Advanced Manufacturing of (S)-3-Cyclohexene-1-Carboxylic Acid for FXa Inhibitors
Advanced Manufacturing of (S)-3-Cyclohexene-1-Carboxylic Acid for FXa Inhibitors
The pharmaceutical industry continuously seeks robust, scalable, and cost-effective pathways for producing chiral intermediates essential for next-generation therapeutics. Patent CN102317248A introduces a groundbreaking production method for optically active carboxylic acids, specifically targeting the synthesis of (S)-3-cyclohexene-1-carboxylic acid. This compound serves as a critical building block for potent Factor Xa (FXa) inhibitors, such as Edoxaban, which are vital in the prevention and treatment of thrombotic diseases. The disclosed technology addresses long-standing inefficiencies in chiral resolution by utilizing a novel solvent system comprising aqueous acetone or aqueous ethyl acetate. This innovation not only streamlines the purification process but also integrates a sustainable recycling loop for stereoisomers, marking a significant leap forward in green chemistry and industrial process design for high-value pharmaceutical intermediates.
For R&D Directors and process chemists, the significance of this patent lies in its ability to bypass the limitations of previous synthetic routes. Traditional methods often relied on expensive chiral auxiliaries like D-pantolactone or complex enzymatic hydrolysis reactions that demanded excessive solvent volumes and offered limited scalability. In contrast, the method described in CN102317248A leverages simple diastereomeric salt formation using (R)-alpha-phenylethylamine. By meticulously tuning the water content within organic solvents, the process achieves high optical purity with significantly fewer recrystallization steps compared to prior art. This level of control over crystallization dynamics ensures that the final product meets the stringent purity specifications required for clinical applications, reducing the burden on downstream purification teams.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of (S)-3-cyclohexene-1-carboxylic acid has been plagued by economic and technical bottlenecks that hindered large-scale adoption. Prior art methods, such as those utilizing asymmetric Diels-Alder reactions with D-pantolactone, incurred prohibitive costs due to the high price of the chiral auxiliary and the complexity of its removal post-reaction. Similarly, enzymatic approaches, while selective, suffered from poor volumetric efficiency, requiring massive amounts of solvent to maintain enzyme activity and substrate solubility. Furthermore, conventional optical resolution techniques often necessitated five or more repetitive recrystallization cycles to achieve acceptable enantiomeric excess, leading to substantial yield losses and increased waste generation. These inefficiencies created a fragile supply chain where minor deviations in process parameters could result in batch failures, making it difficult for procurement managers to secure consistent supplies of high-quality intermediates.
The Novel Approach
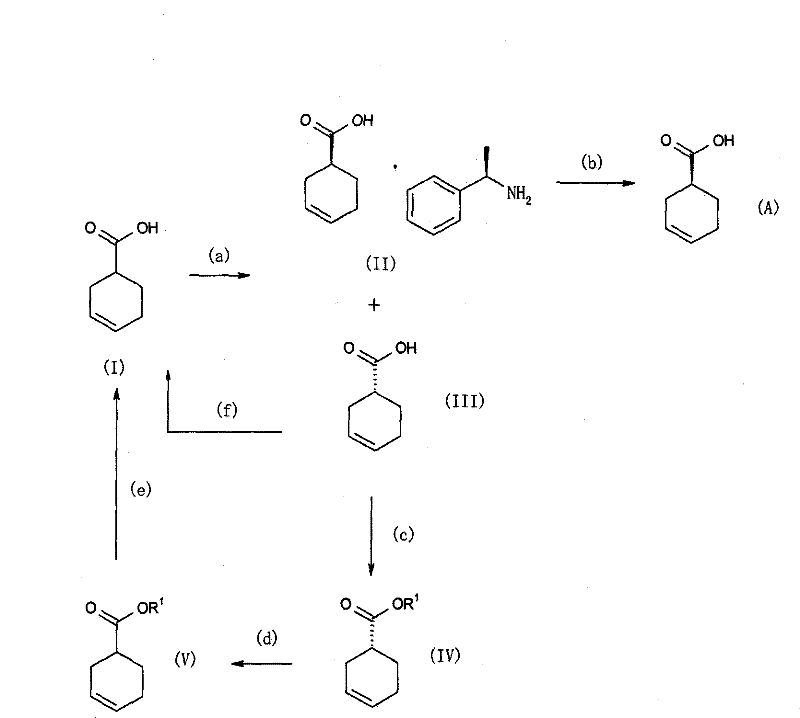
The innovative strategy outlined in this patent fundamentally reshapes the production landscape by introducing a highly efficient resolution and recycling protocol. The core breakthrough involves the use of mixed solvents—specifically water combined with acetone or ethyl acetate—to facilitate the crystallization of the (R)-alpha-phenylethylamine salt of the target acid. This solvent system optimizes the solubility profile, allowing for the precipitation of the desired diastereomer with high purity in as few as two or three recrystallization steps. Moreover, the process incorporates a clever racemization loop where the unwanted (R)-stereoisomer, typically discarded as waste, is chemically converted back into the racemic starting material. This closed-loop system dramatically improves atom economy and reduces raw material consumption, providing a compelling value proposition for supply chain heads focused on sustainability and cost containment.
Mechanistic Insights into Chiral Resolution and Racemization
At the heart of this technology is a sophisticated understanding of diastereomeric salt formation and thermodynamic control during crystallization. The reaction begins with the interaction between racemic 3-cyclohexene-1-carboxylic acid (Compound I) and the chiral base (R)-alpha-phenylethylamine. In the presence of the optimized aqueous solvent system, the solubility difference between the (S)-acid/(R)-amine salt and the (R)-acid/(R)-amine salt is maximized. This allows the less soluble diastereomer to precipitate selectively from the solution. The precise control of water content—ranging from 0.5% to 3.0% in ethyl acetate or 3% to 90% in acetone—is critical, as it modulates the dielectric constant of the medium and influences the hydrogen bonding networks within the crystal lattice. This fine-tuning ensures that impurities and the unwanted enantiomer remain in the mother liquor, thereby enhancing the optical purity of the isolated solid without the need for excessive processing.
Beyond the initial resolution, the patent details a robust mechanism for managing the stereochemical waste stream. The unwanted (R)-3-cyclohexene-1-carboxylic acid (Compound III), which accumulates in the mother liquor, is not discarded but rather subjected to a racemization process. This can be achieved either through esterification followed by base-catalyzed isomerization and hydrolysis or via direct treatment with a strong base like sodium hydride in a polar aprotic solvent such as DMF. These conditions facilitate the abstraction of the acidic alpha-proton adjacent to the carbonyl group, leading to the formation of a planar enolate intermediate. Upon reprotonation, the stereochemical information is lost, regenerating the racemic mixture (Compound I). This regenerated racemate can then be fed back into the resolution step, creating a continuous cycle that theoretically allows for 100% conversion of the starting material into the desired chiral product, a feat rarely achieved in traditional resolution processes.
How to Synthesize (S)-3-Cyclohexene-1-Carboxylic Acid Efficiently
Implementing this synthesis route requires careful attention to solvent preparation and temperature profiling to maximize yield and optical purity. The process begins by dissolving the racemic acid in the pre-mixed aqueous solvent system, followed by the controlled addition of the chiral amine. Maintaining the reaction temperature within the specified range of 0°C to 60°C during the crystallization phase is crucial for controlling crystal growth and preventing the occlusion of impurities. Following the isolation of the salt, the free acid is liberated through acidification with mineral acids like hydrochloric or sulfuric acid. For the recycling portion of the process, the choice of base and solvent for the racemization step must be aligned with the specific ester or acid form being processed to ensure complete isomerization. Detailed standard operating procedures regarding stoichiometry, agitation rates, and filtration temperatures are essential for translating this laboratory-scale success to commercial production.
- React racemic 3-cyclohexene-1-carboxylic acid with (R)-alpha-phenylethylamine in a mixed solvent of water and acetone or water and ethyl acetate to form the diastereomeric salt.
- Purify the resulting salt through controlled recrystallization cycles to achieve high optical purity (greater than 90% de).
- Liberate the free acid using mineral acid treatment and recycle the unwanted (R)-isomer back to the racemic starting material via base-catalyzed isomerization.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented methodology offers transformative benefits for organizations involved in the commercial scale-up of complex pharmaceutical intermediates. The primary advantage lies in the drastic simplification of the purification workflow. By reducing the number of recrystallization steps from five or more down to two or three, manufacturers can significantly lower labor costs, reduce equipment occupancy time, and minimize solvent consumption. This efficiency translates directly into a more competitive cost structure, allowing suppliers to offer better pricing without compromising on quality. Furthermore, the elimination of expensive chiral auxiliaries and enzymes removes volatile cost drivers from the bill of materials, stabilizing the long-term economics of the supply chain and protecting against market fluctuations in specialty reagent prices.
- Cost Reduction in Manufacturing: The implementation of this process leads to substantial cost savings by eliminating the need for costly chiral auxiliaries like D-pantolactone and reducing the reliance on large volumes of solvents associated with enzymatic methods. The ability to recycle the unwanted stereoisomer back into the process effectively doubles the theoretical yield from the same amount of starting racemic material, drastically lowering the raw material cost per kilogram of the final active intermediate. Additionally, the simplified crystallization protocol reduces energy consumption associated with heating and cooling cycles, further contributing to overall operational expenditure reductions.
- Enhanced Supply Chain Reliability: By establishing a closed-loop recycling system for the stereoisomers, manufacturers can mitigate the risks associated with raw material shortages. The process is less dependent on the continuous influx of fresh racemic acid since the waste stream is continuously replenished into the production line. This self-sustaining aspect enhances supply security, ensuring that production schedules are not disrupted by external supply chain bottlenecks. Moreover, the use of common, commodity solvents like acetone and ethyl acetate ensures that solvent availability remains stable, unlike specialized bio-catalytic reagents which may have longer lead times.
- Scalability and Environmental Compliance: The chemistry described relies on standard unit operations such as crystallization, filtration, and distillation, which are easily scalable from pilot plants to multi-ton commercial reactors. This scalability reduces the technical risk during technology transfer. Environmentally, the process aligns with green chemistry principles by minimizing waste generation through the racemization loop and reducing the total solvent load. This results in a lower environmental footprint, simplifying waste disposal compliance and reducing the costs associated with environmental health and safety (EHS) management, making it an attractive option for companies aiming to meet strict sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this production method. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this technology into their existing manufacturing portfolios. The answers provided are derived directly from the technical disclosures within the patent documentation, ensuring accuracy and relevance for decision-makers.
Q: Why is the specific water content in the solvent critical for this resolution process?
A: The patent specifies that controlling water content (e.g., 0.5% to 3.0% in ethyl acetate) is essential for optimizing the solubility differential between the diastereomeric salts, ensuring high yield and optical purity during crystallization.
Q: How does this method handle the unwanted (R)-stereoisomer byproduct?
A: Unlike traditional methods that discard the unwanted isomer, this process includes a racemization step where the (R)-3-cyclohexene-1-carboxylic acid is converted back to the racemic mixture, allowing it to be re-fed into the resolution cycle.
Q: What are the primary advantages over enzymatic hydrolysis methods?
A: This chemical resolution method avoids the large solvent volumes and specialized enzyme handling required in biocatalytic routes, offering a more robust and scalable approach for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-3-Cyclohexene-1-Carboxylic Acid Supplier
As the global demand for anticoagulant therapies continues to rise, securing a dependable source of high-quality intermediates is paramount. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging advanced proprietary technologies like the one described in CN102317248A to deliver superior products. Our facility is equipped with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of multinational pharmaceutical clients. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of (S)-3-cyclohexene-1-carboxylic acid meets the highest international standards for safety and efficacy.
We invite you to collaborate with us to optimize your supply chain for FXa inhibitor production. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our efficient manufacturing processes can reduce your overall procurement costs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us partner with you to drive innovation and efficiency in your drug development pipeline, ensuring a steady supply of critical materials for your life-saving medications.
