Advanced Manufacturing of Brivaracetam Intermediate via Novel Cyclization Route
Advanced Manufacturing of Brivaracetam Intermediate via Novel Cyclization Route
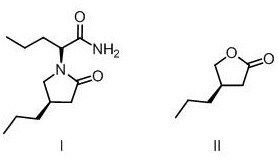
The global demand for third-generation antiepileptic drugs continues to surge, placing immense pressure on supply chains to deliver high-quality active pharmaceutical ingredients (APIs) and their precursors efficiently. Patent CN112521352A, published in March 2021, introduces a significant technological breakthrough in the preparation of Brivaracetam, specifically focusing on the synthesis of its critical chiral intermediate, (R)-4-propyl-dihydrofuran-2-one. This molecule serves as the structural cornerstone for the final drug substance, dictating the stereochemical integrity and therapeutic efficacy of the medication. For R&D directors and procurement strategists, understanding this novel pathway is essential, as it represents a shift away from complex resolution techniques toward a more direct, chiral-pool-based synthesis. The patent outlines a robust four-step sequence that transforms readily available starting materials into the target lactone with impressive yield and purity profiles, addressing key pain points in modern pharmaceutical manufacturing such as cost, scalability, and environmental impact.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral pyrrolidinone derivatives like the Brivaracetam intermediate has been plagued by inefficiencies inherent to traditional asymmetric synthesis or resolution strategies. Conventional routes often rely heavily on the use of expensive chiral catalysts or auxiliaries that require rigorous removal steps to meet stringent regulatory limits on residual metals. Furthermore, many legacy processes involve late-stage resolution of racemic mixtures, which theoretically caps the maximum yield at 50% unless dynamic kinetic resolution is employed, a technique that adds significant complexity and cost. These methods frequently necessitate the use of hazardous reagents and generate substantial volumes of waste solvent, creating bottlenecks for supply chain heads who must manage disposal costs and environmental compliance. The reliance on transition metals also introduces risks of batch-to-batch variability in impurity profiles, complicating the validation process for regulatory filings. Consequently, finding a reliable pharmaceutical intermediate supplier who can navigate these complexities while maintaining cost competitiveness has become a primary challenge for generic drug manufacturers.
The Novel Approach
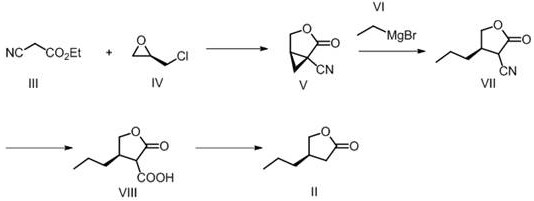
The methodology disclosed in CN112521352A offers a compelling alternative by utilizing a chiral pool strategy centered on R-epichlorohydrin. This approach fundamentally alters the economic landscape of the synthesis by introducing the required stereocenter at the very beginning of the sequence, thereby eliminating the need for downstream resolution. As illustrated in the reaction scheme below, the process begins with a base-catalyzed condensation to form a cyclic precursor, followed by a Grignard addition to install the propyl side chain. This linear progression avoids the thermodynamic equilibria issues associated with racemization. The subsequent hydrolysis and thermal lactonization steps are designed to be telescoped or performed in standard reactors, significantly simplifying the operational workflow. By shifting the complexity to the selection of high-quality starting materials rather than complex catalytic cycles, this novel approach ensures a more predictable impurity profile and facilitates easier scale-up from kilogram to multi-ton production scales.
Mechanistic Insights into Chiral Pool Synthesis and Lactonization
The core of this synthetic innovation lies in the precise orchestration of nucleophilic substitutions and organometallic additions. In the initial step, ethyl cyanoacetate reacts with R-epichlorohydrin in the presence of sodium ethoxide. The base deprotonates the active methylene group of the cyanoacetate, generating a nucleophile that attacks the epoxide ring of the chiral halohydrin. This ring-opening event is stereospecific, preserving the chirality of the epoxide carbon and setting the stage for the subsequent intramolecular cyclization that forms the five-membered ring structure of intermediate (V). The choice of sodium ethoxide in ethanol is critical here, as it provides a homogeneous reaction medium that promotes high conversion rates while minimizing side reactions such as polymerization of the epoxide. Following this, the introduction of the alkyl chain via a Grignard reagent (specifically propyl magnesium bromide as inferred from the final structure, though ethyl magnesium bromide is cited in examples) requires strict temperature control at -30°C to -20°C. This low-temperature regime is essential to prevent the decomposition of the sensitive nitrile-ester functionality and to ensure regioselective addition to the carbonyl or nitrile group, depending on the specific mechanistic pathway intended to generate the ketone precursor.
Following the organometallic step, the transformation of intermediate (VII) to the final lactone (II) involves a two-stage functional group manipulation. First, acidic hydrolysis using concentrated sulfuric acid at elevated temperatures (100-150°C) converts both the nitrile and ester moieties into carboxylic acid functionalities. This harsh condition is necessary to overcome the stability of the nitrile group, ensuring complete conversion to the dicarboxylic acid or keto-acid intermediate (VIII). The final step is a thermal lactonization performed in a high-boiling solvent like toluene at reflux (100-120°C). This dehydration reaction is driven by entropy and the stability of the five-membered lactone ring. From a quality control perspective, this thermal step acts as a self-purification mechanism; volatile impurities and residual solvents are removed during the reflux and subsequent distillation, resulting in a final product with purity levels exceeding 98%. This mechanistic robustness is vital for reducing lead time for high-purity pharmaceutical intermediates, as it minimizes the need for extensive chromatographic purification in the final stages.
How to Synthesize (R)-4-propyl-dihydrofuran-2-one Efficiently
Implementing this synthesis requires careful attention to reaction parameters, particularly temperature gradients and stoichiometry, to maximize yield and safety. The process is divided into four distinct operational units: alkylation/cyclization, Grignard addition, acid hydrolysis, and thermal lactonization. Each step builds upon the previous one, requiring efficient workup procedures to remove inorganic salts and byproducts before proceeding. For instance, the removal of copper salts (if used as a co-catalyst in specific variations) or magnesium salts after the Grignard quench is critical to prevent interference with the subsequent acid hydrolysis. The detailed standardized synthetic steps, including specific molar ratios, solvent volumes, and exact temperature ramps derived from the patent examples, are outlined in the guide below to assist process chemists in replicating this high-efficiency route.
- React ethyl cyanoacetate with R-epichlorohydrin in sodium ethoxide solution to form the cyclic intermediate (V).
- Perform a Grignard reaction using an organomagnesium reagent on compound (V) to introduce the alkyl side chain, yielding compound (VII).
- Hydrolyze the nitrile and ester groups of compound (VII) under acidic conditions to generate the carboxylic acid derivative (VIII).
- Heat compound (VIII) in a high-boiling solvent like toluene to induce intramolecular esterification, forming the final lactone (II).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible strategic benefits beyond mere technical feasibility. The primary advantage lies in the significant reduction of raw material costs associated with chiral sourcing. By utilizing R-epichlorohydrin, a commodity chemical produced on a massive industrial scale, the process bypasses the need for custom-synthesized chiral building blocks or expensive enzymatic resolutions. This shift drastically simplifies the supply chain, reducing dependency on niche suppliers and mitigating the risk of raw material shortages. Furthermore, the avoidance of precious metal catalysts (in the preferred embodiments described in the summary) eliminates the costly and time-consuming heavy metal scavenging steps typically required to meet ICH Q3D guidelines. This not only lowers the direct cost of goods sold (COGS) but also accelerates the batch release cycle by simplifying the analytical testing protocol for residual metals.
- Cost Reduction in Manufacturing: The economic model of this process is strengthened by the use of conventional solvents such as ethanol, tetrahydrofuran, and toluene, which are inexpensive and easily recycled. The elimination of chiral chromatography or enzymatic resolution steps represents a substantial cost saving, as these are often the most expensive unit operations in chiral synthesis. Additionally, the high overall yield reported in the patent examples suggests a more efficient atom economy, meaning less raw material is wasted as byproduct. This efficiency translates directly to lower waste disposal costs and a reduced environmental footprint, aligning with the growing corporate mandates for sustainable manufacturing practices in the fine chemical sector.
- Enhanced Supply Chain Reliability: Sourcing reliability is paramount for long-term API production contracts. Since the key starting materials are bulk chemicals with established global supply networks, the risk of supply disruption is minimized compared to routes relying on specialized, low-volume intermediates. The robustness of the reaction conditions, which tolerate standard industrial equipment rather than requiring specialized high-pressure or cryogenic vessels (beyond standard jacketed reactors), further enhances supply continuity. This accessibility allows for multi-vendor sourcing strategies for raw materials, giving procurement teams greater leverage in negotiations and ensuring that production schedules remain uninterrupted even during market fluctuations.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, with reaction conditions that are manageable in large-scale stainless steel reactors. The use of aqueous workups and standard extractions facilitates the handling of large volumes without requiring exotic separation technologies. From an environmental standpoint, the reduction in heavy metal usage and the potential for solvent recovery contribute to a greener process profile. This is increasingly important for meeting the rigorous environmental, social, and governance (ESG) criteria set by major pharmaceutical buyers. The ability to produce high-purity material with fewer purification steps also reduces the volume of hazardous waste generated per kilogram of product, simplifying regulatory compliance and waste management logistics.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. They are derived from a close analysis of the experimental data and the stated objectives of the invention, providing clarity for stakeholders evaluating this route for potential licensing or contract manufacturing opportunities. Understanding these nuances is critical for making informed decisions about process adoption and risk assessment.
Q: What is the key advantage of using R-epichlorohydrin in this synthesis?
A: Using R-epichlorohydrin leverages the 'chiral pool' strategy, introducing the necessary stereochemistry early in the synthesis without requiring expensive chiral catalysts or resolution steps.
Q: How does this method address impurity control?
A: The process utilizes distinct separation steps including extraction and column chromatography between stages, and the final thermal lactonization allows for purification via distillation, ensuring high purity (up to 98.9%).
Q: Is this process suitable for large-scale industrial production?
A: Yes, the method avoids exotic reagents and uses standard solvents like ethanol, THF, and toluene. The reaction conditions, while requiring specific temperature controls (-30°C to 150°C), are compatible with standard stainless steel reactors used in fine chemical manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Brivaracetam Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires more than just chemical knowledge; it demands engineering excellence and rigorous quality assurance. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising yields seen in the lab are faithfully reproduced on an industrial scale. We understand the critical nature of stereochemical purity in antiepileptic drugs and employ stringent purity specifications alongside our rigorous QC labs to guarantee that every batch of Brivaracetam intermediate meets the highest global standards. Our facility is equipped to handle the specific thermal and cryogenic requirements of this synthesis, providing a seamless bridge between R&D innovation and market supply.
We invite you to collaborate with us to optimize this pathway for your specific production needs. By leveraging our technical expertise, we can provide a Customized Cost-Saving Analysis tailored to your volume requirements, identifying further opportunities for process intensification and cost reduction. We encourage potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments. Let us help you secure a stable, high-quality supply of this critical intermediate, ensuring your downstream API production remains competitive and compliant in the global marketplace.
