Optimizing Arbekacin Production: A High-Yield Route for Pharmaceutical Intermediates
Introduction to Advanced Aminoglycoside Synthesis
The global demand for effective antibiotics against methicillin-resistant Staphylococcus aureus (MRSA) has placed Arbekacin, a semi-synthetic aminoglycoside, at the forefront of pharmaceutical research. However, the commercial viability of this potent drug has historically been constrained by complex synthesis routes and low overall yields. Patent CN101575354B introduces a transformative methodology for synthesizing Arbekacin and its critical intermediate, Dibekacin, addressing these longstanding bottlenecks. By utilizing Kanamycin B as a readily available starting material, this novel approach streamlines the production pathway through optimized protection strategies and a refined elimination process. This technological leap not only enhances the purity profile required for regulatory compliance but also fundamentally alters the cost structure of manufacturing high-purity pharmaceutical intermediates.
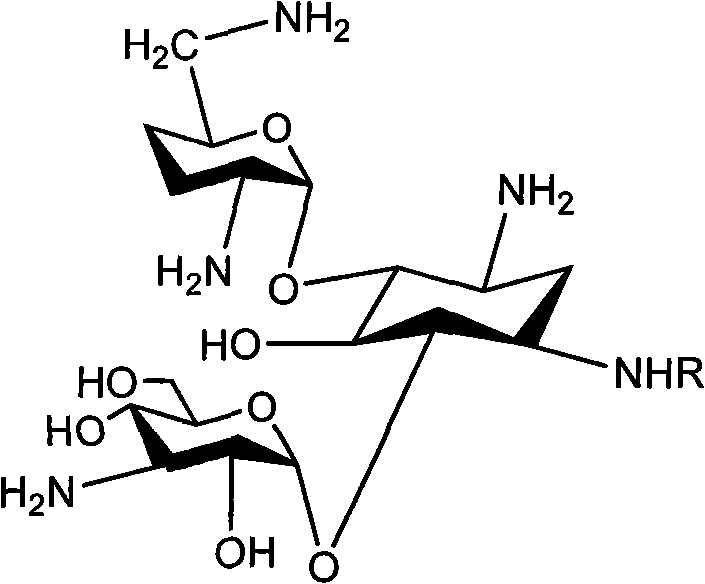
For procurement specialists and supply chain directors, the implications of this patent extend beyond mere chemical curiosity. The ability to produce Dibekacin with a total recovery rate significantly higher than the industry standard of 15% represents a tangible opportunity for cost reduction in API manufacturing. Furthermore, the strategic replacement of hazardous reagents and the simplification of purification steps directly correlate to improved supply chain reliability and reduced environmental liability. As we delve into the technical specifics, it becomes clear that this synthesis route offers a robust framework for the commercial scale-up of complex aminoglycoside derivatives, positioning manufacturers to meet the rigorous demands of the global antimicrobial market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Dibekacin and Arbekacin has been plagued by inefficiencies that hinder large-scale production. Prior art, such as the methods disclosed in English Patent GB1537905, involves an excessive number of reaction steps, leading to a cumulative yield that is economically unsustainable. Specifically, the final deprotection steps in these traditional routes often suffer from extremely low conversion rates, sometimes dropping the total recovery to as low as 15%, despite individual step yields appearing acceptable. Moreover, alternative methods like those in US Patents 4156078 and 4169939 rely heavily on the use of sodium iodide and zinc powder in stoichiometric excesses that are environmentally detrimental. In these legacy processes, zinc powder is often used in amounts 5 to 10 times the weight of the product, creating severe challenges in waste management and downstream processing due to emulsification during extraction.
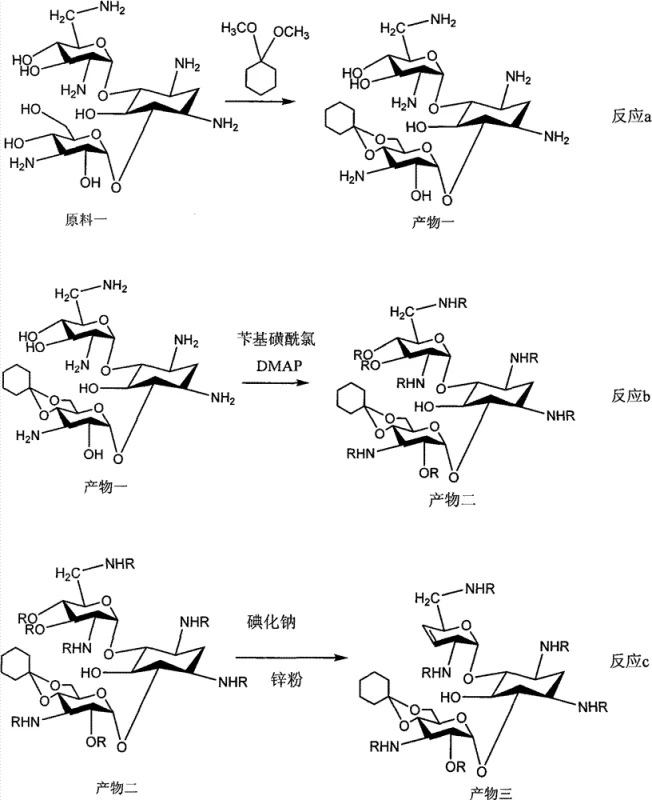
The Novel Approach
The methodology outlined in CN101575354B dismantles these barriers through a series of strategic chemical innovations. The new route initiates with the formation of 4',6'-O-cyclohexylidene kanamycin, establishing a robust protecting group strategy that withstands subsequent harsh reaction conditions. A pivotal advancement is observed in the elimination step, where the consumption of sodium iodide is reduced to 4 to 6 times the molar amount and zinc powder is drastically cut to 0.04 to 0.2 times the product weight. This optimization not only mitigates the risk of heavy metal contamination but also elevates the yield of this critical step to approximately 97% without the need for intermediate purification. Additionally, the introduction of di-tert-butyl dicarbonate (Boc anhydride) for amino protection replaces more expensive and difficult-to-remove groups, simplifying the overall workflow and enhancing the feasibility of industrial application.
Mechanistic Insights into Selective Protection and Elimination
The core of this synthesis lies in the precise control of regioselectivity during the protection and elimination phases. The process begins with the acid-catalyzed condensation of Kanamycin B with 1,1-dimethoxycyclohexane, selectively masking the 4' and 6' hydroxyl groups. This cyclohexylidene acetal is crucial as it prevents unwanted side reactions at these positions during the subsequent sulfonylation. The sulfonylation step, conducted at low temperatures between -20°C and 0°C using benzylsulfonyl chloride and DMAP, ensures that the amino groups are protected without degrading the sensitive glycosidic bonds. This meticulous control over reaction conditions is vital for maintaining the integrity of the aminoglycoside core, ensuring that the final product meets the stringent purity specifications required for pharmaceutical applications.
Following protection, the mechanism shifts to the formation of the 3',4'-double bond, a defining feature of Dibekacin. The use of sodium iodide in DMF facilitates a nucleophilic substitution, which is immediately followed by zinc-mediated elimination. Unlike prior art where excess zinc causes emulsification and difficult workups, the optimized stoichiometry in this patent allows for a clean reaction profile. The subsequent acidic hydrolysis removes the cyclohexylidene group, exposing the hydroxyls for the final hydrogenation step. The use of platinum oxide as a catalyst for hydrogenation under mild conditions ensures the stereoselective reduction of the double bond to form the saturated 3',4'-dideoxy structure of Dibekacin. This mechanistic precision minimizes the formation of impurities, thereby reducing the burden on downstream purification processes and improving the overall impurity profile of the active pharmaceutical ingredient.
How to Synthesize Dibekacin Efficiently
The synthesis of Dibekacin via this patented route involves a sequence of ten distinct chemical transformations, each optimized for maximum yield and minimal waste. The process leverages common industrial solvents and reagents, making it accessible for established chemical manufacturing facilities. From the initial protection of Kanamycin B to the final catalytic hydrogenation, every step has been engineered to avoid the pitfalls of previous methods, such as low yields and difficult separations. For R&D teams looking to implement this technology, the detailed reaction conditions provided in the patent offer a clear roadmap for replication and scale-up. The following guide outlines the standardized synthesis steps derived from the patent data, serving as a foundational reference for process development.
- Protect Kanamycin B with cyclohexylidene and sulfonyl groups to enable selective modification.
- Perform sodium iodide substitution and zinc-mediated elimination to form the 3',4'-double bond.
- Execute deprotection, Birch reduction, and catalytic hydrogenation to finalize the Dibekacin core.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers compelling economic and operational advantages. The primary driver of value is the substantial increase in overall yield, which directly translates to a lower cost of goods sold (COGS) per kilogram of finished intermediate. By improving the total recovery of purified Dibekacin from the historical baseline of 15% to a range of 25% to 35%, manufacturers can significantly reduce the amount of raw Kanamycin B required to meet production targets. This efficiency gain is compounded by the reduction in reagent costs, particularly the drastic decrease in zinc powder and sodium iodide usage, which lowers both material expenses and the associated costs of hazardous waste disposal. These factors combined create a more resilient and cost-effective supply chain for aminoglycoside intermediates.
- Cost Reduction in Manufacturing: The optimized stoichiometry of reagents, specifically the reduction of zinc powder usage from 5-10 equivalents to merely 0.04-0.2 equivalents, results in a dramatic decrease in raw material consumption. This shift eliminates the need for extensive wastewater treatment protocols associated with heavy metal removal, thereby lowering operational expenditures related to environmental compliance. Furthermore, the high yield of the elimination step (approximately 97%) reduces the loss of valuable intermediates, ensuring that more input material is converted into saleable product. The use of cost-effective protecting groups like Boc anhydride further drives down the direct material costs compared to proprietary or complex esters used in older methods.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable reagents such as Kanamycin B, p-toluenesulfonic acid, and di-tert-butyl dicarbonate ensures a stable supply chain不受制于 exotic or hard-to-source chemicals. The simplification of the purification process, where certain intermediates do not require rigorous purification before the next step, shortens the overall production cycle time. This agility allows manufacturers to respond more quickly to fluctuations in market demand for Arbekacin and its precursors. Additionally, the robustness of the reaction conditions, which tolerate a broader range of parameters without significant yield loss, reduces the risk of batch failures and ensures consistent delivery schedules to downstream API producers.
- Scalability and Environmental Compliance: The process is designed with industrial scalability in mind, avoiding the use of dangerous solvents like ether in critical steps and minimizing the generation of toxic byproducts. The significant reduction in zinc and iodine waste aligns with increasingly strict global environmental regulations, reducing the regulatory burden on manufacturing sites. The ability to perform key reactions like the elimination step without complex workup procedures facilitates easier scale-up from pilot plant to commercial production volumes. This environmental and operational efficiency makes the technology attractive for long-term investment, ensuring that production capacity can be expanded sustainably to meet growing global antibiotic needs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of Arbekacin and Dibekacin as described in the patent literature. These insights are derived directly from the experimental data and comparative analysis provided in CN101575354B, offering clarity on the practical implementation of this technology. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this route for their own manufacturing pipelines. The answers reflect the specific improvements in yield, safety, and efficiency that distinguish this method from prior art.
Q: How does the new synthesis method improve upon prior art yields?
A: The patented method increases the total recovery of purified Dibekacin from approximately 15% in conventional routes to between 25% and 35%, significantly enhancing material efficiency.
Q: What are the environmental benefits of this specific Arbekacin route?
A:
Q: Is this process scalable for commercial API manufacturing?
A: Yes, the method utilizes common reagents like Boc anhydride and avoids complex purification steps in intermediate stages, making it highly suitable for large-scale industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Arbekacin Supplier
The synthesis of Arbekacin and its intermediate Dibekacin represents a critical capability in the modern pharmaceutical landscape, particularly for combating resistant bacterial strains. NINGBO INNO PHARMCHEM stands ready to leverage this advanced technology to support your drug development and commercialization goals. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of Arbekacin intermediate meets the highest international standards for safety and efficacy.
We invite you to collaborate with us to explore the full potential of this optimized synthesis route. Our technical procurement team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this efficient process can enhance your bottom line. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in aminoglycoside chemistry can accelerate your path to market while optimizing your supply chain economics.
