Advanced Silver-Catalyzed Carboxylation for Scalable 4-Hydroxyquinoline-2(1H)-one Production
The pharmaceutical and fine chemical industries are increasingly prioritizing sustainable synthetic routes that leverage abundant C1 feedstocks, and patent CN113416166B represents a significant breakthrough in this domain by detailing a novel method for preparing 4-hydroxyquinoline-2(1H)-one compounds. This technology utilizes 2-ethynylaniline derivatives and carbon dioxide as primary raw materials, reacting them in an ionic liquid medium under the catalysis of silver salts to achieve efficient cyclization. The significance of this invention lies in its ability to transform thermodynamically stable carbon dioxide into valuable heterocyclic scaffolds under relatively mild conditions, addressing the growing demand for green chemistry solutions in API intermediate manufacturing. By replacing harsh traditional reagents with tunable ionic liquids and inexpensive CO2, this process offers a compelling alternative for producing biologically active quinolinone derivatives which are known for their antibacterial, antifungal, and antimalarial properties.

The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-hydroxy-2(1H)-quinolinone derivatives has relied on methodologies that pose significant operational and environmental challenges for large-scale manufacturing. Traditional routes often involve the condensation of substituted anilines with malonate derivatives in high-boiling solvents like diphenyl ether at elevated temperatures, which necessitates energy-intensive heating and complex solvent recovery systems. Other established methods utilize corrosive acidic compounds such as phosphorus oxychloride or require scarce and expensive reagents like sodium telluride, creating severe safety hazards and waste disposal issues for production facilities. Furthermore, multi-step sequences involving the reduction of o-nitrobenzoic acid derivatives often suffer from moderate yields and generate substantial amounts of chemical waste, thereby inflating the overall cost of goods and complicating regulatory compliance regarding impurity profiles.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN113416166B introduces a streamlined atom-economical pathway that directly couples 2-ethynylaniline with carbon dioxide using a silver salt catalyst in an ionic liquid system. This innovative approach eliminates the need for corrosive acids and high-temperature condensation steps, operating instead at moderate temperatures ranging from 30°C to 120°C and under manageable CO2 pressures of 0.1 MPa to 2 MPa. The use of task-specific ionic liquids not only acts as a green solvent but also plays a crucial role in activating the carbon dioxide and stabilizing the reaction intermediates, leading to significantly improved reaction kinetics. This shift from hazardous reagents to a catalytic carboxylation strategy represents a paradigm shift in process chemistry, offering a cleaner, safer, and more efficient route to these high-value heterocyclic cores.
Mechanistic Insights into Silver-Catalyzed Cyclization in Ionic Liquids
The core of this transformation relies on the synergistic interaction between the silver catalyst and the ionic liquid medium to overcome the kinetic inertness of carbon dioxide. The silver salt, typically silver nitrate or silver acetate, coordinates with the alkyne moiety of the 2-ethynylaniline substrate, increasing its electrophilicity and facilitating the nucleophilic attack by the amine group or the insertion of CO2. Simultaneously, the ionic liquid, such as [HDBU][Im] or [HTMG][Im], provides a highly polar environment that enhances the solubility of CO2 and stabilizes the charged transition states formed during the cyclization process. This dual activation mechanism allows the reaction to proceed with high selectivity towards the desired 4-hydroxyquinoline-2(1H)-one structure, minimizing the formation of side products that are common in thermal condensation reactions.
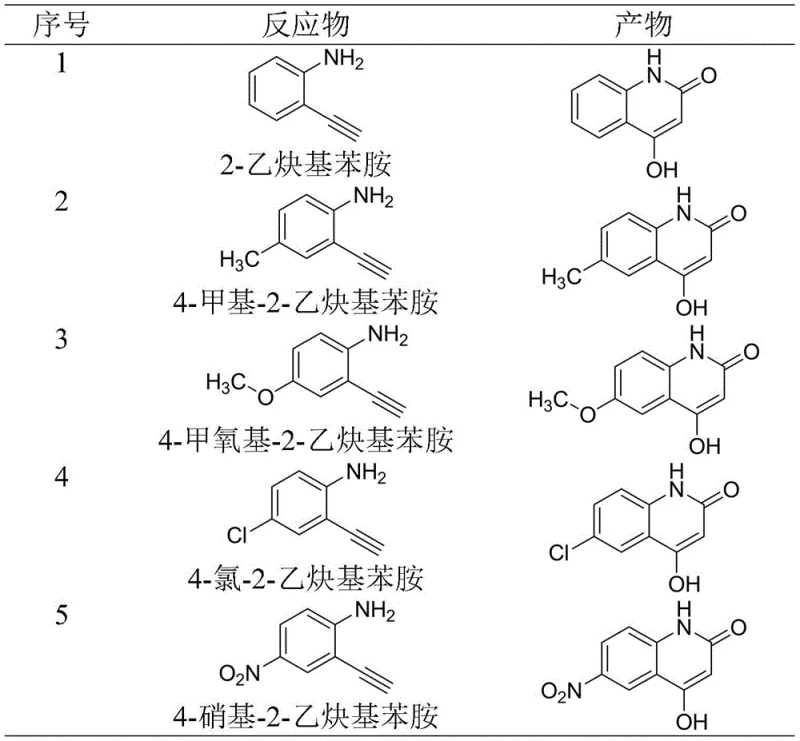
Furthermore, the robustness of this catalytic system is evidenced by its broad substrate tolerance, accommodating a wide range of electronic and steric environments on the benzene ring. As illustrated in the substrate scope data, substituents such as methyl, methoxy, chloro, and even strongly electron-withdrawing nitro groups are well-tolerated, yielding the corresponding quinolinone derivatives in high yields. This versatility is critical for medicinal chemists who require diverse libraries of analogs for structure-activity relationship (SAR) studies. The ability to maintain high efficiency across different substituted anilines suggests that the rate-determining step is likely the CO2 insertion or the subsequent cyclization, both of which are effectively promoted by the silver-ionic liquid couple, ensuring consistent quality regardless of the starting material's electronic nature.
How to Synthesize 4-Hydroxyquinoline-2(1H)-one Efficiently
The practical implementation of this synthesis involves a straightforward procedure where the 2-ethynylaniline substrate is mixed with a catalytic amount of silver salt and a stoichiometric amount of ionic liquid in a reaction vessel. The system is then pressurized with carbon dioxide and heated to the optimal temperature, typically around 60°C, for a duration of 2 to 48 hours depending on the specific substrate reactivity. Following the reaction, the product is isolated through a simple aqueous workup where the addition of water causes the product to precipitate, allowing for easy filtration and washing without the need for extensive chromatographic purification. For detailed standardized operating procedures and specific parameter optimization, please refer to the technical guide below.
- Prepare the reaction mixture by combining 2-ethynylaniline, a silver salt catalyst (such as silver nitrate), and a functionalized ionic liquid in a reaction vessel.
- Introduce carbon dioxide gas into the system to maintain a pressure between 0.1 MPa and 2 MPa while heating the mixture to 30-120°C.
- Upon completion, precipitate the product by adding water, adjusting pH, and filtering the solid to obtain high-purity 4-hydroxyquinoline-2(1H)-one.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented technology offers distinct advantages that directly address the pain points of cost volatility and raw material availability in the fine chemical sector. By utilizing carbon dioxide as a primary building block, the process reduces reliance on complex and expensive C1 synthons that are subject to fluctuating market prices and supply constraints. The substitution of volatile organic solvents with recyclable ionic liquids further mitigates the risks associated with solvent supply chains and reduces the environmental footprint of the manufacturing process, aligning with the sustainability goals of modern pharmaceutical companies.
- Cost Reduction in Manufacturing: The elimination of corrosive reagents and high-energy heating steps translates into significant operational expenditure savings, as the process requires less specialized equipment and lower utility consumption. The use of low catalyst loadings minimizes the cost associated with precious metals, while the simplified isolation procedure reduces the time and resources needed for downstream processing. Additionally, the high atom economy of the carboxylation reaction ensures that raw materials are converted efficiently into the final product, reducing waste disposal costs and maximizing yield per batch.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials like 2-ethynylaniline and industrial-grade carbon dioxide ensures a stable and secure supply chain that is less susceptible to geopolitical disruptions. The robustness of the reaction conditions allows for flexible manufacturing schedules, as the process is not limited by the availability of niche reagents or strict anhydrous conditions. This reliability enables suppliers to maintain consistent inventory levels and meet tight delivery windows, providing a competitive edge in the fast-paced API intermediate market.
- Scalability and Environmental Compliance: The mild reaction conditions and the absence of hazardous byproducts make this process inherently scalable from laboratory to commercial production without significant engineering hurdles. The use of ionic liquids, which have negligible vapor pressure, drastically reduces VOC emissions, simplifying compliance with stringent environmental regulations. This green chemistry profile not only lowers the cost of environmental management but also enhances the brand reputation of the manufacturer as a responsible partner in the global supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this silver-catalyzed carboxylation technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a clear understanding of the method's capabilities and limitations for potential adopters. Understanding these details is essential for evaluating the feasibility of integrating this route into existing manufacturing workflows.
Q: What are the advantages of using ionic liquids over traditional organic solvents in this synthesis?
A: Ionic liquids offer superior CO2 absorption capabilities and thermal stability, allowing the reaction to proceed under milder conditions compared to high-boiling solvents like diphenyl ether, while facilitating easier product separation via water precipitation.
Q: Can this method tolerate various substituents on the aniline ring?
A: Yes, the protocol demonstrates excellent substrate scope, successfully accommodating electron-donating groups like methyl and methoxy, as well as electron-withdrawing groups such as chloro and nitro, yielding corresponding derivatives with high efficiency.
Q: Is the silver catalyst loading economically viable for large-scale production?
A: The process utilizes low catalyst loadings (molar ratios as low as 1:50 relative to substrate), which significantly reduces heavy metal costs and simplifies downstream purification, making it suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Hydroxyquinoline-2(1H)-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this silver-catalyzed carboxylation technology for the production of high-quality pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale innovation to industrial reality is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 4-hydroxyquinoline-2(1H)-one delivered meets the highest standards required for drug substance manufacturing.
We invite forward-thinking R&D and procurement leaders to collaborate with us to leverage this advanced synthetic route for their next-generation therapeutic programs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. We encourage you to reach out today to obtain specific COA data and comprehensive route feasibility assessments that demonstrate how our expertise can optimize your supply chain and accelerate your time to market.
