Scalable Production of High-Purity Calcium 3-Methyl-2-Oxovalerate for Renal Therapeutics
The pharmaceutical and nutritional industries are constantly seeking robust, scalable pathways for producing critical alpha-keto acid derivatives, particularly for applications in renal therapeutics and sports nutrition. A pivotal advancement in this domain is detailed in patent CN103044238A, which discloses a highly efficient method for preparing calcium 3-methyl-2-oxovalerate, also known as racemic ketoprofen isoleucine calcium. This compound serves as a vital precursor in the synthesis of essential amino acid substitutes used to manage chronic renal insufficiency, offering patients a way to reduce nitrogen load while maintaining nutritional status. The disclosed technology represents a significant leap forward by replacing hazardous, energy-intensive legacy processes with a mild, one-pot synthetic strategy that utilizes inexpensive commodity chemicals. For R&D directors and procurement specialists alike, understanding the nuances of this patent is crucial for securing a reliable pharmaceutical intermediates supplier capable of delivering high-quality materials at a competitive cost structure.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of alpha-keto acid calcium salts has been plagued by significant technical and economic hurdles that hinder efficient commercial scale-up of complex pharmaceutical intermediates. Traditional Route 1, often cited in older literature, relies heavily on Grignard reagents reacting with oxalic acid diesters. While chemically feasible, this approach demands strictly anhydrous conditions, high-purity inert gas atmospheres such as argon, and cryogenic temperatures to control exothermic reactions. These severe reaction conditions not only escalate energy consumption but also introduce substantial safety risks associated with handling pyrophoric reagents on a multi-ton scale. Furthermore, alternative pathways involving glycolylurea intermediates, while avoiding Grignard reagents, suffer from the generation of ammonia and carbonate byproducts during hydrolysis. These volatile byproducts create environmental compliance challenges and necessitate complex waste treatment protocols, thereby inflating the overall cost reduction in API manufacturing efforts and complicating the supply chain logistics for bulk producers.
The Novel Approach
In stark contrast to these cumbersome legacy methods, the novel approach outlined in the patent data leverages a streamlined base-catalyzed condensation mechanism that operates under remarkably mild conditions. By utilizing sodium alkoxide in an alcoholic medium, the process facilitates a rapid transesterification and subsequent aldol condensation without the need for extreme temperatures or pressure. The reaction proceeds efficiently at temperatures not exceeding 25°C, drastically simplifying the thermal management requirements for reactor systems. This methodology eliminates the need for isolating unstable intermediates, effectively collapsing multiple synthetic steps into a cohesive one-pot operation. For supply chain heads, this translates to reduced lead time for high-purity pharmaceutical intermediates, as the simplified workflow minimizes batch cycle times and equipment turnover. Moreover, the avoidance of toxic heavy metals and the use of benign solvents align perfectly with modern green chemistry principles, ensuring that the production facility remains compliant with increasingly stringent environmental regulations while maintaining high throughput.
Mechanistic Insights into Base-Catalyzed Condensation and Salification
The core of this technological breakthrough lies in the elegant mechanistic pathway driven by sodium methoxide catalysis, which orchestrates a sequence of transesterification, condensation, and cyclization events. Initially, diethyl oxalate undergoes rapid transesterification in the methanol solution of sodium methylate to form the corresponding dimethyl ester in situ. Upon the addition of 2-methylbutyraldehyde, the strong base promotes an aldol condensation, generating a reactive enolate species that attacks the carbonyl carbon of the oxalate derivative. This is followed by an intramolecular cyclization to form a transient lactone intermediate, which subsequently eliminates a molecule of methyl formate. The final ring-opening step, triggered by the addition of aqueous alkali, yields the sodium salt of the keto acid. This precise control over the reaction trajectory ensures that side reactions are minimized, leading to a clean crude product profile that requires less aggressive downstream purification.
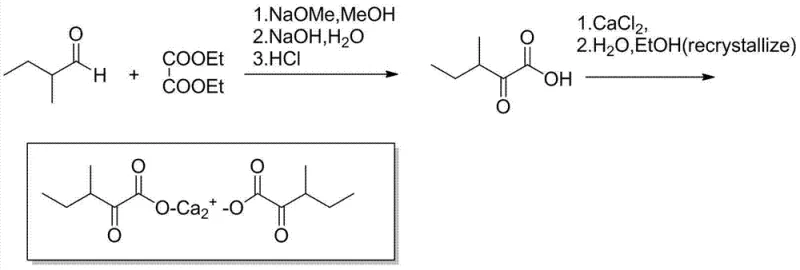
Impurity control is meticulously managed through a sophisticated workup procedure that exploits the pH-dependent solubility of the target molecule. After the reaction reaches completion, the mixture is acidified to a pH range of 1.0 to 4.0, converting the sodium salt into the free keto acid, which is then extracted into an organic solvent such as methyl isobutyl ketone or ethyl acetate. This extraction step effectively separates the organic product from inorganic salts and water-soluble impurities. The organic phase is subsequently washed and back-extracted into an aqueous phase by adjusting the pH to a neutral range of 5 to 7. Finally, the pH is raised to 8 to 9, and calcium chloride is introduced to precipitate the target calcium salt. This multi-stage pH swing purification strategy is critical for achieving the reported liquid phase purity of over 99.5%, ensuring that the final active pharmaceutical ingredient meets the rigorous specifications required for human therapeutic use.
How to Synthesize Calcium 3-Methyl-2-Oxovalerate Efficiently
Implementing this synthesis route requires careful attention to stoichiometry and temperature control to maximize yield and minimize byproduct formation. The process begins with the preparation of the alkoxide solution, followed by the controlled addition of oxalate and aldehyde substrates. The subsequent hydrolysis and salification steps must be monitored closely to ensure complete conversion and optimal crystal formation during the final precipitation. The patent provides a robust framework for operators to follow, detailing specific molar ratios and solvent choices that have been empirically validated to deliver consistent results. For technical teams looking to adopt this methodology, the following standardized steps outline the critical operational parameters derived directly from the experimental embodiments described in the intellectual property documentation.
- Prepare a solution of sodium alkoxide in alcohol and add dialkyl oxalate, maintaining temperature control during the initial transesterification phase.
- Introduce 2-methylbutyraldehyde to the mixture to initiate aldol condensation, followed by the addition of alkali lye for hydrolysis and ring-opening.
- Acidify the reaction mixture to extract the free acid, then neutralize and treat with calcium chloride to precipitate the final calcium salt product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthesis route offers compelling advantages that directly address the pain points of procurement managers and supply chain directors in the fine chemical sector. The shift away from exotic reagents like Grignard solutions to commodity chemicals such as diethyl oxalate and 2-methylbutyraldehyde fundamentally alters the cost structure of the manufacturing process. These raw materials are widely available in the global market, reducing the risk of supply disruptions and price volatility that often plague specialized reagent markets. Furthermore, the elimination of complex intermediate isolation steps reduces the total number of unit operations required, which in turn lowers labor costs and equipment depreciation expenses. This streamlined approach allows manufacturers to offer more competitive pricing without sacrificing quality, making it an attractive option for cost-sensitive projects in the generic pharmaceutical and nutraceutical sectors.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the simplification of the synthetic route and the use of low-cost inputs. By avoiding the need for expensive catalysts or specialized anhydrous solvents, the direct material costs are significantly lowered. Additionally, the one-pot nature of the reaction reduces solvent consumption and waste disposal fees, contributing to substantial cost savings in the overall production budget. The high yield reported in the patent embodiments further enhances economic efficiency, ensuring that a greater proportion of raw materials are converted into saleable product rather than lost to side reactions or purification losses.
- Enhanced Supply Chain Reliability: Operational simplicity translates directly into supply chain resilience. Because the reaction conditions are mild and do not require specialized cryogenic equipment or inert gas manifolds, the process can be executed in a wider range of manufacturing facilities. This flexibility increases the pool of potential contract manufacturing organizations (CMOs) capable of producing the material, thereby diversifying the supply base and mitigating single-source risks. The robustness of the chemistry also means that batch-to-batch variability is minimized, ensuring consistent delivery schedules and reducing the likelihood of production delays caused by technical failures or out-of-specification results.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is straightforward due to the absence of hazardous exotherms and toxic byproducts. The use of aqueous workups and common organic solvents simplifies waste treatment, allowing facilities to meet environmental discharge standards with standard effluent treatment plants. This environmental compatibility is increasingly important for multinational corporations aiming to reduce their carbon footprint and adhere to sustainable sourcing policies. The ability to scale safely and sustainably ensures long-term supply continuity, which is a critical factor for strategic partnerships in the development of life-saving renal therapies.
Frequently Asked Questions (FAQ)
To further clarify the technical and commercial implications of this synthesis method, we have compiled a set of frequently asked questions based on the specific details found in the patent literature. These queries address common concerns regarding yield optimization, purity profiles, and the feasibility of adapting this route for different scales of production. Understanding these aspects is essential for stakeholders evaluating the viability of integrating this intermediate into their existing supply chains or new product development pipelines. The answers provided below are grounded in the experimental data and mechanistic explanations offered within the intellectual property rights documentation.
Q: What are the primary advantages of this synthesis route over traditional Grignard methods?
A: Unlike traditional Grignard routes which require strict anhydrous conditions and cryogenic temperatures, this method operates under mild conditions (below 25°C) using common solvents, significantly reducing operational complexity and safety risks.
Q: How is high purity achieved in the final calcium salt product?
A: High purity exceeding 99.5% is achieved through a rigorous purification process involving pH-controlled extraction, selective salification with calcium chloride, and final recrystallization in a water-alcohol mixed solvent system.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the process is designed for industrial scalability, utilizing cheap and readily available raw materials like diethyl oxalate and 2-methylbutyraldehyde, while avoiding toxic byproducts and complex intermediate isolation steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Calcium 3-Methyl-2-Oxovalerate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of effective renal therapeutics and nutritional supplements. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the demanding volume requirements of global pharmaceutical clients. We are committed to maintaining stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify that every batch of calcium 3-methyl-2-oxovalerate meets or exceeds the 99.5% purity benchmark established in the patent. Our state-of-the-art facilities are equipped to handle the specific solvent systems and pH control requirements of this synthesis, guaranteeing a consistent and reliable supply of this vital building block.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific project needs. By leveraging our expertise in process optimization and supply chain management, we can help you identify opportunities to reduce costs and improve efficiency in your manufacturing operations. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. We are eager to demonstrate how our capabilities align with your strategic goals and to establish a long-term partnership that drives value and innovation in the pharmaceutical industry.
