Advanced Metal-Free Synthesis of Quinazolinedione Derivatives for Pharmaceutical Manufacturing
Advanced Metal-Free Synthesis of Quinazolinedione Derivatives for Pharmaceutical Manufacturing
The pharmaceutical industry is constantly seeking greener, more efficient pathways to synthesize critical heterocyclic scaffolds, particularly those serving as core structures for antihypertensive and anti-diabetic medications. A significant breakthrough in this domain is documented in Chinese Patent CN111454222B, which discloses a novel method for synthesizing 2,4-(1H,3H)-quinazolinedione and its derivatives. This technology leverages carbon dioxide (CO2) as a sustainable C1 building block, reacting it with readily available aminobenzonitrile compounds. Unlike traditional methods that rely on toxic phosgene or harsh high-pressure metal catalysis, this invention utilizes a specialized organic base phenolate catalyst system. This approach not only aligns with global green chemistry initiatives by fixing CO2 but also operates under remarkably mild conditions, typically between 40°C and 120°C, achieving high yields without the need for transition metals. For R&D directors and procurement specialists, this represents a pivotal shift towards safer, more cost-effective, and environmentally compliant manufacturing processes for valuable pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2,4-(1H,3H)-quinazolinedione has been fraught with significant operational and safety challenges. Conventional routes often involve the condensation of anthranilic acid with urea or the reaction of anthranilamide with phosgene. The use of phosgene, a highly toxic and regulated gas, imposes severe safety burdens on manufacturing facilities, requiring specialized containment equipment and rigorous hazard management protocols. Furthermore, alternative methods utilizing metal carbonates or ionic liquids frequently demand extreme reaction conditions, such as high temperatures and pressures exceeding 2.0 MPa, to achieve acceptable conversion rates. These harsh parameters not only escalate energy consumption but also complicate the downstream processing, as removing trace metal residues from the final active pharmaceutical ingredient (API) intermediate is both costly and technically demanding. Additionally, many existing catalytic systems suffer from poor atom economy and generate substantial waste streams, making them increasingly untenable in an era of strict environmental regulations and rising disposal costs.
The Novel Approach
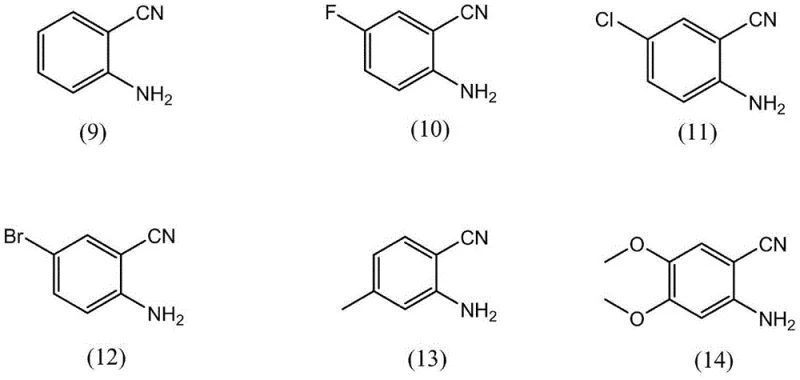
The methodology outlined in patent CN111454222B offers a transformative solution by employing a metal-free organic salt catalyst derived from guanidines or amidines paired with substituted phenols. This system enables the direct cycloaddition of CO2 to aminobenzonitriles in a single step, bypassing the need for toxic reagents like phosgene. The reaction proceeds efficiently at atmospheric or low pressures (0.1-2.0 MPa) and moderate temperatures, significantly reducing the energy footprint of the process. Crucially, the absence of transition metals eliminates the risk of heavy metal contamination, thereby simplifying the purification workflow and ensuring the final product meets stringent quality specifications for pharmaceutical use. The versatility of this approach is demonstrated by its compatibility with a wide range of substrates, including those with halogen, alkyl, and alkoxy substituents, as illustrated by the diverse array of aminobenzonitrile starting materials compatible with this chemistry.
Mechanistic Insights into Organic Base Phenolate Catalysis
The efficacy of this synthesis relies on the unique cooperative activation provided by the organic base phenolate catalyst. The cationic component, typically a protonated guanidine or amidine (such as DBU or TBD), acts as a strong hydrogen bond donor and electrostatic stabilizer, while the phenolate anion serves as a nucleophilic activator. This dual-activation mechanism facilitates the insertion of the thermodynamically stable CO2 molecule into the nitrile group of the aminobenzonitrile substrate. The phenolate anion likely activates the CO2 or the nitrile moiety, lowering the energy barrier for the initial nucleophilic attack. Subsequently, the intramolecular cyclization occurs to form the quinazolinedione ring. The steric bulk of substituents on the phenol ring, such as tert-butyl groups in catalyst variants like structure (6) or (8), plays a critical role in modulating the basicity and solubility of the catalyst, preventing deactivation and enhancing turnover numbers. This precise tuning of the catalyst structure allows for high selectivity and minimizes side reactions, ensuring a clean impurity profile which is paramount for API intermediate production.
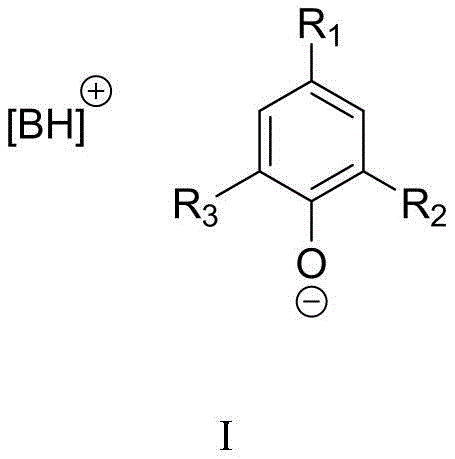
Furthermore, the mechanistic pathway ensures that the reaction remains homogeneous and controllable throughout the process. The interaction between the organic base and the acidic protons of the intermediate species stabilizes the transition states, allowing the reaction to proceed to completion even at lower temperatures (e.g., 60-80°C). This contrasts sharply with metal-catalyzed systems where ligand dissociation or metal aggregation can lead to catalyst death and inconsistent batch-to-batch performance. The robustness of this organic catalytic cycle means that the process is less sensitive to minor fluctuations in reaction parameters, providing a wider operating window for scale-up. For process chemists, understanding this mechanism highlights the potential for optimizing reaction times and catalyst loading to further drive down costs while maintaining the high isolated yields reported in the patent examples, which often exceed 90% for optimized substrate-catalyst pairs.
How to Synthesize 2,4-(1H,3H)-quinazolinedione Efficiently
Implementing this synthesis route requires careful attention to catalyst preparation and reaction conditions to maximize yield and catalyst recovery. The process begins with the in situ formation or pre-isolation of the organic salt catalyst, followed by the introduction of the aminobenzonitrile substrate and CO2 under controlled pressure. The simplicity of the work-up procedure, involving precipitation and filtration, makes this method particularly attractive for large-scale operations where complex chromatographic separations are economically prohibitive. Detailed standardized synthetic steps for preparing the catalyst and executing the cycloaddition reaction are provided below to guide technical teams in replicating these results.
- Prepare the organic base phenolate catalyst by mixing a guanidine or amidine base (e.g., DBU, TBD) with a substituted phenol in a solvent like DCM, followed by washing and drying.
- Load the aminobenzonitrile substrate and the prepared catalyst (1-10 mol%) into a pressure reactor with a suitable solvent such as DMSO or toluene.
- Pressurize the reactor with CO2 (0.1-2.0 MPa), heat to 40-120°C, and stir for 5-20 hours. Isolate the product by precipitation and recover the catalyst from the filtrate.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this CO2 fixation technology offers profound advantages for supply chain resilience and cost management. By shifting away from petrochemical-derived C1 sources and toxic reagents, manufacturers can insulate themselves from volatile raw material markets and regulatory crackdowns on hazardous substances. The ability to utilize CO2, an abundant and inexpensive feedstock, fundamentally alters the cost structure of producing quinazolinedione derivatives. Moreover, the operational simplicity of the process reduces the burden on facility infrastructure, allowing for production in standard reactors without the need for exotic high-pressure alloy vessels required by older technologies. This accessibility lowers the barrier to entry for production and enhances the overall agility of the supply chain.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts removes the necessity for expensive metal scavenging resins and complex purification steps, which traditionally account for a significant portion of manufacturing costs in fine chemical synthesis. Additionally, the recyclability of the organic base phenolate catalyst means that the effective catalyst cost per kilogram of product is drastically reduced over multiple cycles. The mild reaction conditions also translate to lower energy consumption for heating and pressurization, contributing to substantial operational expenditure savings. These factors combined create a leaner, more cost-efficient production model that improves margin potential for high-volume pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The raw materials for this process, specifically aminobenzonitriles and simple phenols, are commodity chemicals with robust global supply chains, reducing the risk of shortages associated with specialized ligands or rare earth metals. The one-step nature of the reaction shortens the overall production cycle time, enabling faster turnaround from order to delivery. This efficiency is critical for meeting the just-in-time delivery demands of modern pharmaceutical supply chains. Furthermore, the stability of the catalyst system ensures consistent product quality, minimizing the risk of batch failures that could disrupt downstream API synthesis schedules.
- Scalability and Environmental Compliance: Scaling this process is straightforward due to the absence of exothermic runaway risks associated with phosgene chemistry and the use of benign organic solvents. The metal-free nature of the reaction simplifies wastewater treatment and waste disposal, as there are no heavy metal contaminants to manage, ensuring full compliance with increasingly stringent environmental protection laws. This "green" credential not only mitigates regulatory risk but also enhances the brand value of the final pharmaceutical product by aligning with the sustainability goals of major multinational drug developers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this CO2 fixation technology. These answers are derived directly from the experimental data and beneficial effects described in the patent literature, providing clarity on catalyst selection, substrate scope, and process robustness. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the advantages of using organic base phenolate catalysts over traditional metal catalysts?
A: Organic base phenolate catalysts eliminate the risk of heavy metal contamination in the final API, simplifying purification and ensuring compliance with strict pharmaceutical residue limits. They also operate under milder conditions and are often recyclable.
Q: Can this synthesis method tolerate various substituents on the aminobenzonitrile ring?
A: Yes, the patented method demonstrates excellent substrate scope, successfully converting aminobenzonitriles with electron-donating groups (like methoxy) and electron-withdrawing groups (like halogens) into the corresponding quinazolinediones with high efficiency.
Q: Is the catalyst reusable in this CO2 fixation process?
A: Absolutely. The protocol allows for the recovery of the organic salt catalyst from the reaction filtrate after product precipitation. The recovered catalyst retains its activity and can be directly reused in subsequent batches, enhancing process economics.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,4-(1H,3H)-quinazolinedione Supplier
The technological advancements presented in patent CN111454222B underscore the immense potential of metal-free CO2 fixation in modern pharmaceutical synthesis. At NINGBO INNO PHARMCHEM, we recognize the strategic value of such innovative pathways and possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle complex organic transformations with stringent purity specifications, supported by rigorous QC labs that ensure every batch meets the highest international standards. We are committed to bridging the gap between laboratory innovation and industrial reality, offering our partners a seamless transition from process development to commercial supply.
We invite procurement leaders and R&D directors to collaborate with us to leverage this cutting-edge synthesis method for your specific project needs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to reach out today to obtain specific COA data for our quinazolinedione intermediates and discuss detailed route feasibility assessments that can optimize your supply chain and reduce your overall cost of goods sold.
