Revolutionizing Antitumor Intermediate Production via Rapid One-Step Phosphorylation Technology
Revolutionizing Antitumor Intermediate Production via Rapid One-Step Phosphorylation Technology
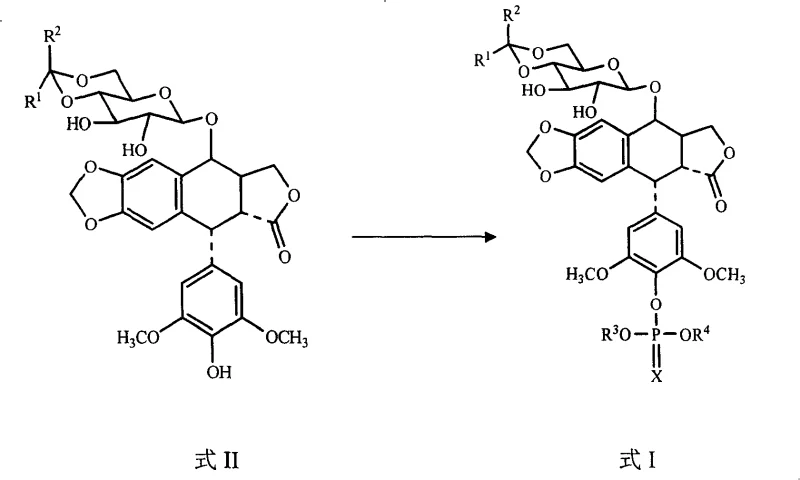
The pharmaceutical industry constantly seeks robust methodologies to enhance the production efficiency of critical antineoplastic agents, particularly water-soluble derivatives of podophyllotoxin. Patent CN100410266C introduces a groundbreaking synthetic approach for preparing podophyllotoxin phosphate derivatives, addressing long-standing challenges in selectivity and process economics. This technology enables the direct phosphorylation of podophyllotoxin derivatives (Formula II) using phosphorous acid derivatives (Formula III) in the presence of a tetrahalomethane, a tertiary amine, and a 4-dimethylaminopyridine catalyst. Unlike traditional multi-step sequences, this innovation allows the reaction to proceed in a single step with a remarkably short reaction time of within 1 hour. For R&D directors and supply chain leaders, this represents a pivotal shift towards leaner, more agile manufacturing of high-value oncology intermediates like etoposide phosphate and teniposide phosphate.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of phosphate esters of podophyllotoxin derivatives has been plagued by significant operational inefficiencies and high costs. As documented in background art such as British Patent 2207674, conventional methods often rely on diphenyl chlorophosphate as the phosphorylating agent. This reagent is not only prohibitively expensive but also necessitates extended reaction times approaching 100 hours to achieve conversion, creating a severe bottleneck in production throughput. Furthermore, alternative strategies described in patents like CN1189499A require a cumbersome protection-deprotection sequence. In these older routes, the hydroxyl groups on the sugar moiety must first be protected to prevent unwanted side reactions, followed by phosphorylation and subsequent removal of the protecting groups. This multi-step manipulation drastically increases material consumption, waste generation, and overall processing time, rendering the final API intermediate economically burdensome for large-scale procurement.
The Novel Approach
The methodology disclosed in CN100410266C fundamentally reengineers this synthetic landscape by enabling a direct, one-pot phosphorylation that bypasses the need for sugar hydroxyl protection. By employing a phosphorous acid derivative activated by a tetrahalomethane and tertiary amine system, the reaction achieves high chemoselectivity for the phenolic hydroxyl group exclusively. This strategic simplification means that all reactants can be added in a streamlined fashion without the need for complex batch-wise additions or intermediate isolation steps. The result is a process that completes in merely 30 to 60 minutes, a drastic reduction from the multi-day timelines of legacy technologies. This leap in efficiency not only accelerates the path from raw materials to finished intermediate but also significantly lowers the barrier to entry for cost-effective manufacturing of these critical anticancer drugs.
Mechanistic Insights into Tetrahalomethane-Mediated Phosphorylation
The core of this technological advancement lies in the in situ generation of a highly reactive phosphorus species through an Atherton-Todd type reaction mechanism. In the presence of a base such as N,N-diisopropylethylamine or triethylamine, the tetrahalomethane (e.g., carbon tetrachloride) activates the P-H bond of the phosphorous acid derivative. This activation generates a reactive halophosphonate intermediate that acts as the true electrophile in the substitution reaction. The inclusion of 4-dimethylaminopyridine (DMAP) as a nucleophilic catalyst further enhances the reaction kinetics by facilitating the transfer of the phosphoryl group to the substrate. Crucially, the reaction conditions are tightly controlled at low temperatures ranging from -10°C to -30°C. This thermal control is essential for modulating the reactivity of the intermediate, ensuring that it reacts preferentially with the more acidic and nucleophilic phenolic oxygen atom rather than the aliphatic hydroxyls on the glycosidic sugar ring.
From an impurity control perspective, this mechanism offers superior cleanliness compared to chlorophosphate-based routes. The avoidance of highly reactive chlorophosphate reagents minimizes the formation of bis-phosphorylated byproducts or degradation products associated with harsh acidic conditions. The patent data indicates that this mechanistic precision yields crude products with purity levels exceeding 90% as determined by HPLC, which can be further purified to greater than 97%. For quality assurance teams, this implies a simplified downstream purification process, reducing the load on chromatography columns and crystallization steps. The high selectivity ensures that the final impurity profile is manageable and consistent, a critical factor for regulatory compliance in the production of injectable antineoplastic agents.
How to Synthesize Etoposide Phosphate Intermediates Efficiently
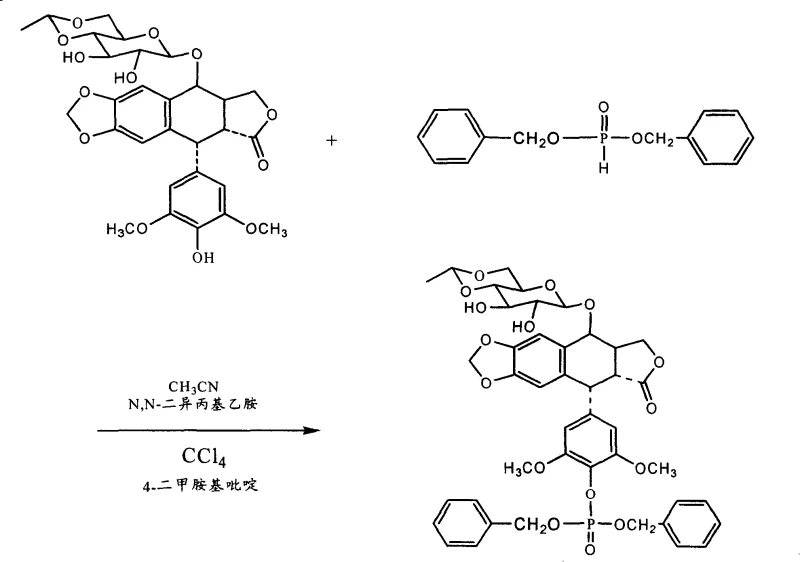
Implementing this synthesis requires precise control over stoichiometry and temperature to maximize the benefits of the novel activation system. The process begins with the dissolution of the podophyllotoxin derivative, such as etoposide, in a polar aprotic solvent like acetonitrile. The reaction mixture is then cooled to sub-zero temperatures before the sequential addition of the activator system. Specifically, carbon tetrachloride, a tertiary amine base (preferably 2 equivalents), and a catalytic amount of DMAP (0.2 equivalents) are introduced. Subsequently, the phosphorous acid derivative, such as dibenzyl phosphite, is added dropwise to maintain thermal stability. The reaction is allowed to proceed for approximately 1 hour until the starting material is fully consumed. The resulting intermediate, such as etoposide dibenzyl phosphate, can then be isolated or directly subjected to catalytic hydrogenation to yield the final free acid or salt form.
- Dissolve the podophyllotoxin derivative (Formula II) in a suitable solvent like acetonitrile and cool the mixture to a temperature range of -10°C to -30°C.
- Add tetrahalomethane (e.g., CCl4), a tertiary amine base (e.g., DIPEA), and a catalytic amount of 4-dimethylaminopyridine (DMAP) to the reaction vessel.
- Dropwise add the phosphorous acid derivative (Formula III) while maintaining low temperature, stir for 30-60 minutes until completion, and proceed to workup or hydrogenation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route translates into tangible strategic advantages regarding cost structure and supply continuity. The elimination of expensive chlorophosphate reagents in favor of readily available phosphorous acid disubstituted derivatives directly impacts the bill of materials, driving down the unit cost of the intermediate. Furthermore, the reduction of reaction time from nearly 100 hours to under 1 hour dramatically increases asset utilization rates, allowing manufacturing facilities to produce significantly higher volumes within the same timeframe. This efficiency gain is compounded by the operational simplicity of the one-pot process, which reduces labor hours and minimizes the risk of human error during complex multi-step manipulations.
- Cost Reduction in Manufacturing: The economic model of this process is heavily favored by the substitution of high-cost reagents with commodity chemicals. By avoiding the use of diphenyl chlorophosphate and eliminating the separate steps for protecting and deprotecting sugar hydroxyls, the overall consumption of solvents and reagents is drastically minimized. This streamlined approach removes entire unit operations from the production schedule, leading to substantial savings in utility costs, waste disposal fees, and equipment occupancy time, thereby optimizing the total cost of ownership for the manufacturing process.
- Enhanced Supply Chain Reliability: The robustness of this one-step chemistry enhances supply chain resilience by reducing the number of potential failure points. Traditional multi-step syntheses are vulnerable to delays at each protection or deprotection stage; in contrast, this consolidated route ensures a faster turnaround from raw material intake to finished goods. The high yield reported (up to 90%) ensures that less raw material is required to meet production targets, mitigating risks associated with raw material scarcity and price volatility in the global market for specialized chiral starting materials.
- Scalability and Environmental Compliance: From an environmental and safety standpoint, the process offers a cleaner profile suitable for modern green chemistry standards. The reduction in reaction time and the absence of multiple workup stages decrease the total volume of organic waste generated per kilogram of product. Additionally, the ability to run the reaction in common solvents like acetonitrile with standard amine bases simplifies solvent recovery and recycling protocols. This scalability ensures that the process can be seamlessly transferred from pilot plant to commercial scale (100 kgs to 100 MT) without encountering the nonlinear scale-up issues often seen in complex, multi-step phosphorylation sequences.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this phosphorylation technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, aimed at clarifying the operational feasibility and competitive advantages of this method for potential licensees and manufacturing partners.
Q: How does this method achieve selectivity for the phenolic hydroxyl group over the sugar hydroxyls?
A: The method utilizes a specific activation system involving tetrahalomethane and tertiary amine which generates a reactive phosphorus species in situ. Under the controlled low-temperature conditions (-10°C to -30°C), this species exhibits high chemoselectivity for the more acidic phenolic hydroxyl group, effectively bypassing the aliphatic hydroxyl groups on the sugar moiety without the need for protecting groups.
Q: What are the primary cost drivers eliminated by this synthetic route compared to British Patent 2207674?
A: This route eliminates the need for expensive diphenyl chlorophosphate reagents and avoids the prolonged reaction times (nearly 100 hours) associated with older methods. By utilizing lower-cost phosphorous acid disubstituted derivatives and completing the reaction within 1 hour, both raw material costs and operational overheads are substantially reduced.
Q: Is this process scalable for commercial manufacturing of etoposide phosphate?
A: Yes, the process is highly scalable due to its one-pot nature and simple operation. It does not require complex protection and deprotection sequences or batch-wise addition of reagents, making it ideal for large-scale production facilities aiming for high throughput and consistent purity profiles above 90%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Podophyllotoxin Phosphate Derivatives Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating advanced patent technologies like CN100410266C into commercial reality. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this rapid phosphorylation method are fully realized in a GMP-compliant environment. Our rigorous QC labs and stringent purity specifications guarantee that every batch of podophyllotoxin phosphate intermediates meets the exacting standards required for oncology drug formulation, providing our partners with unwavering confidence in product quality and consistency.
We invite global pharmaceutical companies to leverage our technical expertise to optimize their supply chains for antineoplastic agents. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific production volumes. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how this innovative synthesis can drive value and efficiency in your manufacturing operations.
