Advanced Copper-Catalyzed Synthesis of 3-Aroyl Indoles for Scalable Pharmaceutical Manufacturing
The pharmaceutical industry continuously seeks robust synthetic routes for bioactive scaffolds, particularly indole derivatives which serve as critical cores for anticancer and antiemetic agents. Patent CN102911105A introduces a transformative methodology for the synthesis of 3-aroyl indole compounds, addressing long-standing challenges in process chemistry regarding cost and operational complexity. This innovation utilizes a copper-salt catalyzed decarboxylative acylation strategy, employing readily available benzoylformic acids as acylating agents rather than traditional acid chlorides or anhydrides. By shifting away from moisture-sensitive reagents and expensive precious metal catalysts, this technology offers a streamlined pathway that is highly attractive for the commercial scale-up of complex pharmaceutical intermediates. The ability to conduct reactions under aerobic conditions without strict anhydrous protocols represents a significant leap forward in process safety and efficiency, making it an ideal candidate for reliable pharmaceutical intermediate supplier networks aiming to optimize their manufacturing portfolios.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 3-aroyl indole motif has relied heavily on classical Friedel-Crafts acylation strategies, which impose severe constraints on industrial applicability. Traditional protocols typically necessitate the use of highly reactive acyl chlorides or acid anhydrides in conjunction with stoichiometric amounts of Lewis acids such as aluminum chloride or diethyl aluminum chloride. These reagents are notoriously sensitive to moisture, mandating the use of absolutely anhydrous solvents like dichloromethane or toluene and requiring rigorous inert gas protection throughout the reaction course. Furthermore, the generation of stoichiometric aluminum waste creates substantial environmental burdens and complicates downstream purification processes. As illustrated in standard literature precedents, the reaction of indoles with acyl chlorides often requires low temperatures and careful quenching procedures to prevent decomposition, thereby increasing energy consumption and operational hazards.
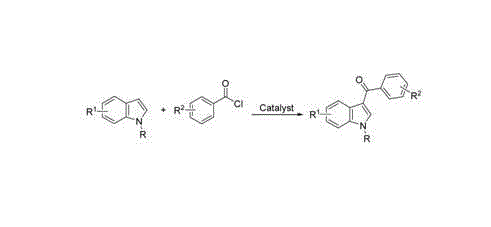
Beyond the environmental and safety concerns, the substrate scope of these conventional methods is often limited by the incompatibility of sensitive functional groups with harsh Lewis acidic conditions. Alternative approaches involving N-acyl benzotriazoles or indole-2,3-dicarboxylic anhydrides have been explored, yet they frequently suffer from multi-step precursor synthesis or the requirement for toxic solvents like 1,2-dichloroethane. The reliance on precious metal catalysts such as palladium or rhodium in newer decarboxylative coupling methods further exacerbates cost issues, as these metals are not only expensive but also require stringent removal processes to meet regulatory limits for residual metals in active pharmaceutical ingredients. Consequently, there is a critical demand for a catalytic system that balances reactivity with economic and operational feasibility.
The Novel Approach
The patented methodology disclosed in CN102911105A fundamentally redefines the synthetic landscape by utilizing benzoylformic acids as direct acylating agents in the presence of inexpensive copper salts and silver oxidants. This decarboxylative acylation proceeds smoothly in polar organic solvents such as dimethyl sulfoxide (DMSO) or N,N-dimethylformamide, eliminating the need for absolute anhydrous conditions or inert atmosphere protection. The reaction mechanism leverages the oxidative decarboxylation of the alpha-keto acid to generate an acyl radical or cationic species in situ, which then attacks the electron-rich C3 position of the indole ring. This approach not only simplifies the reagent profile by avoiding the pre-activation of carboxylic acids into chlorides or anhydrides but also generates carbon dioxide as the primary byproduct, aligning with green chemistry principles.
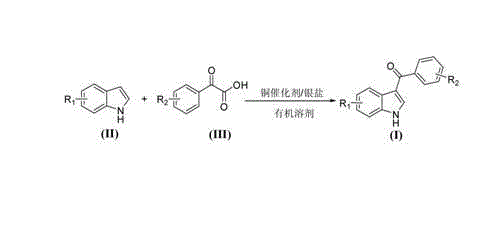
Operational simplicity is a hallmark of this novel approach, as the reaction can be conducted at moderate temperatures ranging from 60°C to 120°C under aerobic conditions. The tolerance to moisture and oxygen drastically reduces the engineering controls required for reactor setup, allowing for simpler equipment configurations and lower capital expenditure. Moreover, the use of copper salts such as copper(II) acetate or cupric oxide, paired with silver carbonate or silver oxide, provides a cost-effective alternative to palladium-based systems while maintaining high catalytic efficiency. This combination ensures that the synthesis of 3-aroyl indoles becomes accessible for large-scale production without compromising on yield or purity, thereby supporting cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Copper-Catalyzed Decarboxylative Acylation
The core of this technological advancement lies in the synergistic interaction between the copper catalyst and the silver oxidant to facilitate the decarboxylation of benzoylformic acid. Mechanistically, the copper species likely coordinates with the carbonyl oxygen of the alpha-keto acid, lowering the activation energy for the loss of carbon dioxide. Simultaneously, the silver salt acts as a terminal oxidant, regenerating the active copper species and driving the catalytic cycle forward. This redox-neutral or oxidative process avoids the formation of stable organometallic intermediates that are prone to side reactions, thus enhancing the chemoselectivity for the C3 position of the indole nucleus. The generation of the acylating species occurs under mild thermal conditions, preventing the degradation of sensitive substituents on either the indole or the aromatic ring of the acylating agent.
From an impurity control perspective, this mechanism offers distinct advantages over traditional electrophilic aromatic substitution. Since the reaction does not rely on strong Brønsted or Lewis acids, there is minimal risk of polymerization or oligomerization of the indole starting material, which are common side reactions in acidic media. Furthermore, the absence of halogenated acylating agents eliminates the formation of halogenated organic impurities that are difficult to remove and potentially genotoxic. The byproduct profile is exceptionally clean, consisting primarily of carbon dioxide gas and insoluble silver/copper residues that can be easily filtered off. This inherent cleanliness simplifies the workup procedure, often requiring only basic extraction and crystallization to achieve high-purity pharmaceutical intermediates suitable for subsequent drug synthesis steps.
How to Synthesize 3-Aroyl Indoles Efficiently
The practical execution of this synthesis involves a straightforward one-pot procedure that combines the substituted indole and benzoylformic acid with the catalytic system in a polar solvent. The reaction mixture is heated to the specified temperature range until the starting materials are consumed, as monitored by HPLC or TLC. Following the reaction, a simple aqueous workup involving extraction with ethyl acetate and washing with saturated sodium bicarbonate solution effectively removes acidic byproducts and catalyst residues. The crude product is then purified via silica gel column chromatography or recrystallization to afford the target 3-aroyl indole in high purity. For detailed standardized operating procedures and specific stoichiometric ratios optimized for different substrates, please refer to the technical guide below.
- Combine substituted indole, benzoylformic acid, copper salt catalyst, and silver salt oxidant in a polar organic solvent such as DMSO.
- Heat the reaction mixture to a temperature between 60°C and 120°C under aerobic conditions without the need for inert gas protection.
- Upon completion, cool the mixture, extract with ethyl acetate and water, wash with saturated sodium bicarbonate, and purify via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this copper-catalyzed protocol presents a compelling value proposition centered on cost stability and operational resilience. By replacing expensive and volatile acyl chlorides with stable benzoylformic acids, the raw material supply chain becomes less susceptible to market fluctuations and logistical disruptions associated with hazardous chemical transport. The elimination of strict anhydrous requirements means that production facilities do not need to invest in specialized drying equipment or maintain complex inert gas manifolds, leading to substantial capital savings and reduced utility costs. Additionally, the use of base metal catalysts instead of precious metals significantly lowers the bill of materials, directly impacting the bottom line through reduced input costs without sacrificing product quality.
- Cost Reduction in Manufacturing: The substitution of precious metal catalysts like palladium or rhodium with abundant copper and silver salts results in a drastic decrease in catalyst costs, which is a major component of the overall production expense. Furthermore, the avoidance of stoichiometric aluminum chloride and the associated waste disposal fees contributes to significant operational expenditure savings. The simplified workup procedure reduces solvent consumption and labor hours required for purification, enhancing the overall economic efficiency of the manufacturing process.
- Enhanced Supply Chain Reliability: Benzoylformic acids and substituted indoles are commercially available commodity chemicals with robust global supply chains, ensuring consistent availability and reducing the risk of production delays. The tolerance of the reaction to moisture and oxygen minimizes the risk of batch failures due to environmental excursions, thereby improving yield consistency and on-time delivery performance. This reliability is crucial for maintaining continuous supply to downstream API manufacturers who depend on just-in-time delivery models.
- Scalability and Environmental Compliance: The mild reaction conditions and lack of toxic byproducts make this process highly scalable from kilogram to multi-ton production scales without significant re-engineering. The generation of carbon dioxide as the primary gaseous byproduct simplifies废气 treatment compared to processes releasing hydrogen chloride or other corrosive gases. This alignment with green chemistry principles facilitates easier regulatory approval and reduces the environmental footprint of the manufacturing site, supporting corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic method in an industrial setting. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this technology into existing production lines.
Q: Does this copper-catalyzed method require strict anhydrous conditions?
A: No, unlike traditional Friedel-Crafts acylations requiring absolute anhydrous environments, this patented process tolerates moisture and can be conducted in aerobic systems, significantly simplifying operational requirements.
Q: What are the cost advantages of using copper and silver salts over palladium catalysts?
A: Copper and silver salts are substantially more economical and stable compared to precious metal catalysts like palladium or rhodium, leading to significant raw material cost reductions and eliminating the need for expensive heavy metal scavenging steps.
Q: What is the typical yield range for this decarboxylative acylation reaction?
A: The method demonstrates high efficiency with isolated yields ranging from moderate to excellent levels, often exceeding 90% for specific substrates like 5-methoxy-3-(4-nitrobenzoyl) indoles, ensuring high material throughput.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Aroyl Indole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient synthetic routes for high-value pharmaceutical intermediates like 3-aroyl indoles. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical techniques to verify identity and assay. Our capability to implement the copper-catalyzed decarboxylative acylation method allows us to offer competitive pricing and reliable supply continuity for clients developing anticancer and CNS therapeutics.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this greener, more efficient protocol. We encourage you to contact us today to obtain specific COA data for our catalog items and to receive comprehensive route feasibility assessments for your custom synthesis projects, ensuring that your supply chain is built on a foundation of technical excellence and commercial viability.
