Scalable Production of 19-Demethyl-4-Androstenedione via Improved Estrone Route
The pharmaceutical industry constantly seeks more efficient and safer pathways for synthesizing critical steroid intermediates, particularly 19-demethyl-4-androstenedione, a pivotal precursor for drugs like Norethisterone and Nandrolone Phenpropionate. Patent CN102675393B introduces a groundbreaking method that fundamentally shifts the production paradigm from hazardous decarboxylation techniques to a streamlined, high-yield sequence starting from estrone. This innovation addresses long-standing issues regarding toxicity and low conversion rates inherent in legacy processes. By replacing carcinogenic methylating agents with benign dimethyl carbonate and optimizing the reduction steps, this technology offers a robust solution for modern API manufacturing. The strategic integration of etherification, ketal protection, Birch reduction, and hydrolysis creates a cohesive workflow that maximizes atom economy while minimizing hazardous waste generation. For global supply chains, this represents a significant opportunity to secure a more reliable and environmentally compliant source of high-value hormonal intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of 19-demethyl-4-androstenedione has relied heavily on decarboxylation reactions, which suffer from inherent inefficiencies and safety concerns. Traditional protocols often involve the oxidation of 4-androstene-19-methyl alcohol-3,17-diketone using chromic acid-sulfuric acid mixtures, followed by thermal decarboxylation in ethanol-hydrochloric acid or pyridine. These methods typically struggle with substrate conversion issues, resulting in overall yields that hover around merely 60%, which is economically suboptimal for large-scale operations. Furthermore, alternative addition-elimination routes reported in prior art often yield even lower results, approximately 50%, due to complex purification requirements and side reactions. Beyond yield limitations, the conventional methylation of phenolic groups frequently employs dimethyl sulfate or methyl iodide, both of which are classified as high-toxicity carcinogens. The handling of these reagents necessitates stringent safety protocols and expensive waste treatment systems, creating a substantial burden on operational costs and environmental compliance.
The Novel Approach
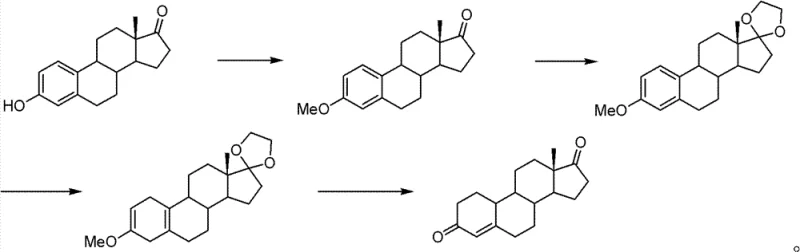
In stark contrast, the methodology disclosed in CN102675393B presents a sophisticated four-step synthesis that dramatically enhances both safety and efficiency. The process initiates with an improved etherification reaction where estrone is treated with dimethyl carbonate and potassium carbonate, eliminating the need for toxic methylating agents entirely. This is followed by a ketalization step to protect the C17 carbonyl, ensuring selectivity during the subsequent reduction phase. The core transformation involves a Birch reduction using metallic lithium in liquid ammonia, which effectively reduces the aromatic A-ring without compromising the structural integrity of the steroid backbone. Finally, a mild hydrolysis step removes the protecting groups to reveal the target 19-demethyl-4-androstenedione. This route achieves a remarkable total recovery rate between 80% and 86%, representing a substantial improvement over traditional decarboxylation techniques. The use of inexpensive inorganic bases and non-toxic organic carbonates not only lowers raw material costs but also simplifies the downstream processing requirements.
Mechanistic Insights into Green Etherification and Birch Reduction
The cornerstone of this technological advancement lies in the optimization of the initial etherification step, which traditionally acts as a bottleneck for both safety and yield. By utilizing dimethyl carbonate (DMC) as the methylating agent in conjunction with potassium carbonate in a high-boiling polar solvent like DMF, the reaction achieves conversion rates exceeding 99%. The mechanism involves the nucleophilic attack of the phenoxide ion, generated in situ by the weak base, on the methyl carbon of the carbonate. This transesterification-like process releases methanol and carbon dioxide as benign byproducts, avoiding the formation of hazardous alkyl halides. Screening of solvents revealed that DMF is superior to crown ether-based systems, pushing the yield from a mediocre 65% to over 97%. This specific solvent choice stabilizes the transition state and enhances the solubility of the inorganic base, facilitating a homogeneous reaction environment that is crucial for consistent batch-to-batch quality in industrial settings.

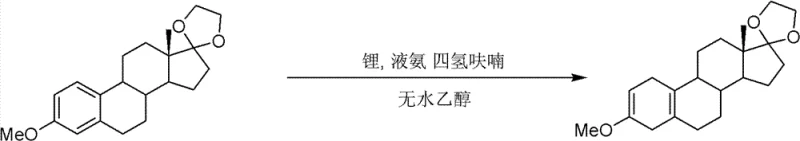
Following protection, the process employs a classic yet highly effective Birch reduction to aromatize the A-ring, a critical step for generating the 19-nor steroid skeleton. In this stage, metallic lithium dissolves in liquid ammonia to generate solvated electrons, which act as powerful reducing agents. The ketal-protected intermediate is introduced in a tetrahydrofuran mixture, allowing for controlled electron transfer to the aromatic system. The reaction temperature is meticulously maintained between -78°C and -40°C to prevent over-reduction or side reactions. Upon quenching with anhydrous ethanol and acetic acid, the dihydro-intermediate is formed with high stereoselectivity. The subsequent acid hydrolysis serves a dual purpose: it cleaves the ethylene ketal to restore the C17 ketone and simultaneously demethylates the phenolic ether to regenerate the conjugated enone system at the C3 position. This tandem deprotection strategy streamlines the synthesis, reducing the total number of unit operations required to reach the final API intermediate.
How to Synthesize 19-Demethyl-4-Androstenedione Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for reproducing these high-yield results in a pilot or production plant. The process begins with the precise mixing of estrone, potassium carbonate, and dimethyl carbonate in DMF, heated to 100-150°C for 10 to 24 hours to ensure complete etherification. Following isolation of the etherate, the material undergoes ketalization with ethylene glycol and triethyl orthoformate catalyzed by boron trifluoride diethyl etherate at ambient temperatures. The resulting ketal is then subjected to the cryogenic Birch reduction conditions described previously. Finally, the crude Birch product is refluxed in methanol with hydrochloric acid to effect hydrolysis. Detailed standardized operating procedures regarding stoichiometry, solvent volumes, and workup techniques are essential for maintaining the reported purity levels above 97%. For a comprehensive guide on the specific molar ratios and quenching protocols, please refer to the structured synthesis guide below.
- Perform etherification of estrone using dimethyl carbonate and potassium carbonate in a high-boiling polar solvent like DMF at 100-150°C.
- Protect the C17 ketone group via ketalization using ethylene glycol and triethyl orthoformate with boron trifluoride diethyl etherate catalyst.
- Execute Birch reduction using metallic lithium in liquid ammonia and tetrahydrofuran to reduce the aromatic A-ring.
- Conduct acid hydrolysis in methanol to remove the ketal protecting group and demethylate the phenolic ether, yielding the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented process translates into tangible strategic benefits beyond mere chemical yield. The substitution of hazardous reagents like methyl iodide with dimethyl carbonate drastically reduces the regulatory burden associated with storing and transporting controlled substances. This shift minimizes the risk of supply chain disruptions caused by strict environmental regulations or safety audits. Furthermore, the use of potassium carbonate as a base instead of expensive organic bases like tetramethylguanidine significantly lowers the direct material costs. The elimination of toxic byproducts simplifies waste disposal protocols, leading to substantial cost savings in environmental compliance and effluent treatment. These factors combined create a more resilient and cost-effective supply chain for critical steroid intermediates.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the replacement of high-cost, high-toxicity reagents with commodity chemicals. By utilizing dimethyl carbonate and potassium carbonate, manufacturers can avoid the premium pricing associated with specialized alkylating agents and expensive organic bases. Additionally, the high conversion rates mean less raw material is wasted, and the simplified purification steps reduce solvent consumption and energy usage during distillation and crystallization. The overall effect is a leaner manufacturing process with a significantly reduced cost of goods sold, allowing for more competitive pricing in the global API market without sacrificing margin.
- Enhanced Supply Chain Reliability: Sourcing hazardous chemicals like dimethyl sulfate often involves complex logistics and limited supplier pools due to their dual-use nature and toxicity. In contrast, dimethyl carbonate and estrone are widely available bulk chemicals with stable supply chains. This accessibility ensures that production schedules are less likely to be impacted by raw material shortages or shipping restrictions. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in reagent quality, further enhancing the reliability of supply for downstream pharmaceutical customers who depend on consistent intermediate availability for their own formulation timelines.
- Scalability and Environmental Compliance: From a sustainability perspective, this route aligns perfectly with modern green chemistry principles. The absence of heavy metal oxidants like chromic acid eliminates the need for costly heavy metal scavenging and disposal, which is a major pain point in steroid synthesis. The process generates primarily benign byproducts such as methanol and carbon dioxide, which are easier to manage and recycle. This environmental profile facilitates easier permitting for new production facilities and reduces the risk of fines or shutdowns due to environmental non-compliance. The scalability is proven by the high yields maintained even at larger scales, making it suitable for meeting the demands of blockbuster hormone therapies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. They are derived directly from the comparative data and experimental examples provided in the patent documentation. Understanding these nuances is critical for R&D teams evaluating technology transfer and for procurement specialists assessing vendor capabilities. The answers highlight the specific advantages in terms of purity, safety, and operational efficiency that distinguish this method from legacy technologies currently in use.
Q: How does this patent improve safety compared to traditional methylation methods?
A: Traditional methods often utilize highly toxic and carcinogenic reagents such as dimethyl sulfate or methyl iodide for phenolic methylation. This patented process replaces those hazardous chemicals with dimethyl carbonate, a non-toxic alternative, and uses inexpensive potassium carbonate as the base, significantly reducing environmental hazards and operator health risks.
Q: What is the overall yield advantage of this estrone-based route?
A: While conventional decarboxylation methods typically achieve yields around 60%, this improved four-step synthesis starting from estrone achieves a total recovery rate between 80% and 86%. Each individual step maintains a high conversion rate, with the etherification step alone exceeding 97% yield when optimized with DMF solvent.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the process is specifically designed for scalability. It utilizes readily available raw materials like estrone and avoids complex purification steps associated with heavy metal catalysts. The use of standard solvents like DMF and methanol, along with robust reaction conditions, facilitates easy scale-up from laboratory to commercial tonnage production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 19-Demethyl-4-Androstenedione Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving hormonal therapies. Our technical team has extensively analyzed the pathway described in CN102675393B and possesses the expertise to implement this green chemistry approach at an industrial scale. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of 19-demethyl-4-androstenedione meets the highest pharmacopeial standards. We are committed to delivering not just a chemical product, but a reliable partnership that supports your long-term commercial goals.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific project requirements. By leveraging our manufacturing capabilities, you can access a Customized Cost-Saving Analysis tailored to your volume needs. We encourage potential partners to contact us directly to request specific COA data and route feasibility assessments. Let us help you secure a sustainable and cost-effective supply chain for your steroid API projects, ensuring continuity and quality from gram scale to commercial tonnage.
