Revolutionizing Fluorinated Intermediate Production: Continuous Synthesis of Diaryl Hexafluoropropane via Ionic Liquids
Revolutionizing Fluorinated Intermediate Production: Continuous Synthesis of Diaryl Hexafluoropropane via Ionic Liquids
The global demand for high-performance fluorinated polymers and advanced electronic materials has necessitated a paradigm shift in the synthesis of key intermediates like diaryl hexafluoropropane. Patent CN112409125A introduces a groundbreaking methodology that transitions the production of these critical molecules from hazardous batch processes to a safe, efficient continuous flow system. This innovation leverages the unique properties of acidic ionic liquids to catalyze the condensation of hexafluoroacetone trihydrate with various aryl compounds, effectively eliminating the need for corrosive hydrogen fluoride and toxic gaseous reagents. For R&D directors and procurement strategists, this represents a significant opportunity to secure a reliable fluorine material supplier capable of delivering high-purity intermediates with a vastly improved environmental footprint. The technology not only addresses the chronic safety issues associated with traditional fluorination but also unlocks new potentials for cost reduction in advanced materials manufacturing through catalyst recycling and energy-efficient continuous operation.
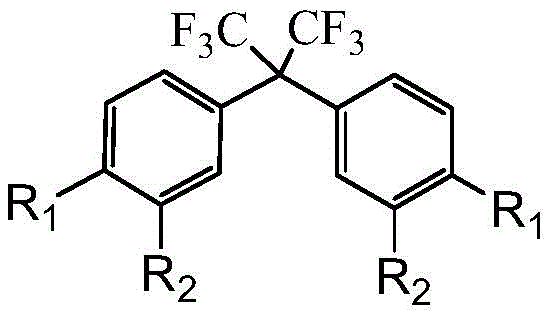
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of diaryl hexafluoropropanes, such as the vital vulcanizing agent Bisphenol AF, has been plagued by severe safety and operational challenges. Conventional routes typically rely on the direct reaction of phenol with gaseous hexafluoroacetone (HFA) in the presence of anhydrous hydrogen fluoride (HF) as a catalyst. This approach presents a triple threat: first, HFA is a highly toxic gas that requires stringent containment measures to prevent lethal exposure; second, anhydrous HF is notoriously corrosive, demanding expensive Hastelloy-lined reactors and imposing heavy maintenance costs on production facilities; and third, these processes are predominantly batch-operated, leading to inconsistent heat transfer, potential hot spots, and prolonged reaction times ranging from 8 to 12 hours. Furthermore, alternative routes involving hexafluoropropylene oxide still suffer from the generation of corrosive byproducts like hydrogen chloride, while fluorination of Bisphenol A using elemental fluorine involves extreme hazards due to the explosive nature of F2 gas. These legacy methods create substantial bottlenecks in the commercial scale-up of complex fluorinated intermediates, limiting supply reliability and inflating the total cost of ownership for downstream polymer manufacturers.
The Novel Approach
In stark contrast, the methodology disclosed in CN112409125A redefines the synthetic landscape by introducing a continuous loop reactor system driven by reusable ionic liquid catalysts. Instead of handling dangerous gases, the process utilizes hexafluoroacetone trihydrate, a stable solid adduct that releases the reactive ketone in situ under controlled conditions, thereby inherently enhancing process safety. The core innovation lies in the use of imidazolium or quaternary ammonium ionic liquids, such as [C6mim][HSO4], which function dually as the reaction medium and the acid catalyst. This eliminates the need for volatile organic solvents and corrosive mineral acids. By pumping the reactant mixture through a circulating loop reactor at temperatures between 80°C and 150°C, the system achieves rapid mixing and superior heat dissipation, reducing the reaction residence time to merely 5 to 10 minutes. This transition from batch to continuous processing not only drastically improves throughput but also ensures consistent product quality, making it an ideal solution for reducing lead time for high-purity fluorine chemicals in a competitive global market.
Mechanistic Insights into Ionic Liquid-Catalyzed Condensation
The efficacy of this novel process is rooted in the unique physicochemical properties of the acidic ionic liquids employed. These salts possess tunable acidity, allowing them to protonate the carbonyl oxygen of the hexafluoroacetone moiety effectively, thereby activating it towards electrophilic aromatic substitution with the aryl compound (e.g., phenol or o-xylene). Unlike traditional homogeneous acid catalysts that are difficult to separate, the ionic liquid forms a distinct phase or remains in the solution post-reaction, facilitating easy separation of the solid product upon cooling. The mechanism likely proceeds via a Friedel-Crafts type alkylation where the electron-deficient hexafluoroisopropylidene cation attacks the electron-rich aromatic ring. The presence of the ionic liquid stabilizes the transition state and intermediates, leading to the high selectivity observed in the experimental data. Moreover, the addition of antioxidants like sodium sulfite plays a crucial role in scavenging any oxidative impurities that might arise during the high-temperature continuous flow, ensuring the final high-purity diaryl hexafluoropropane meets the rigorous specifications required for polyimide and fluororubber applications.
From a process engineering perspective, the circulating loop reactor is instrumental in maximizing the contact between the hydrophobic aryl substrate and the polar ionic liquid phase. The high shear forces generated within the loop ensure that mass transfer limitations, which often plague multiphase reactions, are virtually eliminated. This intense mixing allows the reaction to proceed to near-completion (conversion rates up to 99%) within minutes, a feat unachievable in static batch vessels. Furthermore, the thermal stability of the ionic liquid allows it to withstand the reaction temperatures without degradation, enabling its recovery and reuse. Experimental data indicates that the catalyst retains its activity over at least five consecutive cycles after simple vacuum drying, demonstrating a robust catalytic cycle that minimizes waste generation. This mechanistic robustness provides R&D teams with a predictable and scalable platform for synthesizing a wide range of diaryl hexafluoropropane derivatives by simply varying the R1 and R2 substituents on the aromatic rings.
How to Synthesize Diaryl Hexafluoropropane Efficiently
Implementing this continuous synthesis route requires precise control over stoichiometry and flow dynamics to maximize yield and purity. The process begins with the preparation of a homogeneous feed solution containing hexafluoroacetone trihydrate, the chosen aryl compound in a molar excess (typically 1:1.5 to 1:3 ratio), and the ionic liquid catalyst. An antioxidant is added to protect the product from oxidation during the high-temperature phase. This mixture is then pumped into the pre-heated circulating loop reactor, where the residence time is strictly controlled between 5 and 10 minutes depending on the specific reactivity of the aryl substrate. Upon exiting the reactor, the effluent is cooled to induce crystallization of the target diaryl hexafluoropropane, which is then filtered off. The remaining mother liquor, rich in the ionic liquid catalyst, is subjected to vacuum drying to remove water and trace volatiles before being recycled back into the feed tank. For detailed operational parameters and specific stoichiometric ratios tailored to your production scale, please refer to the standardized synthesis steps outlined below.
- Mix hexafluoroacetone trihydrate, the selected aryl compound, and an acidic ionic liquid catalyst along with a stabilizing antioxidant.
- Pump the mixture into a circulating loop reactor maintained at 80-150°C, ensuring a retention time of 5-10 minutes for optimal conversion.
- Cool the effluent to precipitate the solid product, filter to separate the crude material, and recycle the ionic liquid phase for subsequent batches.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this ionic liquid-based continuous process offers transformative economic and logistical benefits. The most immediate impact is the drastic simplification of the supply chain regarding raw material handling; by replacing toxic gaseous hexafluoroacetone and corrosive hydrogen fluoride with stable solids and non-volatile liquids, the facility requirements are significantly downgraded, removing the need for specialized high-pressure gas containment and exotic alloy reactors. This shift directly translates to lower capital expenditure (CapEx) for new production lines and reduced maintenance costs for existing infrastructure. Furthermore, the continuous nature of the process allows for a "just-in-time" manufacturing model, where production can be ramped up or down quickly to match market demand without the inefficiencies inherent in large batch campaigns. This flexibility is crucial for maintaining supply continuity in the volatile specialty chemicals market.
- Cost Reduction in Manufacturing: The elimination of expensive corrosion-resistant equipment and the ability to recycle the ionic liquid catalyst multiple times without significant loss of activity leads to substantial operational cost savings. Unlike traditional processes that generate large volumes of acidic wastewater requiring neutralization and treatment, this method produces minimal waste, thereby reducing environmental compliance costs and waste disposal fees. The high conversion rates and selectivity mean that raw material utilization is maximized, further driving down the cost per kilogram of the final API intermediate or polymer additive.
- Enhanced Supply Chain Reliability: By utilizing safer raw materials like hexafluoroacetone trihydrate, the risks associated with transportation and storage are markedly reduced, ensuring a more stable inbound logistics network. The continuous flow setup minimizes the risk of batch failures due to human error or thermal runaway, which are common in exothermic batch reactions, thus guaranteeing a consistent output of qualified product. This reliability allows downstream customers to reduce their safety stock levels and optimize their inventory management, knowing that the reliable fluorine material supplier can deliver consistent quality on a predictable schedule.
- Scalability and Environmental Compliance: The modular nature of loop reactors makes scaling from pilot to commercial production straightforward, avoiding the non-linear scale-up issues often encountered with batch reactors. The process aligns perfectly with green chemistry principles by avoiding volatile organic compounds (VOCs) and hazardous reagents, making it easier to obtain environmental permits and meet increasingly stringent global sustainability standards. This environmental advantage not only future-proofs the production facility against regulatory changes but also enhances the brand value of the end-products in eco-conscious markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the continuous synthesis of diaryl hexafluoropropane. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a clear understanding of the technology's capabilities and limitations. Understanding these details is essential for evaluating the feasibility of integrating this advanced manufacturing route into your existing supply chain.
Q: How does the ionic liquid catalyst improve safety compared to traditional HF methods?
A: Traditional methods rely on highly corrosive hydrogen fluoride (HF) and toxic gaseous hexafluoroacetone. The patented process utilizes non-volatile, reusable acidic ionic liquids and safer hexafluoroacetone trihydrate, drastically reducing equipment corrosion risks and operator exposure to toxic gases.
Q: What are the typical conversion rates achieved in this continuous flow process?
A: The continuous loop reactor system demonstrates exceptional efficiency, achieving conversion rates of up to 99% based on hexafluoroacetone trihydrate, with selectivity often exceeding 95% for target products like Bisphenol AF.
Q: Can the ionic liquid catalyst be recycled effectively?
A: Yes, the ionic liquid serves as both solvent and catalyst. After simple vacuum drying, it can be reused for multiple cycles (demonstrated for at least 5 runs) without significant loss of catalytic activity, contributing to substantial waste reduction.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diaryl Hexafluoropropane Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced continuous flow chemistry requires a partner with deep technical expertise and robust manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves seamlessly from laboratory concept to industrial reality. Our facilities are equipped with state-of-the-art continuous flow reactors and rigorous QC labs capable of meeting stringent purity specifications for high-value fluorinated intermediates. We understand the critical importance of impurity control in polymer and pharmaceutical applications, and our dedicated analytical teams work tirelessly to characterize and minimize trace contaminants, guaranteeing a product that performs consistently in your final formulations.
We invite you to collaborate with us to leverage this cutting-edge synthesis technology for your specific application needs. Whether you require custom synthesis of novel diaryl hexafluoropropane derivatives or optimization of existing supply chains, our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. Please contact our technical procurement team today to request specific COA data, discuss route feasibility assessments, and discover how we can drive efficiency and safety in your fluorine chemistry projects.
