Advanced Synthesis of 10-Methoxy Iminostilbene for High-Spec Pharmaceutical Manufacturing
Advanced Synthesis of 10-Methoxy Iminostilbene for High-Spec Pharmaceutical Manufacturing
The pharmaceutical industry constantly demands higher purity standards for key intermediates, particularly for antiepileptic drugs like oxcarbazepine and carbamazepine. Patent CN102807528A introduces a groundbreaking preparation method for 10-methoxy iminostilbene that addresses critical limitations in yield and purity found in legacy manufacturing processes. This technical insight report analyzes the novel four-step synthetic route which leverages dibromohydantoin (DBDMH) mediated bromination to achieve product purity exceeding 99% without the need for energy-intensive recrystallization steps. For R&D directors and procurement specialists, this methodology represents a significant leap forward in process chemistry, offering a robust pathway to high-quality active pharmaceutical ingredient (API) precursors.
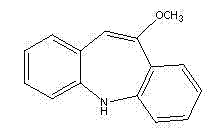
Traditionally, the synthesis of this critical intermediate has been plagued by inconsistent quality and excessive processing times. The new protocol described in the patent utilizes readily available iminostilbene-5-formyl halides as starting materials, reacting them through a sequence of esterification, controlled low-temperature bromination, elimination, and final hydrolysis. This approach not only streamlines the operational workflow but also fundamentally alters the impurity profile of the final bulk drug substance, ensuring compliance with stringent international pharmacopoeia standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods, such as those described in GB943277 and WO2005096709, suffer from inherent inefficiencies that impact both cost and quality. The legacy route depicted below typically involves a two-step reaction requiring extensive reaction times, often totaling over 30 hours for completion. More critically, these conventional pathways frequently result in yields of 65% or lower, with product purity struggling to reach 98%.
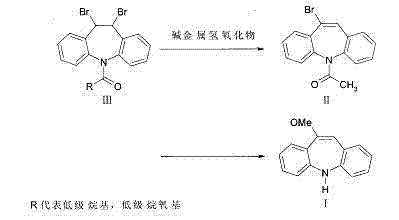
Furthermore, alternative methods utilizing sodium methylate directly on precursor IV have demonstrated severe issues with side reactions. Experimental data indicates that these older techniques often generate substantial amounts of dimer impurities, resulting in crude product purity as low as 70% to 80%. Such low-quality outputs necessitate complex and costly purification downstream, creating bottlenecks in the supply chain and increasing the overall cost of goods sold (COGS) for the final API manufacturer.
The Novel Approach
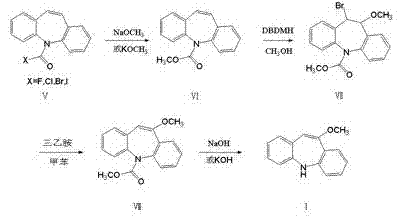
In stark contrast, the novel approach detailed in CN102807528A introduces a sophisticated control mechanism over the bromination step. By employing dibromohydantoin (DBDMH) at strictly controlled low temperatures ranging from -5°C to 5°C, the process ensures selective monobromination. This precision prevents the formation of unwanted by-products and dimers that plague earlier methods. The subsequent elimination step using triethylamine in toluene further refines the molecular structure, setting the stage for a high-yield hydrolysis.
The result is a streamlined process where the final product precipitates as glassy yellow crystals with purity consistently above 99%. This eliminates the need for additional recrystallization steps, drastically reducing solvent consumption and processing time. The ability to use mixtures of iminostilbene-5-formyl halides (chloride, bromide, fluoride, or iodine) also provides flexibility in raw material sourcing, allowing manufacturers to optimize costs based on market availability without compromising the chemical integrity of the synthesis.
Mechanistic Insights into DBDMH-Mediated Bromination and Elimination
The core innovation of this synthesis lies in the mechanistic control exerted during the bromination phase. Unlike traditional bromination agents that may lead to polybromination or radical side reactions, dibromohydantoin acts as a stable source of electrophilic bromine. When reacted with the iminostilbene-5-methyl formate intermediate in methanol at sub-zero temperatures, the reagent facilitates the addition of a single bromine atom across the double bond. This forms the 10-methoxy-11-bromo-10,11-dihydroiminostilbene-5-methyl formate intermediate with high regioselectivity.
Following the bromination, the elimination mechanism is driven by the presence of triethylamine in a non-polar solvent like toluene under reflux conditions. Triethylamine acts as a base to abstract a proton, triggering the elimination of hydrogen bromide (HBr) and restoring the double bond character essential for the stilbene structure. This step is crucial for establishing the correct conjugation in the final molecule. The subsequent hydrolysis using alkali metal hydroxides cleaves the methyl formate protecting group, revealing the free secondary amine and yielding the final 10-methoxy iminostilbene. This sequence ensures that impurities formed in earlier steps do not carry over, as the crystallization of intermediates acts as an in-process purification.
How to Synthesize 10-Methoxy Iminostilbene Efficiently
The synthesis protocol outlined in the patent provides a clear, scalable roadmap for producing high-purity 10-methoxy iminostilbene suitable for GMP manufacturing. The process begins with the esterification of formyl halides, followed by the critical low-temperature bromination and elimination steps. Each stage is monitored via HPLC to ensure reaction completion before proceeding, minimizing the risk of carrying unreacted starting materials into the final product. The detailed standardized synthesis steps below outline the precise stoichiometry and conditions required to replicate the high yields reported in the patent examples.
- Esterify iminostilbene-5-formyl halide with methanol and alkali metal alkoxide to form iminostilbene-5-methyl formate.
- React the formate with dibromohydantoin (DBDMH) at low temperature (-5 to 5°C) to obtain the brominated intermediate.
- Perform elimination using triethylamine in toluene under reflux, followed by hydrolysis with alkali metal hydroxide to yield the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route offers tangible economic and logistical benefits. The primary advantage stems from the optimized stoichiometry of the brominating agent. The patent explicitly notes that because only one bromine atom from the dibromohydantoin molecule participates in the reaction, the usage quantity of the reagent is effectively reduced by half compared to traditional dibromination routes. This direct reduction in reagent consumption translates to significant raw material cost savings and a smaller environmental footprint due to reduced chemical waste disposal.
- Cost Reduction in Manufacturing: The elimination of the recrystallization step is a major driver for cost efficiency. Traditional methods often require multiple purification cycles to meet purity specifications, consuming vast amounts of solvents and energy. By achieving >99% purity directly from the reaction mixture, this new method drastically simplifies the downstream processing. Additionally, the ability to use mixed formyl halides allows procurement teams to source the most cost-effective available halide derivatives without altering the process outcome, providing flexibility against market price fluctuations.
- Enhanced Supply Chain Reliability: The reliance on iminostilbene-5-formyl halides as starting materials addresses a critical vulnerability in the supply chain. Unlike specialized precursors used in older methods which may have limited suppliers, formyl halides are large-scale industrial chemicals with stable sources and established markets. This ensures a continuous supply of raw materials, reducing the risk of production stoppages due to material shortages. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, further securing supply continuity.
- Scalability and Environmental Compliance: The process is designed for scalability, utilizing common solvents like methanol and toluene which are easily recovered and recycled in standard chemical plants. The reduction in brominating agent usage by approximately 50% significantly lowers the load of halogenated waste, simplifying wastewater treatment and ensuring compliance with increasingly strict environmental regulations. The high yield (reported up to 97% in intermediate steps) maximizes the throughput of existing reactor capacity, allowing manufacturers to scale production from pilot batches to multi-ton commercial volumes with minimal capital investment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this method outperforms legacy technologies in terms of purity, yield, and operational efficiency.
Q: How does the new DBDMH method improve purity compared to traditional routes?
A: The novel method utilizes a controlled monobromination strategy using dibromohydantoin, which significantly reduces the formation of dimer impurities common in older methods, achieving purity levels exceeding 99% without the need for recrystallization.
Q: What are the cost advantages of using iminostilbene-5-formyl halide mixtures?
A: Using mixtures of formyl halides (chloride/bromide) allows for the utilization of stable, industrially available raw materials, avoiding the supply chain volatility associated with specialized precursors used in legacy processes.
Q: Does this process reduce hazardous waste generation?
A: Yes, the process is more atom-economical regarding the brominating agent. By ensuring only one bromine atom participates in the reaction rather than two, the consumption of the brominating reagent is effectively halved, reducing chemical waste.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 10-Methoxy Iminostilbene Supplier
At NINGBO INNO PHARMCHEM, we understand the critical role that high-purity intermediates play in the efficacy and safety of final pharmaceutical products. Our technical team has extensively analyzed the route described in CN102807528A and possesses the expertise to implement this advanced synthesis at an industrial scale. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to full-scale manufacturing is seamless. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 10-methoxy iminostilbene meets the highest global standards.
We invite pharmaceutical manufacturers and procurement leaders to engage with us for a Customized Cost-Saving Analysis. By leveraging this optimized synthetic route, we can help you reduce your overall cost of goods while improving supply security. Please contact our technical procurement team to request specific COA data and route feasibility assessments tailored to your project requirements. Let us partner with you to engineer a more efficient and reliable supply chain for your antiepileptic drug portfolio.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
