Advanced Ring-Opening Technology for Pyranose Derivatives: Scalable Synthesis for Pharma Intermediates
The landscape of carbohydrate chemistry is undergoing a significant transformation with the introduction of novel synthetic methodologies that prioritize both efficiency and environmental sustainability. Patent CN114671849A represents a pivotal advancement in this field, disclosing a sophisticated ring-opening method for pyranose rings that overcomes the limitations of classical approaches. This technology enables the conversion of methyl-alpha-D-glucopyranoside into valuable open-chain derivatives through a unique strain-induced mechanism. By leveraging specific sulfonate esterification strategies, the process achieves high yields under remarkably mild conditions, avoiding the extreme temperatures and hazardous reagents often associated with glycoside manipulation. For industry leaders seeking a reliable pharmaceutical intermediate supplier, this innovation offers a robust pathway to complex sugar derivatives essential for drug development.
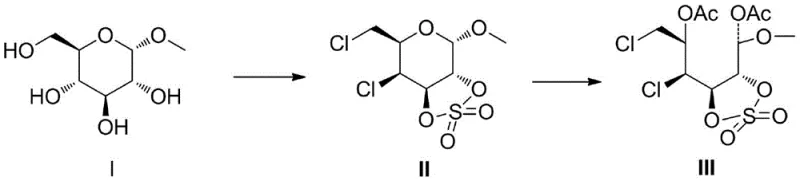
The strategic value of this patent lies in its ability to generate versatile intermediates applicable in the synthesis of glycals, tumor diagnostic reagents, and targeted therapeutics. The core innovation involves transforming the stable pyranose skeleton into a reactive open-chain structure without compromising the stereochemical integrity of adjacent centers. This capability is crucial for the commercial scale-up of complex pharmaceutical intermediates, where consistency and purity are paramount. The method described provides a clear competitive edge by streamlining the synthesis of precursors used in high-value applications such as 18F-FDG production, thereby addressing critical bottlenecks in the supply chain for radiopharmaceuticals and oncology drugs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the opening of oxygen-containing pyranose rings has relied heavily on reactions with thiols and thiophenols or the use of strong Lewis acid catalysts. These conventional techniques present substantial drawbacks that hinder their adoption in modern, green manufacturing environments. A primary concern is the generation of large quantities of gases with pungent and irritating odors, which pose significant challenges for waste gas treatment and worker safety compliance. Furthermore, these traditional methods often necessitate harsh reaction conditions, including elevated temperatures and strictly anhydrous environments, which increase energy consumption and operational complexity. The reliance on specialized catalysts can also introduce difficulties in downstream purification, potentially leading to metal contamination issues that are unacceptable in high-purity pharmaceutical intermediate production. These factors collectively contribute to higher production costs and extended lead times, making conventional routes less attractive for large-scale industrial applications.
The Novel Approach
In stark contrast, the methodology disclosed in CN114671849A introduces a paradigm shift by utilizing a strain-release strategy to facilitate ring opening. The process begins with the selective sulfonation of the 2,3-positions of the glucopyranoside, which induces a twisted conformation in the ring structure. This structural distortion dramatically increases the internal ring tension, effectively priming the molecule for cleavage. Consequently, the ring opening can be achieved under mild acidic conditions at room temperature, eliminating the need for aggressive thermal input or noxious sulfur-based reagents. This approach not only enhances the environmental profile of the synthesis but also significantly improves the overall yield and selectivity. For procurement teams focused on cost reduction in pharmaceutical intermediate manufacturing, this translates to a more efficient process with lower utility costs and simplified waste management protocols, ensuring a more sustainable and economically viable supply chain.
Mechanistic Insights into Strain-Induced Ring Opening
The mechanistic elegance of this synthesis lies in the precise manipulation of stereoelectronic effects to drive the reaction forward. The initial step involves the formation of a cyclic sulfonate ester at the 2 and 3 positions of the pyranose ring. This modification forces the six-membered ring into a distorted, twisted boat-like conformation that deviates significantly from the stable chair form. This distortion stores potential energy within the molecular framework in the form of torsional strain. When the anomeric center is subsequently exposed to an electrophilic environment, typically generated by acid catalysis in the presence of anhydrides, the system seeks to relieve this strain. The release of ring tension acts as a powerful thermodynamic driving force, lowering the activation energy barrier for the cleavage of the C-O bond at the anomeric position. This results in the formation of an open-chain carbocation intermediate which is rapidly trapped by the nucleophilic anhydride to form the final esterified product.
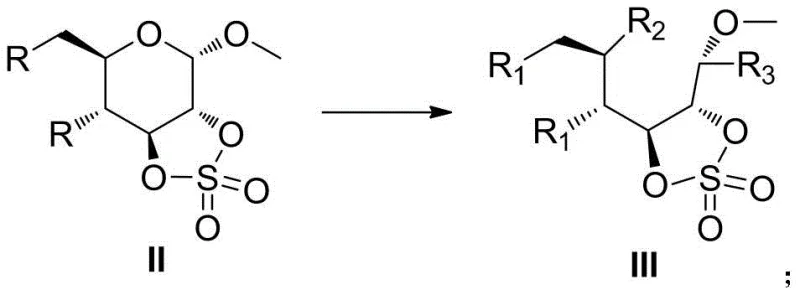
From an impurity control perspective, this mechanism offers distinct advantages over random bond cleavage methods. Because the ring opening is driven by specific geometric constraints and electronic activation at the anomeric carbon, the reaction exhibits high regioselectivity. This minimizes the formation of side products that typically arise from non-specific hydrolysis or elimination reactions. The mild conditions further suppress degradation pathways that might occur at higher temperatures, such as caramelization or polymerization of the sugar backbone. For R&D directors, this implies a cleaner crude reaction profile, which simplifies the purification process and ensures that the final pharmaceutical intermediates meet stringent quality specifications. The ability to control the reaction trajectory through structural design rather than brute-force conditions exemplifies a mature level of process chemistry that is highly desirable for regulatory compliance and consistent batch-to-batch reproducibility.
How to Synthesize Ring-Opened Pyranose Derivatives Efficiently
The synthesis protocol outlined in the patent provides a clear and actionable roadmap for producing these valuable derivatives. The process is divided into two main stages: the preparation of the strained sulfonate intermediate and the subsequent acid-catalyzed ring opening. The first stage requires careful control of stoichiometry and temperature to ensure complete sulfonation without over-reaction. The second stage leverages the inherent instability of the strained intermediate to achieve rapid conversion upon exposure to acid. Detailed operational parameters, including solvent ratios and reaction times, are optimized to maximize yield while maintaining safety. For technical teams looking to implement this technology, the following guide summarizes the critical steps involved in the standardized synthesis procedure.
- Sulfonate the 2,3-positions of methyl-alpha-D-glucopyranoside using sulfonyl chloride and pyridine to form the twisted cyclic sulfonate intermediate.
- Perform the ring-opening reaction by treating the sulfonate intermediate with anhydrides and concentrated sulfuric acid at room temperature.
- Purify the resulting open-chain ester derivative using standard silica gel column chromatography to obtain the high-purity target compound.
Commercial Advantages for Procurement and Supply Chain Teams
The adoption of this novel ring-opening technology presents compelling economic and logistical benefits for organizations managing the supply of complex carbohydrate derivatives. By shifting away from hazardous and odorous reagents, manufacturers can significantly reduce the overhead costs associated with environmental compliance and specialized waste disposal. The mild reaction conditions also allow for the use of standard glass-lined or stainless steel reactors without the need for exotic materials resistant to extreme corrosion or high pressure. This flexibility enhances the agility of the supply chain, allowing for faster turnaround times and the ability to respond quickly to fluctuating market demands. Furthermore, the high yields reported in the patent examples suggest a material-efficient process that maximizes the output from raw materials, directly contributing to improved margins and cost reduction in pharmaceutical intermediate manufacturing.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the reduction in energy consumption due to room temperature operation create a leaner cost structure. The process avoids the need for complex scrubbing systems required for thiol-based methods, thereby lowering capital expenditure for new production lines. Additionally, the high selectivity reduces the loss of valuable starting materials to byproducts, ensuring that the cost of goods sold remains competitive even at smaller scales. This economic efficiency makes the technology accessible for both pilot-scale development and full commercial production.
- Enhanced Supply Chain Reliability: The reagents utilized in this synthesis, such as sulfonyl chloride, acetic anhydride, and common organic solvents, are commodity chemicals with robust global supply chains. This reduces the risk of supply disruptions that can occur with specialized or proprietary catalysts. The simplicity of the workup procedure, involving standard extraction and chromatography, further ensures that production schedules are not delayed by complex purification bottlenecks. For supply chain heads, this reliability translates to reducing lead time for high-purity pharmaceutical intermediates and ensuring consistent availability for downstream drug manufacturing.
- Scalability and Environmental Compliance: The benign nature of the byproducts and the absence of toxic gases make this process inherently safer and easier to scale. Regulatory approval for manufacturing sites is often expedited when the environmental impact is minimized, facilitating faster market entry for new drugs utilizing these intermediates. The process aligns with green chemistry principles by reducing waste generation and energy usage, which is increasingly becoming a requirement for partnerships with major pharmaceutical companies. This alignment ensures long-term viability and sustainability of the supply source.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ring-opening technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation. Understanding these aspects is crucial for evaluating the feasibility of integrating this method into existing production workflows or for sourcing these intermediates from external partners. The answers provide clarity on the operational advantages and the specific chemical transformations involved.
Q: What are the advantages of this ring-opening method over traditional thiol-based methods?
A: Unlike traditional methods that rely on thiols or thiophenols which produce pungent odors and require harsh conditions, this patented method utilizes sulfonate esterification to induce ring strain. This allows the reaction to proceed under mild acidic conditions at room temperature, significantly improving environmental friendliness and operational safety.
Q: Can this method be scaled for commercial production of tumor diagnostic reagents?
A: Yes, the method is designed for scalability. The use of common reagents like acetic anhydride and sulfonyl chloride, combined with room temperature reaction conditions, simplifies the thermal management requirements for large-scale reactors, making it highly suitable for commercial manufacturing of intermediates for tumor diagnostics.
Q: What is the key mechanistic driver for the ring opening in this process?
A: The key driver is the formation of a twisted ring structure via 2,3-sulfonate esterification. This structural modification greatly enhances the internal tension of the pyranose ring. When the anomeric carbon is subjected to electrophilic attack under acidic conditions, this stored tension facilitates the cleavage of the ring bond, leading to efficient ring opening.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Methyl-alpha-D-glucopyranoside Derivative Supplier
As the demand for specialized carbohydrate intermediates continues to grow in the oncology and diagnostic sectors, having a partner with deep technical expertise is essential. NINGBO INNO PHARMCHEM stands at the forefront of this industry, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our commitment to quality is underscored by our stringent purity specifications and rigorous QC labs, ensuring that every batch of methyl-alpha-D-glucopyranoside derivatives meets the exacting standards required for clinical applications. We understand the critical nature of these materials in the drug development timeline and are equipped to handle the complexities of strain-induced synthesis with precision and reliability.
We invite you to collaborate with us to leverage this advanced technology for your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements and quality targets. We encourage potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments. By partnering with NINGBO INNO PHARMCHEM, you secure a supply chain that is not only cost-effective but also technologically superior, ensuring your projects proceed without interruption.
