Advanced Synthesis of 9,10-Substituted Anthracene for High-Performance OLED Host Materials
Advanced Synthesis of 9,10-Substituted Anthracene for High-Performance OLED Host Materials
The rapid evolution of Organic Light Emitting Diode (OLED) technology has placed immense pressure on the supply chain for high-performance host materials, particularly those capable of emitting deep blue light with extended operational lifetimes. Patent CN110963876A introduces a groundbreaking preparation and purification method for 9,10-substituted anthracene derivatives, specifically targeting the synthesis of 9-(naphthalene-1-yl)-10-(4-(naphthalene-2-yl)phenyl)anthracene. This compound represents a critical class of blue fluorescent host materials where molecular aggregation must be strictly controlled to prevent efficiency roll-off and color purity degradation. The disclosed technology addresses the persistent industry challenge of achieving ultra-high chemical purity (>99.99%) while maintaining high synthetic yields, a balance that is often difficult to strike with conventional Suzuki coupling protocols.
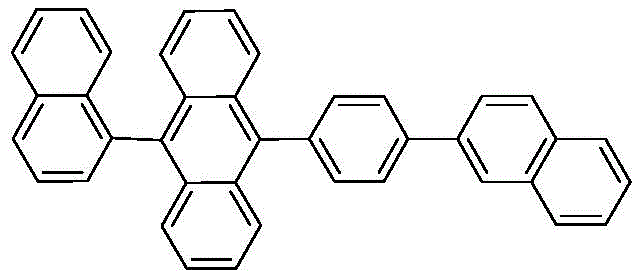
Traditional synthesis routes for such complex anthracene derivatives often suffer from the formation of stubborn impurities, including boric acid self-coupling products and debrominated by-products, which necessitate energy-intensive sublimation processes to meet device-grade specifications. The innovation detailed in this patent leverages a specialized Pd-132 catalyst system that dramatically suppresses these side reactions, enabling a cleaner reaction profile. By integrating a robust five-step synthetic pathway with a novel multi-stage recrystallization purification protocol, this method offers a viable solution for the commercial scale-up of complex OLED materials, ensuring that the final product meets the stringent single impurity limits of less than 100 ppm required for next-generation display applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 9,10-disubstituted anthracenes has relied heavily on standard palladium catalysts such as Pd(PPh3)4. While these catalysts are effective for general cross-coupling, they frequently exhibit insufficient selectivity when dealing with sterically hindered substrates like 9-bromoanthracene derivatives. The primary drawback lies in the generation of significant quantities of by-products, specifically homocoupling of the boronic acid species and hydrodeboronation, which complicates the downstream purification landscape. These impurities are structurally similar to the target molecule, making them exceptionally difficult to remove via standard crystallization techniques, often forcing manufacturers to rely on repeated vacuum sublimation. This reliance not only drives up energy consumption and production costs but also results in substantial material loss, thereby reducing the overall process yield and economic viability for mass production.
The Novel Approach
The methodology presented in CN110963876A fundamentally shifts the paradigm by introducing the Pd-132 catalyst for key coupling steps, which demonstrates superior activity and selectivity. This advanced catalytic system facilitates the coupling of bulky naphthyl and anthracene groups with remarkable efficiency, achieving conversion rates exceeding 97% while minimizing the formation of boric acid removal products to less than 1%. Furthermore, the process incorporates a sophisticated purification strategy that bypasses the immediate need for sublimation by utilizing a heat-preservation silica gel column followed by sequential recrystallization in toluene and DMF. This approach not only streamlines the workflow but also ensures that the final high-purity OLED material is obtained with minimal loss, directly addressing the cost and efficiency bottlenecks inherent in legacy manufacturing processes.
Mechanistic Insights into Pd-132 Catalyzed Suzuki Coupling
The core of this synthetic breakthrough lies in the strategic application of the Pd-132 catalyst during the formation of the carbon-carbon bonds at the 9 and 10 positions of the anthracene core. Unlike traditional phosphine ligands that may dissociate under harsh conditions leading to palladium black precipitation and reduced catalytic turnover, the ligand system in Pd-132 maintains stability throughout the reaction cycle. This stability is crucial for the coupling of Intermediate 4 (the boronic acid derivative) with Intermediate 1, where steric hindrance is significant. The catalyst promotes the oxidative addition of the aryl bromide and the subsequent transmetallation with the boronic acid species at a rapid rate, completing the reaction within just 1 to 2 hours under reflux conditions. This kinetic acceleration reduces the exposure time of sensitive intermediates to thermal stress, thereby limiting decomposition pathways that typically generate colored impurities detrimental to optical performance.
Impurity control is further enhanced by the specific reaction conditions employed, such as the dropwise addition of base and the use of phase transfer catalysts like tetrabutylammonium bromide in earlier steps. These additives facilitate the transport of reactive species across phase boundaries, ensuring homogeneous reaction kinetics that prevent localized high concentrations of reagents which could trigger side reactions. The subsequent purification mechanism relies on the differential solubility of the target anthracene derivative versus its impurities in specific solvent systems. By passing the crude product through a silica gel column maintained at elevated temperatures (60-80°C), polar impurities and catalyst residues are effectively retained, while the target molecule elutes cleanly. This is followed by crystallization steps that exploit the subtle solubility differences between the target and structural isomers, ensuring that the final electronic chemical meets the rigorous purity standards demanded by the display industry.
How to Synthesize 9-(naphthalene-1-yl)-10-(4-(naphthalene-2-yl)phenyl)anthracene Efficiently
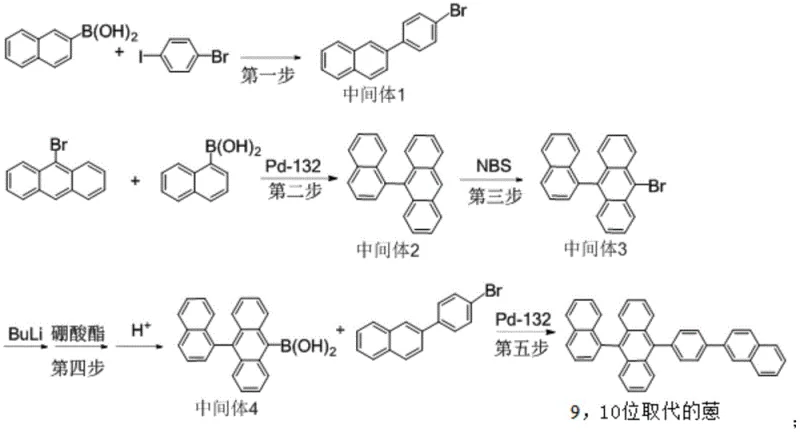
The synthesis of this complex anthracene derivative is achieved through a logical five-step sequence that balances yield and purity at every stage. The process begins with the preparation of key building blocks via Suzuki coupling, followed by functionalization of the anthracene core through bromination and lithiation-boration. The final assembly involves a second high-efficiency coupling reaction to join the two major fragments. Detailed operational parameters, including precise molar ratios, temperature controls, and solvent volumes, are critical for replicating the high yields reported in the patent. For a comprehensive guide on executing this synthesis with optimal results, please refer to the standardized protocol below.
- Perform initial Suzuki coupling of 2-naphthalene boric acid and p-bromoiodobenzene to form Intermediate 1.
- Execute Pd-132 catalyzed Suzuki coupling between 9-bromoanthracene and 1-naphthalene boric acid to yield Intermediate 2.
- Conduct bromination of Intermediate 2 with NBS, followed by lithiation and boration to generate boronic acid Intermediate 4.
- Finalize synthesis via Pd-132 catalyzed coupling of Intermediate 4 and Intermediate 1, followed by rigorous toluene/DMF recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers tangible benefits that extend beyond mere technical specifications. The primary advantage lies in the drastic simplification of the purification workflow, which traditionally acts as a bottleneck in fine chemical manufacturing. By eliminating the immediate dependency on multiple sublimation cycles and reducing the burden on chromatographic separation, the process significantly lowers the operational expenditure associated with energy and consumables. This efficiency gain translates directly into a more competitive cost structure for the final OLED terminal material, allowing buyers to secure high-quality inputs without the premium pricing often associated with ultra-pure electronic chemicals.
- Cost Reduction in Manufacturing: The utilization of the Pd-132 catalyst leads to substantially higher conversion rates and minimizes the formation of difficult-to-remove impurities. This high selectivity means that less raw material is wasted in side products, and the downstream purification requires fewer cycles to achieve the target purity. Consequently, the overall consumption of expensive reagents and solvents is optimized, driving down the unit cost of production. Furthermore, the avoidance of extensive sublimation steps reduces energy consumption and equipment wear, contributing to long-term operational savings and a more sustainable manufacturing footprint.
- Enhanced Supply Chain Reliability: The robustness of the synthetic route, characterized by mild reaction conditions and the use of commercially available solvents like toluene and ethanol, ensures high process reliability. This stability reduces the risk of batch failures due to sensitive reaction parameters, thereby guaranteeing consistent output volumes. For supply chain planners, this predictability is invaluable, as it mitigates the risk of shortages and allows for more accurate forecasting. The ability to produce device-grade material through crystallization rather than relying solely on sublimation also diversifies the processing capabilities, adding resilience to the supply network against equipment downtime.
- Scalability and Environmental Compliance: The process is explicitly designed with industrial scale-up in mind, utilizing standard reactor configurations and avoiding exotic or highly hazardous reagents. The streamlined purification process generates less waste solvent and solid residue compared to traditional methods that rely heavily on chromatography. This reduction in waste volume simplifies effluent treatment and aligns with increasingly stringent environmental regulations. The scalability of the method ensures that as demand for blue OLED materials grows, production capacity can be expanded rapidly without the need for complex process re-engineering, securing a stable long-term supply for downstream device manufacturers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of this 9,10-substituted anthracene derivative. These answers are derived directly from the experimental data and technical disclosures within the patent documentation, providing clarity on the process capabilities and product specifications.
Q: What distinguishes the Pd-132 catalyst from traditional Pd(PPh3)4 in this synthesis?
A: The Pd-132 catalyst significantly reduces side reactions such as boric acid self-coupling and debromination, resulting in fewer impurities and higher conversion rates compared to traditional tetrakis catalysts.
Q: How is the >99.99% purity achieved without extensive sublimation?
A: The process utilizes a specialized three-stage purification involving heat-preservation silica gel chromatography followed by sequential recrystallization in toluene and DMF, effectively removing trace impurities to device-grade standards.
Q: Is this synthetic route scalable for industrial production?
A: Yes, the method employs robust reaction conditions, common solvents like toluene and ethanol, and avoids extremely sensitive reagents, making it highly suitable for large-scale commercial manufacturing of OLED intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 9,10-Substituted Anthracene Supplier
The technological advancements detailed in CN110963876A underscore the critical importance of precise catalytic control and advanced purification in the production of next-generation display materials. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to deliver exceptional value to our global partners. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of 9,10-substituted anthracene meets the exacting standards required for high-performance OLED devices. We understand that consistency is key in the electronics sector, and our commitment to quality assurance guarantees a supply chain you can trust.
We invite potential partners to engage with our technical team to explore how this optimized synthesis route can benefit your specific application requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this high-efficiency process. We encourage you to contact our technical procurement team today to obtain specific COA data and route feasibility assessments, ensuring that your project moves forward with the highest quality materials and the most efficient supply strategy available in the market.
