Scalable Asymmetric Synthesis of Etazocine Intermediates for Commercial Pharmaceutical Manufacturing
Introduction to Advanced Etazocine Intermediate Manufacturing
The pharmaceutical industry continuously seeks robust synthetic pathways for potent analgesics like Etazocine Hydrobromide, a kappa-opioid receptor agonist developed for managing severe postoperative and cancer pain. Patent CN110563605B introduces a groundbreaking methodology for synthesizing key intermediates, specifically compounds 4 through 8, which serve as the structural backbone for the final active pharmaceutical ingredient. This innovation addresses critical bottlenecks in traditional manufacturing by replacing hazardous stoichiometric reactions with catalytic asymmetric processes. The disclosed technology leverages chiral phase transfer catalysis to establish stereocenters with high fidelity, ensuring that the resulting intermediates possess the requisite optical purity for biological efficacy without the need for wasteful resolution steps. For global supply chain stakeholders, this represents a pivotal shift towards more sustainable and economically viable production models.
Furthermore, the strategic implementation of this synthetic route allows for significant optimization in process safety and environmental compliance. By circumventing the use of highly reactive organometallic reagents typically employed in legacy methods, manufacturers can mitigate risks associated with exothermic runaways and pyrophoric materials. The patent details a sequence that transitions smoothly from simple starting materials to complex bridged morphinan structures, demonstrating a clear understanding of industrial scalability. This report analyzes the technical merits of this approach, providing R&D directors and procurement officers with the insights needed to evaluate its potential for integration into existing supply chains for high-purity opioid intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
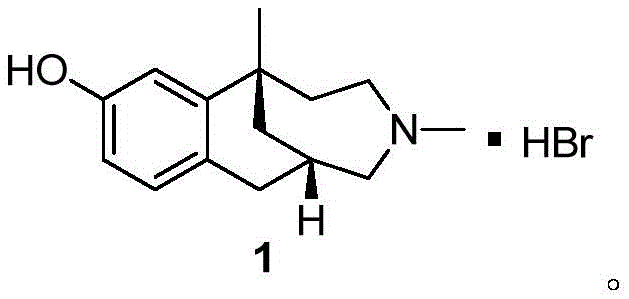
Historically, the industrial synthesis of Etazocine Hydrobromide has relied heavily on routes that involve classical Grignard reactions followed by chiral resolution, as exemplified by earlier Japanese patents such as JP59130872. These conventional pathways are inherently inefficient because they generate racemic mixtures that require subsequent separation using resolving agents like L-tartaric acid. This resolution step inevitably discards nearly half of the synthesized material as unwanted isomers, leading to poor atom economy and inflated raw material costs. Additionally, the reliance on Grignard reagents necessitates strict anhydrous conditions and often cryogenic temperatures to control reactivity, which imposes heavy energy burdens and requires specialized reactor infrastructure that increases capital expenditure.
Beyond the economic inefficiencies, the traditional multi-step sequences often involve harsh oxidizing agents and protection-deprotection strategies that complicate the workflow. For instance, the use of chromium-based oxidants in older routes poses significant environmental and health hazards due to the toxicity of hexavalent chromium, creating substantial waste treatment challenges for manufacturing facilities. The cumulative effect of these long, cumbersome synthetic lines is a lower overall yield and a higher risk of batch-to-batch variability. For procurement managers, these factors translate into volatile pricing and potential supply disruptions, as the complexity of the process makes it difficult to ramp up production quickly in response to market demand fluctuations.
The Novel Approach
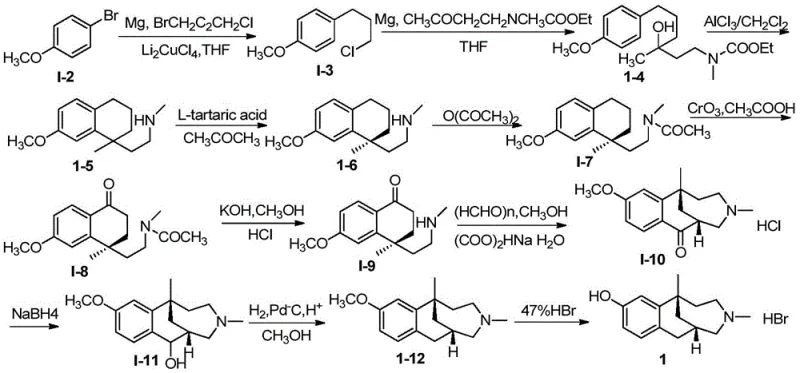
In stark contrast, the methodology disclosed in CN110563605B utilizes a convergent strategy centered on asymmetric phase transfer catalysis to construct the quaternary carbon center with high stereocontrol. This novel approach initiates with the alkylation of a tetralone derivative using a chiral quaternary ammonium salt catalyst, which effectively differentiates between enantiotopic faces of the substrate under mild basic conditions. By establishing chirality at the very beginning of the synthesis, the entire downstream process benefits from high optical purity, eliminating the need for resolution and effectively doubling the theoretical yield compared to racemic routes. The reaction conditions are notably温和 (mild), often proceeding at 0°C to room temperature in common organic solvents like toluene or chlorobenzene, which simplifies process engineering requirements.
The subsequent transformations, including reduction, dehydration, and oxidation, are optimized to maintain the integrity of the chiral center while building the necessary functional groups for ring closure. The use of reagents such as DDQ for oxidation and Raney-Ni for hydrogenation reflects a preference for selective transformations that minimize side reactions and impurity formation. This streamlined pathway not only reduces the total number of unit operations but also enhances the robustness of the manufacturing process. For supply chain heads, this means a more reliable source of intermediates with consistent quality attributes, reducing the need for extensive incoming quality control testing and facilitating smoother integration into final API synthesis workflows.
Mechanistic Insights into Phase Transfer Catalyzed Asymmetric Alkylation
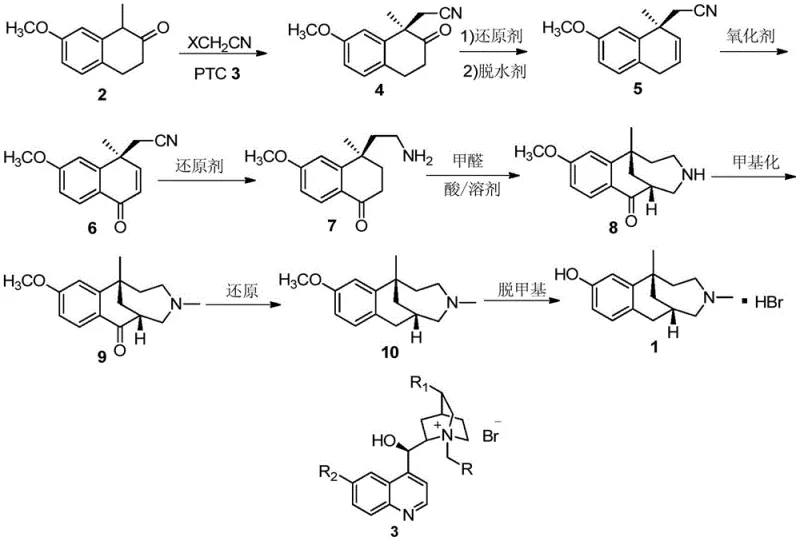
The cornerstone of this innovative synthesis is the enantioselective alkylation of 1-methyl-7-methoxy-2-tetralone (Compound 2) with haloacetonitriles, mediated by a chiral phase transfer catalyst (Compound 3). Mechanistically, this reaction operates at the interface of two immiscible phases: an aqueous base layer and an organic solvent layer containing the substrate and alkylating agent. The chiral catalyst, typically a cinchona alkaloid derivative, shuttles the deprotonated enolate of the tetralone from the aqueous phase into the organic phase as a tight ion pair. Within the chiral environment of the catalyst's quaternary nitrogen center, the enolate is shielded on one face, forcing the incoming electrophile (the haloacetonitrile) to attack from the less hindered side. This steric guidance is what dictates the formation of the desired (R)-enantiomer with high enantiomeric excess, often exceeding 99% after recrystallization.
Controlling impurities in this step is critical for downstream success, and the patent highlights the importance of catalyst structure, specifically the substituents on the benzyl and quinoline moieties (R, R1, R2 groups). Variations in these groups modulate the steric bulk and electronic properties of the catalyst, fine-tuning the selectivity and reaction rate. For R&D directors, understanding this structure-activity relationship is vital for troubleshooting; for instance, switching from a benzyl group to a trifluoromethylbenzyl group on the catalyst can significantly enhance solubility and selectivity in different solvent systems. Furthermore, the choice of base concentration (25%-50% NaOH or KOH) influences the equilibrium of enolate formation, ensuring that the reaction proceeds without excessive hydrolysis of the nitrile group, which is a common side reaction in alkaline media.
How to Synthesize Etazocine Intermediates Efficiently
The execution of this synthetic pathway requires precise control over reaction parameters to maximize yield and optical purity. The process begins with the preparation of the key chiral intermediate 4, followed by a series of functional group manipulations that build complexity while preserving stereochemistry. Operators must pay close attention to temperature control during the alkylation phase and the stoichiometry of reducing agents in subsequent steps to prevent over-reduction or epimerization. The patent provides detailed experimental protocols that serve as a baseline for process development teams aiming to adapt this chemistry for pilot plant operations. Detailed standardized synthesis steps are provided in the guide below.
- Perform asymmetric alkylation of compound 2 with haloacetonitrile using a chiral phase transfer catalyst to form intermediate 4.
- Execute reduction and dehydration sequences to convert intermediate 4 into the dihydronaphthyl acetonitrile derivative 5.
- Complete the synthesis via oxidation, hydrogenation, Mannich condensation, and final demethylation to yield Etazocine Hydrobromide.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this novel synthetic route offers profound commercial benefits that extend beyond simple chemical yield improvements. For procurement managers, the elimination of chiral resolution steps translates directly into substantial cost savings by removing the need for expensive resolving agents and the associated loss of 50% of the material mass. This improvement in atom economy means that less raw material is required to produce the same amount of final product, effectively lowering the cost of goods sold (COGS). Additionally, the avoidance of hazardous Grignard reagents reduces the logistical burden and safety costs associated with handling pyrophoric substances, allowing for more flexible sourcing of starting materials without stringent special transport requirements.
- Cost Reduction in Manufacturing: The streamlined process significantly lowers operational expenditures by reducing the total number of reaction steps and eliminating the need for cryogenic cooling systems. By operating at near-ambient temperatures and using common solvents like toluene and methanol, energy consumption is drastically reduced compared to traditional low-temperature Grignard processes. Furthermore, the high selectivity of the phase transfer catalyst minimizes the formation of by-products, which simplifies purification workflows and reduces the volume of solvent and silica gel required for chromatography, leading to a leaner and more cost-effective manufacturing footprint.
- Enhanced Supply Chain Reliability: The robustness of this chemistry ensures consistent batch quality, which is essential for maintaining uninterrupted supply to downstream API manufacturers. Because the process does not rely on scarce or highly regulated reagents, the risk of supply chain disruption due to raw material shortages is minimized. The ability to achieve high chiral purity directly from the reaction mixture reduces the lead time for quality assurance testing, allowing for faster release of intermediates. This reliability is crucial for pharmaceutical companies managing tight production schedules for pain management medications where inventory buffers are often kept low.
- Scalability and Environmental Compliance: From an environmental perspective, this route aligns with green chemistry principles by avoiding toxic heavy metal oxidants like chromium trioxide in favor of safer alternatives like DDQ or catalytic oxidation methods. The reduction in hazardous waste generation simplifies effluent treatment and lowers compliance costs related to environmental regulations. Moreover, the process is inherently scalable; the use of heterogeneous catalysts like Raney-Ni and Pd/C in later steps facilitates easy filtration and recycling, making the transition from kilogram-scale laboratory synthesis to multi-ton commercial production seamless and technically feasible without major process redesigns.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis. These answers are derived directly from the experimental data and claims within CN110563605B, providing clarity on the feasibility and advantages of the technology. Understanding these details helps stakeholders make informed decisions about adopting this route for their specific manufacturing needs.
Q: What is the primary advantage of the PTC method over traditional Grignard routes?
A: The Phase Transfer Catalysis (PTC) method eliminates the need for hazardous Grignard reagents and cryogenic conditions, significantly improving operational safety and reducing energy consumption while achieving high enantiomeric excess without resolution steps.
Q: How does this process improve atom economy for Etazocine production?
A: By utilizing asymmetric synthesis directly rather than racemic synthesis followed by chiral resolution, the process prevents the discard of 50% of unwanted isomers, thereby maximizing raw material utilization and minimizing waste disposal costs.
Q: Is this synthetic route suitable for large-scale industrial production?
A: Yes, the patent explicitly states the method uses mild reaction conditions and common solvents like toluene and methanol, avoiding specialized high-pressure equipment for most steps, which facilitates straightforward scale-up from laboratory to commercial tonnage.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Etazocine Intermediate Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom pharmaceutical manufacturing, possessing the technical expertise to translate complex patent methodologies like CN110563605B into commercial reality. Our team of process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to reactor train is managed with precision. We adhere to stringent purity specifications and utilize rigorous QC labs equipped with advanced chiral HPLC and NMR capabilities to guarantee that every batch of Etazocine intermediate meets the highest international standards for optical purity and chemical identity.
We invite global pharmaceutical partners to collaborate with us to leverage this cost-effective and scalable synthesis route. By engaging our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain for this critical analgesic intermediate is both secure and economically optimized for the long term.
