Advanced Spirofluorene Xanthene Derivatives: Scaling High-Purity Blue Emitters for Next-Gen OLEDs
The rapid evolution of flat panel display technology has necessitated the development of advanced organic electroluminescent materials that surpass the limitations of traditional liquid crystal displays. Patent CN103666455A introduces a groundbreaking class of novel spirofluorene xanthene derivatives designed specifically to address the critical demand for high-efficiency, stable blue light emitters in next-generation organic light-emitting diode (OLED) devices. This intellectual property outlines a robust synthetic strategy that functionalizes the 2-position of the spirofluorene xanthene core with various nitrogen-containing groups, such as secondary amines, pyrroles, indoles, carbazoles, and benzimidazoles. By leveraging a streamlined single-stage palladium-catalyzed coupling reaction, this technology enables the production of materials with exceptional thermal stability and morphological integrity, which are paramount for the longevity and performance of commercial display panels. For R&D directors and procurement specialists alike, this patent represents a significant leap forward in material science, offering a pathway to high-purity intermediates that can be reliably scaled for industrial mass production without compromising on the stringent quality standards required by the optoelectronics industry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex spiro-compounds for optoelectronic applications has been plagued by multi-step processes that suffer from low overall yields and difficult purification protocols. Traditional routes often require the protection and deprotection of functional groups, leading to excessive waste generation and increased consumption of hazardous solvents, which drives up both the environmental footprint and the manufacturing cost. Furthermore, conventional methods frequently struggle to achieve the high levels of purity necessary for OLED applications, where trace impurities can act as quenching sites that drastically reduce luminous efficiency and device lifespan. The reliance on harsh reaction conditions in older methodologies can also compromise the structural integrity of sensitive heterocyclic moieties, resulting in inconsistent batch-to-batch performance. These inefficiencies create significant bottlenecks in the supply chain, making it challenging for manufacturers to secure a steady stream of high-quality materials at a price point that supports the mass adoption of advanced display technologies.
The Novel Approach
In stark contrast to these legacy techniques, the methodology described in CN103666455A utilizes a direct, single-stage coupling reaction that dramatically simplifies the synthetic landscape. By employing a highly active palladium catalyst system in conjunction with strong bases like sodium tert-butoxide, the process facilitates the efficient formation of carbon-nitrogen bonds under relatively mild reflux conditions. This approach eliminates the need for tedious intermediate isolation steps, thereby reducing the total processing time and minimizing the exposure of reactive intermediates to potential degradation pathways. The result is a cleaner reaction profile that yields products with superior purity directly after standard column chromatography, significantly lowering the burden on downstream purification units. This technological shift not only enhances the economic viability of producing spirofluorene xanthene derivatives but also aligns perfectly with modern green chemistry principles by reducing solvent usage and energy consumption throughout the manufacturing lifecycle.
Mechanistic Insights into Palladium-Catalyzed Amination
The core of this innovative synthesis lies in the sophisticated mechanism of the palladium-catalyzed cross-coupling reaction, specifically a variation of the Buchwald-Hartwig amination tailored for sterically hindered spiro-systems. The catalytic cycle initiates with the oxidative addition of the palladium(0) species into the carbon-bromine bond of the 2-bromo-spirofluorene-9,9'-xanthene precursor, forming a reactive organopalladium intermediate. This step is crucial as it activates the otherwise inert aryl halide for subsequent nucleophilic attack. The presence of bulky phosphine ligands, such as tri-butyl phosphine, plays a pivotal role in stabilizing the active catalyst species and facilitating the reductive elimination step, which ultimately releases the desired amine-substituted product and regenerates the palladium(0) catalyst. This precise control over the catalytic cycle ensures high turnover numbers and minimizes the formation of homocoupling byproducts, which are common pitfalls in similar cross-coupling reactions involving complex aromatic systems.
Furthermore, the choice of reaction conditions is meticulously optimized to suppress the formation of impurities that could detrimentally affect the electronic properties of the final material. The use of anhydrous weak polar solvents like toluene creates an ideal environment for the solubility of both the organic substrates and the catalyst complex, ensuring homogeneous reaction kinetics. The strict exclusion of moisture and oxygen prevents the oxidation of the phosphine ligands and the deactivation of the palladium center, which are critical factors in maintaining consistent reaction rates. By carefully controlling the stoichiometry of the base and the amine substrate, the process effectively drives the equilibrium towards product formation while neutralizing the hydrogen bromide byproduct. This rigorous attention to mechanistic detail results in a product profile that meets the exacting standards of the electronic chemical industry, where even parts-per-million levels of impurities can lead to device failure.
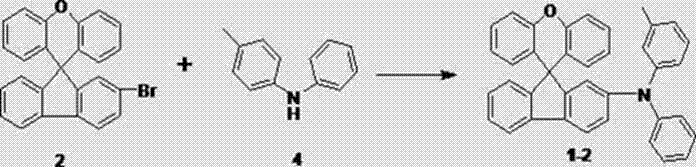
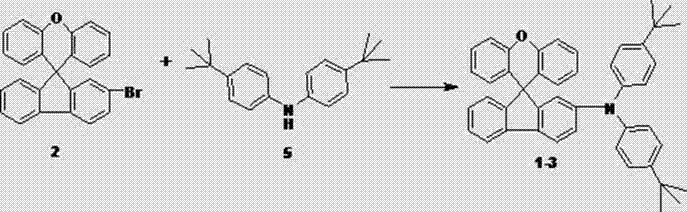
How to Synthesize Spirofluorene Xanthene Derivatives Efficiently
The practical implementation of this synthesis route is designed for seamless integration into existing chemical manufacturing infrastructure, requiring only standard reactor setups and common laboratory equipment. The process begins with the preparation of two distinct solutions: one containing the brominated spirofluorene xanthene core, the amine functional group, and the base, and another containing the pre-activated palladium catalyst system. These solutions are then combined under an inert atmosphere and heated to reflux, allowing the reaction to proceed to completion over a period of 5 to 20 hours depending on the specific steric demands of the amine substituent. Following the reaction, a straightforward workup procedure involving aqueous extraction and drying removes inorganic salts and catalyst residues, yielding a crude product that is easily purified via column chromatography using a dichloromethane and petroleum ether eluent system. Detailed standardized synthesis steps for specific derivatives are outlined in the guide below.
- Combine 2-bromo-spirofluorene-9,9'-xanthene with the chosen amine derivative and a strong base like sodium tert-butoxide in an anhydrous weak polar solvent such as toluene.
- Prepare a catalyst solution by dissolving an organic palladium catalyst (e.g., Pd(OAc)2) and an organic phosphine ligand (e.g., tri-butyl phosphine) in dry toluene.
- Mix the reactant and catalyst solutions, reflux the mixture for 5 to 20 hours, then perform standard workup including extraction, washing, drying, and column chromatography to isolate the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this single-stage synthesis technology translates into tangible strategic advantages that extend far beyond simple unit cost metrics. The elimination of multiple synthetic steps inherently reduces the complexity of the supply chain, minimizing the number of raw materials that need to be sourced, qualified, and inventory-managed. This simplification leads to a more resilient supply network that is less susceptible to disruptions caused by the shortage of niche intermediates. Moreover, the high efficiency of the reaction means that less reactor volume is required to produce the same amount of final product, effectively increasing the throughput capacity of existing manufacturing facilities without the need for significant capital expenditure on new equipment. These factors combine to create a robust and cost-effective production model that can scale rapidly to meet the surging global demand for high-performance OLED materials.
- Cost Reduction in Manufacturing: The streamlined nature of the single-stage process significantly lowers operational expenditures by reducing the consumption of solvents, energy, and labor hours associated with multi-step syntheses. By avoiding the isolation of unstable intermediates, the process minimizes material loss and waste disposal costs, leading to substantial overall savings in the cost of goods sold. Additionally, the high selectivity of the palladium catalyst reduces the need for expensive and time-consuming purification techniques, further enhancing the economic efficiency of the manufacturing operation. This cost structure allows suppliers to offer competitive pricing while maintaining healthy margins, providing a distinct advantage in the highly competitive electronic materials market.
- Enhanced Supply Chain Reliability: The use of commercially available and stable starting materials, such as 2-bromo-spirofluorene-9,9'-xanthene and common amine derivatives, ensures a secure and consistent supply of raw inputs. The robustness of the reaction conditions means that the process is less sensitive to minor variations in temperature or reagent quality, resulting in highly reproducible batches that meet strict quality specifications every time. This reliability is crucial for long-term supply agreements with major display manufacturers, who require guaranteed continuity of supply to maintain their own production schedules. By mitigating the risks associated with complex synthetic routes, this technology provides a stable foundation for building enduring partnerships between material suppliers and device fabricators.
- Scalability and Environmental Compliance: The simplicity of the reaction protocol facilitates easy scale-up from laboratory gram quantities to multi-tonne commercial production without encountering the engineering challenges often associated with exothermic or hazardous multi-step processes. The reduced solvent usage and waste generation align with increasingly stringent environmental regulations, helping manufacturers maintain compliance and avoid potential fines or shutdowns. Furthermore, the ability to recycle solvents and recover catalyst residues contributes to a more sustainable manufacturing footprint, which is becoming a key differentiator for suppliers seeking to partner with environmentally conscious global corporations. This scalability ensures that the technology can grow alongside the market, supporting the transition from niche applications to mass-market consumer electronics.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these novel spirofluorene xanthene materials. These answers are derived directly from the technical specifications and beneficial effects detailed in the patent documentation, providing clarity on performance metrics and process capabilities. Understanding these aspects is essential for stakeholders evaluating the feasibility of integrating these materials into their product development pipelines.
Q: What are the primary advantages of spirofluorene xanthene derivatives in OLED devices?
A: These derivatives offer superior luminous quantum efficiency, excellent charge transport properties, and remarkable thermal and morphological stability, making them ideal for long-lasting blue light emission in organic electroluminescent devices.
Q: How does the single-stage synthesis method impact production costs?
A: The single-stage method significantly simplifies the manufacturing process by eliminating multiple intermediate isolation steps, thereby reducing solvent consumption, energy usage, and overall operational complexity compared to traditional multi-step routes.
Q: Can these materials be used for applications other than light emission?
A: Yes, beyond serving as luminescent materials, these compounds function effectively as hole transport or electron transfer materials, broadening their utility in organic solar batteries, laser diodes, and field-effect transistors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Spirofluorene Xanthene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance organic electroluminescent materials play in the advancement of display technology. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of high-purity spirofluorene xanthene derivatives. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee that every batch meets stringent purity specifications required for OLED applications. We are committed to delivering not just chemicals, but comprehensive solutions that accelerate your time-to-market and enhance the performance of your final devices through superior material quality.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs through the adoption of this advanced synthesis technology. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. Please contact us today to request specific COA data and route feasibility assessments for your next project, and let us demonstrate how our expertise can drive value and innovation in your organization.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
