Revolutionizing Medroxyprogesterone Acetate Production Through Non-Hydrogenation Catalytic Translocation
The pharmaceutical industry continuously seeks robust synthetic pathways that balance high purity with economic viability, particularly for critical hormonal intermediates. Patent CN102911233A discloses a groundbreaking synthesis method for Medroxyprogesterone Acetate that fundamentally restructures the traditional manufacturing logic. By replacing the conventional catalytic hydrogenation step with a sophisticated acid-catalyzed translocation reaction, this technology addresses long-standing inefficiencies in yield and cost. The process initiates with 17 Alpha-hydroxy Progesterone, subjecting it to a series of protective group manipulations and stereochemical adjustments that culminate in the target acetate. This approach not only circumvents the reliance on expensive palladium catalysts but also mitigates the safety risks associated with high-pressure hydrogenation reactors. For R&D directors and procurement specialists, understanding this shift from heterogeneous catalysis to homogeneous acid catalysis represents a pivotal opportunity for supply chain optimization.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of Medroxyprogesterone Acetate has relied heavily on a classical route involving etherification followed by a Mannich reaction and a critical hydrogenation step. The most significant bottleneck in this legacy technology is the dependence on Palladium on Carbon (Pd/C) as a heterogeneous catalyst for the reduction phase. This reliance introduces substantial variable costs, as precious metal prices fluctuate wildly in the global market, directly impacting the cost of goods sold. Furthermore, the handling of pyrophoric catalysts requires specialized equipment and rigorous safety protocols to prevent fire hazards. The conventional process also suffers from moderate overall yields, typically ranging between 72% and 82%, largely due to losses during the refining and decolorization stages which are necessary to remove trace metal residues. These factors combine to create a manufacturing profile that is both capital intensive and operationally rigid.
The Novel Approach
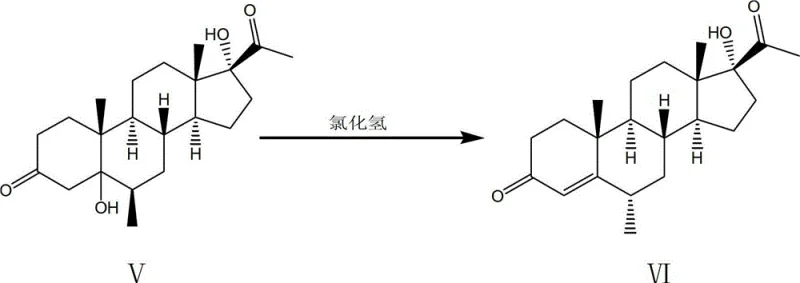
The innovative methodology presented in the patent data offers a decisive break from these constraints by introducing a six-step sequence that entirely eliminates the hydrogenation step. Instead of reducing a double bond with hydrogen gas, the new route employs a hydrogen chloride-catalyzed translocation reaction to achieve the desired 6-alpha methyl configuration. This chemical transformation is not only cheaper but also proceeds under much milder conditions, avoiding the violent heat release and potential deflation phenomena often observed in exothermic hydrogenation processes. The reported data indicates a dramatic improvement in efficiency, with the new route claiming a total recovery significantly higher than the traditional baseline. By utilizing common organic solvents like benzene and chloroform which are easier to recycle, the process further enhances its economic profile through reduced waste treatment costs and simplified solvent recovery loops.
Mechanistic Insights into Acid-Catalyzed Translocation and Grignard Addition
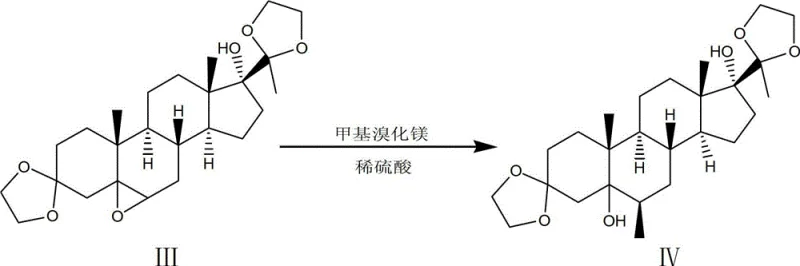
The core of this synthetic advancement lies in the precise orchestration of functional group interconversions, specifically the epoxidation and subsequent Grignard addition. In the second step, the protected ketal intermediate undergoes epoxidation using a peracetic acid solution buffered with anhydrous sodium acetate. This buffering is critical to prevent acid-catalyzed degradation of the sensitive steroid backbone while ensuring high conversion to the epoxide. Following this, the introduction of the methyl group at the 6-beta position is achieved via a Grignard reaction with methylmagnesium bromide in tetrahydrofuran. The mechanism involves the nucleophilic attack of the methyl anion on the epoxide ring, opening it to form a tertiary alcohol. This step is meticulously controlled at temperatures between 75°C and 78°C to ensure complete reaction while minimizing side products. The subsequent hydrolysis with dilute sulfuric acid quenches the excess Grignard reagent and stabilizes the intermediate for the next phase.
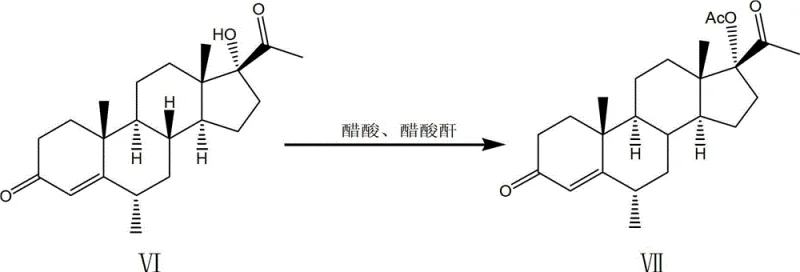
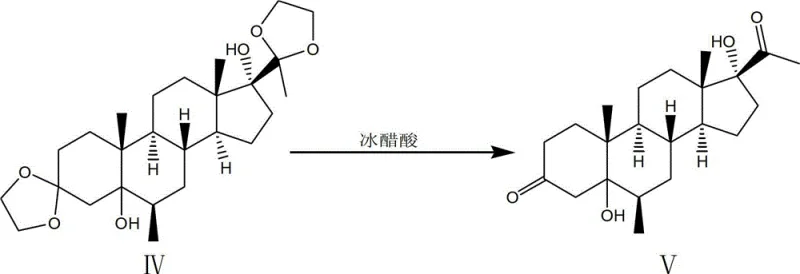
The true mechanistic brilliance, however, is reserved for the fifth step, where the 5-alpha, 17-alpha-dihydroxy-6-beta-methyl progesterone is converted into the 6-alpha-methyl isomer. Under the influence of hydrogen chloride in a chloroform-ethanol system, a translocation reaction occurs. This acid-catalyzed rearrangement likely proceeds through a carbocation intermediate that allows for the thermodynamic equilibration of the methyl group from the beta to the alpha position, which is the biologically active configuration required for Medroxyprogesterone Acetate. This eliminates the need for stereoselective hydrogenation, which is often difficult to control. The final acetylation using acetic anhydride and acetic acid, catalyzed by sulfosalicylic acid, then caps the 17-hydroxyl group to complete the synthesis. Each step is designed to maximize atom economy and minimize purification burdens.
How to Synthesize Medroxyprogesterone Acetate Efficiently
Implementing this synthesis requires strict adherence to the specified reaction parameters to ensure the high yields reported in the patent documentation. The process begins with the ketalization of the starting material, followed by the critical epoxidation and Grignard sequences described above. Operators must pay close attention to the temperature controls during the Grignard addition, maintaining the reflux between 75°C and 78°C for 8 to 12 hours to ensure full conversion. The subsequent deprotection using glacial acetic acid must be conducted at 95°C to 115°C to effectively remove the ketal protecting groups without degrading the newly formed alcohol functionalities. The translocation step is particularly sensitive to acid concentration and reaction time, requiring a balance to drive the isomerization to completion.
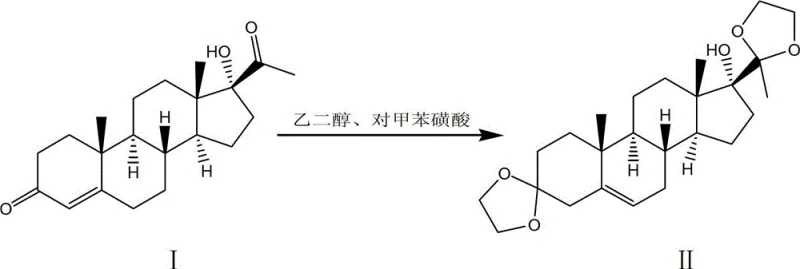
- Perform ketalization of 17 Alpha-hydroxy Progesterone with ethylene glycol and p-toluenesulfonic acid in benzene to form the protected ketal intermediate.
- Execute epoxidation using peracetic acid solution followed by a Grignard reaction with methylmagnesium bromide and subsequent hydrolysis.
- Conduct deprotection with glacial acetic acid, followed by hydrogen chloride-catalyzed translocation and final acetylation to yield the target API.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this non-hydrogenation route offers compelling strategic benefits that extend beyond simple unit cost calculations. The most immediate impact is the removal of the precious metal catalyst from the bill of materials. Palladium on Carbon is not only expensive to purchase but also incurs significant costs for recovery and disposal to meet environmental regulations. By eliminating this input, the manufacturing process becomes inherently more stable against commodity price shocks. Additionally, the solvents employed in this route, such as benzene and chloroform, are standard industrial chemicals with well-established recycling infrastructure. This facilitates a closed-loop solvent management system, drastically reducing the volume of hazardous waste generated and lowering the associated disposal fees. The mild reaction conditions also translate to lower energy consumption, as there is no need for high-pressure reactors or extreme cooling systems typically required for controlling exothermic hydrogenation.
- Cost Reduction in Manufacturing: The elimination of the palladium catalyst removes a major cost driver from the production equation. Without the need for expensive noble metals, the direct material cost is significantly lowered. Furthermore, the absence of metal residues simplifies the downstream purification process, reducing the need for activated carbon decolorization and complex filtration steps. This streamlining of the workflow leads to substantial operational savings and a more predictable cost structure for long-term contracts.
- Enhanced Supply Chain Reliability: Relying on precious metal catalysts introduces supply chain vulnerability, as the availability of palladium can be constrained by geopolitical factors and mining output. By switching to a chemical translocation method that uses readily available acids and solvents, the supply chain becomes more resilient. The raw materials for this process are commodity chemicals with multiple global suppliers, ensuring continuity of supply even during market disruptions. This reliability is crucial for maintaining consistent production schedules for critical hormone intermediates.
- Scalability and Environmental Compliance: The safety profile of this method makes it highly scalable. The avoidance of high-pressure hydrogenation removes a significant barrier to increasing batch sizes, allowing for efficient commercial scale-up from pilot plants to multi-ton production. Moreover, the reduced generation of heavy metal waste aligns with increasingly stringent environmental regulations. The ability to recycle solvents efficiently minimizes the environmental footprint, making the facility more compliant with green chemistry initiatives and reducing the risk of regulatory penalties.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. They are derived from the specific advantages and procedural details outlined in the patent literature, providing clarity on how this method compares to established industry standards. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this new process for large-scale manufacturing.
Q: How does this synthesis route eliminate the need for precious metal catalysts?
A: Unlike conventional methods requiring Palladium on Carbon (Pd/C) for hydrogenation, this novel route utilizes a hydrogen chloride-catalyzed translocation reaction. This chemical rearrangement achieves the necessary structural modification without expensive noble metals, drastically reducing raw material costs and simplifying waste treatment.
Q: What are the safety advantages of this method compared to traditional hydrogenation?
A: The process avoids high-pressure hydrogenation equipment and the associated risks of violent heat release or deflation phenomena. All reaction steps, including the critical translocation, proceed under mild atmospheric conditions with controlled temperatures, significantly enhancing production safety and operational stability.
Q: Why is the solvent recovery system considered more efficient in this protocol?
A: The protocol utilizes solvents such as benzene, chloroform, and tetrahydrofuran, which are explicitly noted for their ease of recycling compared to complex mixed solvent systems. This facilitates a closed-loop recovery process, minimizing chemical waste discharge and lowering the overall environmental compliance burden.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Medroxyprogesterone Acetate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the adoption of advanced synthetic routes like the one described in CN102911233A requires a partner with deep technical expertise and robust manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. Our facilities are equipped with state-of-the-art reactors capable of handling the specific solvent systems and temperature profiles required for this translocation chemistry. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Medroxyprogesterone Acetate meets the highest pharmacopeial standards, regardless of the synthetic route employed.
We invite pharmaceutical partners to engage with our technical procurement team to discuss how this optimized synthesis can benefit your specific supply chain. By leveraging our expertise, you can access a Customized Cost-Saving Analysis that quantifies the potential reductions in material and waste treatment costs for your operations. We encourage you to request specific COA data and route feasibility assessments to validate the performance of this non-hydrogenation method against your current benchmarks. Together, we can drive efficiency and reliability in the production of this vital hormonal intermediate.