Advanced Metal-Free Oxidation Technology for Commercial Scale Aromatic Aldehyde Manufacturing
The pharmaceutical and agrochemical industries are constantly seeking robust, scalable, and environmentally compliant synthetic routes for key intermediates. A pivotal advancement in this domain is detailed in Chinese Patent CN100363320C, which discloses a highly efficient method for producing aromatic aldehyde compounds. This technology represents a significant departure from traditional oxidation protocols that rely on hazardous heavy metals or expensive transition metal catalysts. By leveraging a specific combination of bromate oxidants and acid catalysts, the process achieves exceptional selectivity for the aldehyde functionality while minimizing the formation of over-oxidized carboxylic acid byproducts. For R&D directors and process chemists, this patent offers a compelling alternative for synthesizing critical building blocks such as benzaldehyde derivatives, which are ubiquitous in drug discovery and development pipelines. The ability to operate under relatively mild conditions without the need for complex catalytic systems positions this method as a cornerstone for modern, sustainable chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the oxidation of benzyl alcohols or ethers to their corresponding aldehydes has been fraught with significant technical and environmental challenges. Traditional methods often employ stoichiometric amounts of chromium-based reagents, active manganese dioxide, or Swern oxidation conditions, all of which generate substantial quantities of toxic waste. For instance, chromium oxidation requires rigorous post-reaction treatment to remove residual heavy metals, a step that is not only costly but also poses severe environmental liabilities. Furthermore, methods utilizing transition metal catalysts like ruthenium often necessitate expensive ligand systems and complex purification steps to ensure the final product meets stringent ppm-level metal specifications required for API manufacturing. Additionally, older bromate-based methods often suffered from poor selectivity or required excessive amounts of oxidants, leading to difficult waste streams containing unreacted bromide salts. These limitations create bottlenecks in supply chains, increase the cost of goods sold, and complicate regulatory filings due to impurity profiles.
The Novel Approach
The methodology described in the patent data introduces a transformative approach by utilizing a controlled amount of oxybromide, such as sodium bromate, in the presence of an organic acid catalyst like acetic acid. This system effectively activates the oxidant to selectively convert benzyl alcohols and benzyl ethers into aromatic aldehydes with high efficiency. Unlike previous art that might require harsh conditions or excess reagents, this novel route operates effectively within a temperature range of 20°C to 100°C, allowing for precise thermal management during scale-up. The use of acetic acid serves a dual purpose as both a solvent and a catalyst, simplifying the reaction matrix and reducing the need for additional auxiliary chemicals. Crucially, the process demonstrates remarkable tolerance for various substituents on the aromatic ring, including electron-withdrawing groups like nitro and chloro, which typically deactivate the substrate in other oxidation systems. This versatility ensures that a wide library of functionalized aldehydes can be accessed through a single, unified platform technology.
Mechanistic Insights into Acid-Catalyzed Bromate Oxidation
The core of this technological breakthrough lies in the synergistic interaction between the acid catalyst and the bromate species. In the absence of a strong acid promoter, bromates are relatively stable and unreactive towards organic substrates at moderate temperatures. However, upon protonation by carboxylic acids such as acetic acid, the bromate ion generates reactive bromine species in situ that are capable of abstracting hydrogen from the benzylic position. This mechanism proceeds through a pathway that favors the formation of the aldehyde over the carboxylic acid, largely due to the specific redox potential established by the acid-bromate couple. The reaction kinetics are carefully balanced; if the acidity is too low, the reaction stalls, but if the oxidant concentration is too high without proper control, over-oxidation can occur. The patent data highlights that maintaining the molar ratio of the oxidant within a specific stoichiometric window is critical for maximizing yield and selectivity. This precise control allows manufacturers to stop the reaction at the aldehyde stage with minimal formation of the corresponding acid, thereby simplifying downstream purification.
Furthermore, the impurity profile generated by this method is significantly cleaner compared to metal-catalyzed alternatives. In traditional transition metal oxidations, trace metal residues can coordinate with the product or catalyze decomposition pathways during storage. In contrast, the byproducts of this bromate oxidation are primarily inorganic salts and water, which are easily removed via aqueous workup. The patent examples demonstrate that even with sensitive substrates like o-nitrobenzyl alcohol, the reaction proceeds smoothly to give high conversions without degrading the nitro group. This chemoselectivity is vital for multi-step synthesis where protecting groups might otherwise be required. The structural integrity of the resulting aromatic aldehyde, as represented by the general formula in the patent, is preserved, ensuring that the material is suitable for subsequent condensation or coupling reactions common in medicinal chemistry.
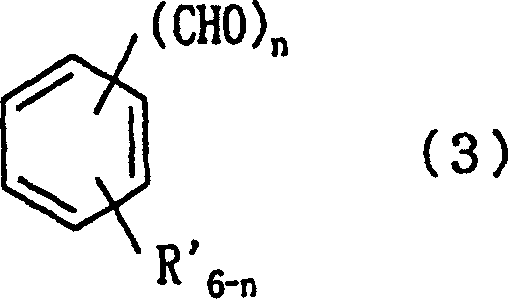
How to Synthesize Aromatic Aldehydes Efficiently
Implementing this oxidation technology in a production environment requires adherence to specific operational parameters to ensure safety and reproducibility. The process generally involves dissolving the benzyl substrate in a mixture of acetic acid and water, followed by the controlled addition of the sodium bromate oxidant. Temperature control is paramount, as the reaction can be exothermic; therefore, semi-batch addition of the oxidant is recommended to manage heat release. The reaction progress is typically monitored by gas chromatography to determine the endpoint, ensuring complete consumption of the starting material while avoiding prolonged exposure to oxidative conditions. Once the reaction is complete, the mixture is neutralized, and the product is extracted using standard organic solvents. This straightforward workflow minimizes unit operations and reduces the overall processing time, making it highly attractive for contract manufacturing organizations looking to optimize throughput.
- Prepare the reaction mixture by combining the benzyl alcohol or ether substrate with an acid catalyst such as acetic acid and a solvent system.
- Slowly add the oxidizing agent, typically sodium bromate dissolved in water, to the reaction vessel while maintaining temperature control between 20°C and 100°C.
- Monitor the reaction progress until the oxidant is consumed, then perform standard workup procedures including neutralization and extraction to isolate the high-purity aldehyde.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this metal-free oxidation technology offers profound strategic benefits. The elimination of expensive transition metal catalysts such as ruthenium or palladium directly translates to a reduction in raw material costs, which is a primary driver for margin improvement in high-volume chemical manufacturing. Moreover, the avoidance of heavy metals removes the necessity for specialized scavenging resins or complex filtration steps designed to meet strict residual metal limits, further streamlining the production process. This simplification not only lowers operational expenditures but also reduces the dependency on volatile precious metal markets, thereby stabilizing the cost structure for long-term supply agreements. The reliance on commodity chemicals like sodium bromate and acetic acid ensures a robust and resilient supply chain, as these reagents are widely available from multiple global vendors, mitigating the risk of single-source bottlenecks.
- Cost Reduction in Manufacturing: The economic impact of switching to this bromate-based oxidation is substantial when analyzed through the lens of total cost of ownership. By removing the need for precious metal catalysts, companies eliminate a significant line item from their bill of materials. Additionally, the simplified workup procedure reduces solvent consumption and energy usage associated with extensive purification steps. The high selectivity of the reaction means that yield losses due to over-oxidation to carboxylic acids are minimized, maximizing the output per batch. This efficiency gain allows manufacturers to produce more product with the same reactor volume, effectively increasing asset utilization rates without requiring capital investment in new infrastructure. The cumulative effect of these factors results in a highly competitive cost position for aromatic aldehyde intermediates.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the scarcity of specialized reagents or the logistical challenges of handling hazardous materials. This technology mitigates those risks by utilizing stable, solid oxidants that are easy to transport and store compared to gaseous oxidants or pyrophoric reagents. The robustness of the reaction conditions means that production can be maintained across different geographical locations without significant re-validation efforts. Furthermore, the reduced generation of hazardous waste simplifies disposal logistics and lowers compliance costs associated with environmental regulations. For supply chain heads, this translates to a more predictable lead time and a lower risk of production stoppages due to regulatory audits or waste disposal capacity issues, ensuring a steady flow of materials to downstream customers.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to commercial production often reveals hidden complexities, particularly regarding heat management and safety. This oxidation method is inherently scalable because the reaction rate can be easily controlled by the addition rate of the oxidant solution. The use of aqueous acetic acid as a solvent system provides a high heat capacity, which helps dampen thermal runaways. From an environmental standpoint, the process aligns with green chemistry principles by avoiding toxic heavy metals and generating benign inorganic byproducts. This compliance facilitates easier permitting for new manufacturing lines and enhances the corporate sustainability profile. For clients demanding eco-friendly supply chains, this technology offers a verifiable pathway to producing high-purity intermediates with a significantly reduced environmental footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this aromatic aldehyde synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding the nuances of substrate compatibility, reaction safety, and purification requirements is essential for project managers evaluating this route for inclusion in their manufacturing portfolio. The answers below reflect the practical realities of operating this chemistry at scale.
Q: How does this method prevent over-oxidation to carboxylic acids?
A: The process utilizes a specific molar ratio of bromate oxidant relative to the substrate and employs an acid catalyst that promotes selective oxidation, stopping primarily at the aldehyde stage rather than proceeding to the carboxylic acid.
Q: Is this method suitable for substrates with electron-withdrawing groups?
A: Yes, the technology demonstrates excellent reactivity even with challenging substrates containing nitro or chloro groups, which often struggle in traditional oxidation methods, ensuring broad applicability for diverse intermediate synthesis.
Q: What are the waste disposal advantages compared to chromium-based oxidation?
A: By eliminating toxic heavy metals like chromium or manganese, the process significantly reduces hazardous waste treatment costs and simplifies environmental compliance, making it ideal for large-scale green manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aromatic Aldehyde Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced, metal-free synthetic routes is critical for the future of pharmaceutical and agrochemical manufacturing. Our technical team has extensively evaluated the oxidation methodology described in CN100363320C and possesses the expertise to implement it seamlessly across our production facilities. We offer extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves from benchtop to bulk supply without interruption. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify the absence of heavy metal contaminants and ensure consistent batch-to-batch performance. We understand that reliability is the currency of the chemical industry, and we are dedicated to being a partner you can trust for critical intermediates.
We invite you to engage with our technical procurement team to discuss how this innovative oxidation technology can be tailored to your specific project needs. Whether you require a Customized Cost-Saving Analysis for an existing product or need to develop a new route for a complex aldehyde derivative, we have the capabilities to deliver. Please contact us to request specific COA data and route feasibility assessments that demonstrate the viability of this approach for your supply chain. Let us collaborate to optimize your manufacturing process, reduce costs, and secure a sustainable supply of high-quality aromatic aldehydes for your global operations.
