Revolutionizing Sartanbiphenyl Production via Continuous Flow Microreactor Technology
Revolutionizing Sartanbiphenyl Production via Continuous Flow Microreactor Technology
The pharmaceutical industry is witnessing a paradigm shift in the manufacturing of critical antihypertensive intermediates, driven by the urgent need for safer, more efficient, and scalable synthetic routes. Patent CN115611769A introduces a groundbreaking method for synthesizing sartanbiphenyl, chemically known as 2-cyano-4'-methyl biphenyl, utilizing advanced continuous flow microreactor technology. This molecule serves as the pivotal structural backbone for a wide array of angiotensin II receptor blockers (ARBs), including Valsartan, Irbesartan, and Telmisartan, which are essential first-line treatments for hypertension globally. 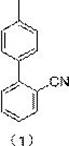 The traditional batch processes for producing this key intermediate have long been plagued by safety hazards associated with Grignard reagent preparation, inconsistent reaction kinetics, and significant environmental waste generation. By transitioning to a continuous flow regime, this patented innovation effectively resolves the thermal management issues inherent in exothermic Grignard formations, ensuring a stable and continuous supply of high-quality intermediates that meet the rigorous purity standards required for modern API manufacturing.
The traditional batch processes for producing this key intermediate have long been plagued by safety hazards associated with Grignard reagent preparation, inconsistent reaction kinetics, and significant environmental waste generation. By transitioning to a continuous flow regime, this patented innovation effectively resolves the thermal management issues inherent in exothermic Grignard formations, ensuring a stable and continuous supply of high-quality intermediates that meet the rigorous purity standards required for modern API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of sartanbiphenyl has relied on several distinct methodologies, each carrying substantial operational burdens that hinder optimal commercial scalability. The classical Meyer o-anisic acid method, while chemically viable, suffers from an excessively long reaction sequence, generating prohibitive amounts of three wastes and requiring complex equipment setups that drive up capital expenditure. Alternatively, transition metal-catalyzed cross-coupling reactions such as the Suzuki method utilize 4-methyl phenylboronic acid and 2-bromobenzonitrile; however, this route is economically disadvantaged by the high cost of palladium catalysts and ligand systems, alongside the necessity for stringent alkaline conditions that complicate workup procedures. 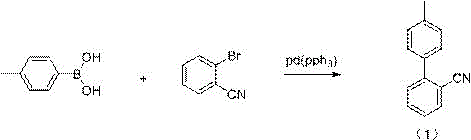 Furthermore, the Negishi coupling approach necessitates the in-situ preparation of organozinc reagents via Grignard intermediates, introducing severe handling challenges due to the hygroscopic nature of anhydrous zinc chloride and the increased difficulty in separating metal residues from the final product. These conventional batch processes often struggle with hot-spot formation during the highly exothermic Grignard step, leading to potential runaway reactions, inconsistent conversion rates, and a broader impurity profile that necessitates costly and time-consuming purification steps.
Furthermore, the Negishi coupling approach necessitates the in-situ preparation of organozinc reagents via Grignard intermediates, introducing severe handling challenges due to the hygroscopic nature of anhydrous zinc chloride and the increased difficulty in separating metal residues from the final product. These conventional batch processes often struggle with hot-spot formation during the highly exothermic Grignard step, leading to potential runaway reactions, inconsistent conversion rates, and a broader impurity profile that necessitates costly and time-consuming purification steps.
The Novel Approach
In stark contrast to these legacy methods, the continuous flow synthesis described in patent CN115611769A leverages the unique physical properties of microreactors to fundamentally alter the reaction landscape for sartanbiphenyl production. This novel approach decouples the Grignard reagent formation from the subsequent coupling step, allowing each stage to be optimized independently within a controlled flow environment. By continuously feeding magnesium powder suspended in an organic solvent along with p-chlorotoluene into a heated microreactor, the system achieves rapid and uniform mixing that eliminates the thermal gradients typical of large batch vessels. The subsequent coupling with o-chlorobenzonitrile is performed in a second reactor zone under precise temperature control, facilitating immediate contact between the reactive Grignard species and the metal catalyst. 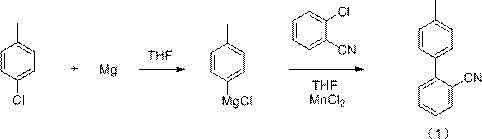 This seamless integration not only drastically reduces the residence time required for full conversion but also minimizes the accumulation of unstable intermediates, thereby enhancing overall process safety. The result is a streamlined manufacturing protocol that delivers superior product quality with significantly reduced solvent consumption and waste generation, aligning perfectly with the principles of green chemistry and modern industrial efficiency.
This seamless integration not only drastically reduces the residence time required for full conversion but also minimizes the accumulation of unstable intermediates, thereby enhancing overall process safety. The result is a streamlined manufacturing protocol that delivers superior product quality with significantly reduced solvent consumption and waste generation, aligning perfectly with the principles of green chemistry and modern industrial efficiency.
Mechanistic Insights into Continuous Flow Kumada Coupling
The core mechanistic advantage of this patented process lies in its ability to maintain a steady-state concentration of the highly reactive p-tolylmagnesium chloride intermediate, which is notoriously difficult to manage in batch operations. In the first stage of the continuous flow system, magnesium powder is activated and reacted with p-chlorotoluene at temperatures ranging from 50°C to 150°C, typically around 120°C, within a dynamic microreactor environment. The high surface-area-to-volume ratio of the microchannels ensures that the heat generated by the Grignard formation is dissipated almost instantaneously, preventing local overheating that could lead to Wurtz-type coupling byproducts or reagent decomposition. This precise thermal control allows for the use of stoichiometric ratios that maximize atom economy while maintaining the stability of the organomagnesium species as it flows directly into the second reaction zone without isolation.
Upon entering the second microreactor, the Grignard stream meets a solution of o-chlorobenzonitrile and a transition metal catalyst, such as manganese chloride (MnCl2), nickel, or palladium complexes, at temperatures between -20°C and 50°C. The catalytic cycle likely proceeds through a standard Kumada-Corriu mechanism where the metal catalyst undergoes oxidative addition with the aryl halide, followed by transmetallation with the Grignard reagent and reductive elimination to form the biaryl bond. The continuous flow setup ensures that the catalyst is utilized with maximum efficiency, as the reactants are forced into intimate contact under laminar or turbulent flow regimes depending on the reactor design. This enhanced mass transfer capability significantly suppresses the formation of homocoupling impurities and unreacted starting materials, resulting in a crude product profile that is far cleaner than what is achievable in stirred tank reactors, thus simplifying the final recrystallization steps.
How to Synthesize Sartanbiphenyl Efficiently
Implementing this continuous flow methodology requires a specialized setup comprising at least three interconnected microreactors designed to handle heterogeneous suspensions and exothermic reactions safely. The process begins with the preparation of a magnesium suspension in a solvent like tetrahydrofuran (THF), which is pumped alongside the aryl chloride into the first heated reactor zone to generate the Grignard reagent. This stream is then immediately merged with the electrophile and catalyst solution in the second reactor, where the coupling occurs under strictly controlled thermal conditions to ensure high selectivity. Finally, the reaction mixture flows into a third unit for quenching with dilute acid, followed by phase separation and solvent removal to isolate the target biphenyl derivative. For detailed operational parameters and specific equipment configurations, please refer to the standardized synthesis guide below.
- Prepare the Grignard reagent by continuously pumping magnesium powder suspended in THF and p-chlorotoluene into a heated microreactor at 50-150°C.
- Mix the resulting p-tolylmagnesium chloride stream with o-chlorobenzonitrile and a metal catalyst solution (e.g., MnCl2) in a second microreactor maintained at -20 to 50°C.
- Quench the reaction mixture with dilute hydrochloric acid in a third reactor, separate phases, and recrystallize the organic layer to obtain high-purity sartanbiphenyl.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this continuous flow technology represents a strategic opportunity to optimize the cost structure and reliability of the sartanbiphenyl supply chain. Traditional batch manufacturing of Grignard-based intermediates often incurs hidden costs related to safety mitigation, extensive waste treatment, and variable batch-to-batch yields that disrupt production planning. By shifting to a continuous process, manufacturers can achieve a drastic simplification of the operational workflow, eliminating the need for large-scale containment systems required for hazardous batch Grignard reactions. This transition inherently lowers the barrier for scaling production capacity, allowing suppliers to respond more agilely to market demand fluctuations without the lengthy lead times associated with building new batch infrastructure.
- Cost Reduction in Manufacturing: The elimination of expensive palladium catalysts in favor of more abundant metals like manganese or nickel significantly reduces the raw material cost per kilogram of the final product. Furthermore, the improved conversion rates and selectivity inherent to flow chemistry minimize the loss of valuable starting materials, leading to substantial cost savings in feedstock procurement. The reduction in solvent usage and the simplified purification process also contribute to a lower overall cost of goods sold, making the final API intermediate more price-competitive in the global market.
- Enhanced Supply Chain Reliability: Continuous manufacturing systems are inherently more robust against process upsets, providing a consistent output of high-purity material that reduces the risk of supply interruptions due to failed batches. The ability to run the process for extended periods without shutdowns for cleaning or recharging ensures a steady flow of inventory, which is critical for maintaining the production schedules of downstream API manufacturers. This reliability fosters stronger partnerships between intermediate suppliers and pharmaceutical companies, as it guarantees the availability of key building blocks for life-saving antihypertensive medications.
- Scalability and Environmental Compliance: The modular nature of microreactor technology allows for capacity expansion through numbering-up rather than scaling-up, which avoids the engineering challenges and safety risks of enlarging batch vessels. This approach facilitates easier compliance with increasingly stringent environmental regulations by significantly reducing the volume of hazardous waste and solvent emissions generated per unit of product. The greener profile of this synthesis route not only lowers waste disposal costs but also enhances the sustainability credentials of the supply chain, a factor of growing importance to multinational pharmaceutical corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the continuous flow synthesis of sartanbiphenyl, derived directly from the insights provided in patent CN115611769A. These answers are designed to clarify the operational benefits and chemical rationale behind this advanced manufacturing technique for stakeholders evaluating its adoption. Understanding these details is crucial for assessing the feasibility of integrating this technology into existing production lines or for sourcing intermediates produced via this superior method.
Q: Why is continuous flow preferred over batch processing for Sartanbiphenyl synthesis?
A: Continuous flow microreactors offer superior heat and mass transfer compared to traditional batch reactors, significantly mitigating the safety risks associated with exothermic Grignard reactions while improving catalyst contact and reducing byproduct formation.
Q: What are the advantages of using MnCl2 catalyst in this specific coupling reaction?
A: Manganese chloride provides a cost-effective alternative to precious metal catalysts like palladium, offering mild reaction conditions and high selectivity for the Kumada coupling, which simplifies downstream purification and reduces heavy metal contamination risks.
Q: How does this method address the limitations of the Suzuki and Negishi coupling routes?
A: Unlike the Suzuki method which requires expensive boronic acids and palladium, or the Negishi method which involves moisture-sensitive organozinc reagents, this continuous flow Kumada approach utilizes stable starting materials and avoids the handling difficulties of pyrophoric or hygroscopic intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sartanbiphenyl Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of continuous flow chemistry in delivering high-value pharmaceutical intermediates with unmatched consistency and quality. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a supply of sartanbiphenyl that meets the most stringent purity specifications. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying the low impurity profiles achieved through advanced flow synthesis, guaranteeing that every batch supports the safety and efficacy of your final drug products.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage these technological advancements for their supply chains. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our continuous flow capabilities can drive efficiency and reduce costs in your API manufacturing operations.
