Scalable Manufacturing of High-Purity Fexofenadine Intermediate via Novel Base-Catalyzed Route
The pharmaceutical industry continuously seeks robust and cost-effective pathways for producing critical antihistamine intermediates, particularly for second-generation drugs like Fexofenadine Hydrochloride. Patent CN101585762A introduces a transformative synthetic methodology for 2-(4-cyclopropoxycarbonylphenyl)-2-methylpropanoic acid, a pivotal building block in the manufacture of this widely prescribed medication. Unlike traditional routes that rely on hazardous oxidants or scarce transition metals, this innovation leverages a mild, base-catalyzed cyclization strategy that dramatically simplifies the production workflow. The structural integrity and chemical identity of this high-value intermediate are defined by its unique cyclopropyl-carbonyl motif attached to a dimethyl-substituted phenylacetic acid backbone.
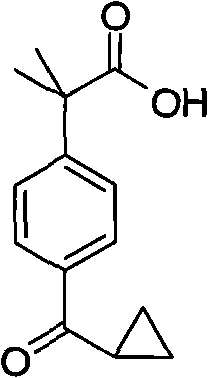
This novel approach not only addresses the longstanding challenges of low yields and environmental toxicity associated with legacy processes but also establishes a new benchmark for operational efficiency in fine chemical manufacturing. By utilizing readily available alkali metal hydroxides and common alcoholic solvents, the process ensures a stable supply chain free from the volatility of precious metal markets. For R&D directors and procurement specialists alike, understanding the mechanistic nuances and commercial implications of this patent is essential for optimizing the sourcing strategies of next-generation allergy medications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-(4-cyclopropoxycarbonylphenyl)-2-methylpropanoic acid has been plagued by inefficient multi-step sequences that impose significant burdens on both cost and environmental compliance. Prior art, such as the methods disclosed in US2002007068(A1) and various Indian patent filings, typically necessitates a cumbersome five-step reaction sequence involving the initial formation of esters followed by complex hydrolysis and cyclopropanation stages. These conventional pathways often suffer from mediocre overall yields, frequently reported in the range of 28% to 40%, which drastically inflates the cost of goods sold due to material loss at each stage. Furthermore, older oxidation-based strategies, like those described in U.S. Pat 6242606(B1), rely heavily on the use of ruthenium trichloride, a precious metal catalyst that introduces substantial expense and requires rigorous removal protocols to meet pharmaceutical purity standards. The reliance on strong oxidants such as potassium permanganate or sodium nitrite in these legacy methods also generates significant quantities of hazardous waste, creating severe disposal challenges and increasing the ecological footprint of the manufacturing facility.
The Novel Approach
In stark contrast, the methodology outlined in CN101585762A streamlines the synthesis into a concise two-step procedure that bypasses the need for expensive catalysts and harsh oxidizing agents entirely. The core innovation lies in the direct intramolecular cyclization of a chlorobutyryl precursor using a simple alkali metal hydroxide in an alcoholic medium, effectively closing the cyclopropane ring under mild thermal conditions between 20°C and 50°C. This is immediately followed by a hydrolysis step that converts the Weinreb amide intermediate directly into the target carboxylic acid, eliminating the need for separate esterification and saponification steps found in older routes. The result is a process that boasts significantly higher yields, often exceeding 90% in the cyclization step and maintaining high efficiency through hydrolysis, thereby maximizing atom economy. By removing the dependency on ruthenium and permanganate, this novel approach not only reduces raw material costs but also simplifies the workup procedure, leading to a cleaner crude product that requires less intensive purification efforts.
Mechanistic Insights into Base-Catalyzed Cyclization and Amide Hydrolysis
The chemical elegance of this synthesis is rooted in a dual-mechanism pathway that prioritizes selectivity and mild reaction conditions to preserve the integrity of the sensitive cyclopropyl ring. In the first stage, the alkali metal hydroxide acts as a base to deprotonate the alpha-position or facilitate a nucleophilic attack by the enolate equivalent on the terminal chloride of the butyryl side chain, driving the intramolecular substitution that forms the three-membered cyclopropane ring. This cyclization is conducted in alcoholic solvents like methanol or ethanol, which serve both as the reaction medium and potentially as proton donors to stabilize intermediates, ensuring that the reaction proceeds smoothly at temperatures as low as 30°C without inducing ring-opening side reactions. The use of N-methyl-N-methoxy-2-[4-(4-chlorobutyryl)phenyl]-2-methylpropanamide as the starting material is critical, as the Weinreb amide functionality provides a stable handle that resists premature hydrolysis during the basic cyclization phase, allowing the ring closure to occur with high fidelity.
Following the successful formation of the cyclopropyl-containing amide, the second mechanistic phase involves the hydrolysis of the Weinreb amide to the corresponding carboxylic acid under reflux conditions. The addition of excess alkali metal hydroxide in a higher boiling alcohol, such as tert-butanol or isopropanol, facilitates the nucleophilic attack of the hydroxide ion on the carbonyl carbon of the amide. This step cleaves the N-O bond and releases the N,O-dimethylhydroxylamine byproduct, ultimately yielding the free carboxylic acid after acidic workup. A key advantage of this mechanism is the suppression of impurities; because the reaction avoids strong oxidants, there is no risk of over-oxidation of the aromatic ring or the cyclopropyl moiety, which are common degradation pathways in ruthenium-catalyzed methods. The precise control of pH during the final isolation step, adjusting to pH 3 with hydrochloric acid, ensures that the product precipitates or extracts efficiently while leaving water-soluble impurities behind, resulting in a final API intermediate with a superior impurity profile.
How to Synthesize 2-(4-Cyclopropoxycarbonylphenyl)-2-Methylpropanoic Acid Efficiently
The execution of this synthesis requires careful attention to stoichiometry and temperature control to maximize the conversion of the chlorobutyryl precursor into the desired cyclopropyl acid. The process begins with the preparation of a concentrated alkali metal hydroxide solution in an anhydrous alcoholic solvent, into which the chlorobutyryl amide substrate is added dropwise to manage the exotherm and ensure uniform mixing. Detailed standard operating procedures regarding specific molar ratios, solvent volumes, and agitation speeds are critical for reproducibility and are essential for any technical team looking to implement this route at scale. For a comprehensive breakdown of the exact experimental parameters, including specific gram-scale examples and workup details, please refer to the standardized synthesis guide below.
- Cyclization: React N-methyl-N-methoxy-2-[4-(4-chlorobutyryl)phenyl]-2-methylpropanamide with alkali metal hydroxide in alcohol solvent at 20-50°C for 10-30 hours to form the cyclopropyl ring.
- Hydrolysis: Reflux the resulting amide intermediate with alkali metal hydroxide in alcohol solvent for 20-40 hours to cleave the amide bond.
- Isolation: Adjust pH to 3 using hydrochloric acid, extract with dichloromethane, and purify to obtain the final carboxylic acid product.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this base-catalyzed synthesis route offers profound advantages that extend far beyond simple yield improvements, fundamentally altering the cost structure and risk profile of the supply chain. By eliminating the requirement for ruthenium trichloride and other precious metal catalysts, manufacturers can decouple their production costs from the volatile fluctuations of the rare earth and precious metal markets, ensuring more predictable budgeting and pricing stability for long-term contracts. Furthermore, the reduction in reaction steps from five or more down to just two significantly compresses the manufacturing cycle time, allowing for faster turnover of reactor vessels and increased annual production capacity without the need for capital-intensive infrastructure expansion. The avoidance of hazardous oxidants like potassium permanganate also translates to substantial savings in waste treatment and environmental compliance costs, as the effluent streams are far less toxic and easier to neutralize compared to those generated by traditional oxidation methods.
- Cost Reduction in Manufacturing: The economic impact of switching to this novel route is driven primarily by the complete removal of expensive catalytic systems and the drastic reduction in solvent usage associated with fewer purification steps. Without the need for specialized ligands or precious metals, the raw material bill of materials is significantly lowered, while the high yields observed in the cyclization step minimize the loss of valuable starting materials. Additionally, the simplified workup procedure, which relies on standard extraction and filtration rather than complex chromatographic separations often needed to remove metal residues, reduces labor hours and consumable costs, leading to a leaner and more cost-efficient production model.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as sodium hydroxide, potassium hydroxide, and common alcohols ensures that the supply chain is robust and resilient against disruptions that might affect the availability of specialized reagents. Since these base materials are produced globally in massive quantities, the risk of supply shortages is negligible, providing procurement managers with the confidence to secure long-term supply agreements for critical pharmaceutical intermediates. The scalability of the process further enhances reliability, as the reaction conditions are mild enough to be performed in standard glass-lined or stainless steel reactors found in most multipurpose chemical plants, facilitating easy technology transfer between different manufacturing sites.
- Scalability and Environmental Compliance: The environmental profile of this synthesis aligns perfectly with modern green chemistry principles, making it an attractive option for companies aiming to reduce their carbon footprint and meet stringent regulatory standards. The absence of heavy metal waste streams simplifies the permitting process for new production lines and reduces the liability associated with hazardous waste disposal. Moreover, the mild reaction temperatures and atmospheric pressure conditions enhance operational safety, lowering the risk of thermal runaways and allowing for safer scale-up from pilot plant to commercial tonnage production, thereby ensuring a continuous and uninterrupted supply of high-quality intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method, drawing directly from the specific advantages and procedural details outlined in the patent literature. Understanding these aspects is crucial for technical teams evaluating the feasibility of adopting this route for their own manufacturing portfolios. The answers provided reflect the consensus on the operational benefits and chemical realities of this base-catalyzed approach.
Q: What are the primary advantages of this synthesis method over traditional oxidation routes?
A: This method eliminates the need for expensive and toxic precious metal catalysts like ruthenium trichloride or harsh oxidants like potassium permanganate, significantly reducing environmental impact and raw material costs while improving overall yield.
Q: How does this process ensure high purity for pharmaceutical applications?
A: By utilizing a mild base-catalyzed cyclization followed by controlled hydrolysis, the process avoids the formation of complex oxidative by-products common in older methods, simplifying downstream purification and ensuring stringent purity specifications.
Q: Is this synthetic route suitable for large-scale commercial production?
A: Yes, the use of common solvents like methanol and ethanol, along with inexpensive alkali metal hydroxides, makes the process highly scalable and economically viable for industrial manufacturing without requiring specialized high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(4-Cyclopropoxycarbonylphenyl)-2-Methylpropanoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global supply of life-saving antihistamines, and we have positioned ourselves as a leader in the commercialization of advanced synthetic routes like the one described in CN101585762A. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of 2-(4-cyclopropoxycarbonylphenyl)-2-methylpropanoic acid meets the exacting standards required by top-tier pharmaceutical companies worldwide.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis that demonstrates exactly how switching to our optimized manufacturing process can improve your bottom line. By contacting our technical procurement team today, you can request specific COA data, route feasibility assessments, and sample quantities to validate the superior quality and consistency of our supply. Let us partner with you to secure a sustainable, cost-effective, and reliable source of this essential Fexofenadine intermediate for your future production needs.
