Revolutionizing Chiral Intermediate Synthesis with Direct C-H Activation Technology
The pharmaceutical industry continuously seeks more efficient pathways to construct chiral centers, particularly within strained ring systems like cyclopropanes which are prevalent in bioactive molecules. Patent CN112312902B introduces a groundbreaking Pd(II)-catalyzed enantioselective C-H arylation method that operates directly on free carboxylic acids. This innovation bypasses the traditional necessity for installing and removing exogenous directing groups, representing a significant leap forward in atom economy and step efficiency. By leveraging novel acetyl-protected aminoethylamine (APAA) ligands, this technology enables the stereoselective functionalization of beta-carbon atoms in cyclopropanecarboxylic acids and related substrates. For R&D directors and procurement specialists, this translates to a robust platform for generating high-purity pharmaceutical intermediates with reduced material throughput and simplified processing workflows.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional strategies for enantioselective C-H activation typically rely heavily on the presence of strong coordinating directing groups attached to the substrate. These groups, such as amides or pyridines, must be chemically installed prior to the reaction and subsequently removed after the desired transformation is complete. This multi-step sequence inherently increases the overall production cost, extends the manufacturing lead time, and generates additional chemical waste that requires disposal. Furthermore, the installation of these directing groups often involves harsh reaction conditions that may not be compatible with sensitive functional groups present in complex drug candidates. The rigidity of these traditional methods limits their applicability in late-stage functionalization scenarios where molecular complexity is already high.
The Novel Approach
The patented methodology overcomes these historical barriers by utilizing the weakly coordinating carboxylic acid group itself as the native directing moiety. Through the strategic design of bidentate APAA ligands, the system achieves sufficient binding affinity and stereocontrol to activate inert C(sp3)-H bonds without auxiliary groups. This direct approach significantly streamlines the synthetic route, effectively collapsing multiple operational steps into a single catalytic event. The use of hexafluoroisopropanol (HFIP) as a solvent further enhances reactivity and selectivity, allowing for mild reaction temperatures around 80°C. This shift from indirect to direct functionalization offers a compelling value proposition for supply chain managers looking to optimize vendor qualification and reduce the carbon footprint of chemical manufacturing processes.
Mechanistic Insights into Pd(II)-Catalyzed Enantioselective C-H Arylation
The core of this technological advancement lies in the synergistic interaction between the palladium catalyst and the chiral APAA ligand. The mechanism initiates with the coordination of the free carboxylic acid to the Pd(II) center, facilitated by the bidentate nature of the ligand which creates a rigid chiral environment. This precise spatial arrangement is critical for distinguishing between enantiotopic C-H bonds, ensuring that the arylation occurs with high fidelity at the beta-position. The catalytic cycle proceeds through a concerted metalation-deprotonation (CMD) pathway, where the base assists in cleaving the C-H bond to form a stable palladacycle intermediate. Subsequent oxidative addition of the aryl iodide and reductive elimination releases the chiral product while regenerating the active catalyst species.
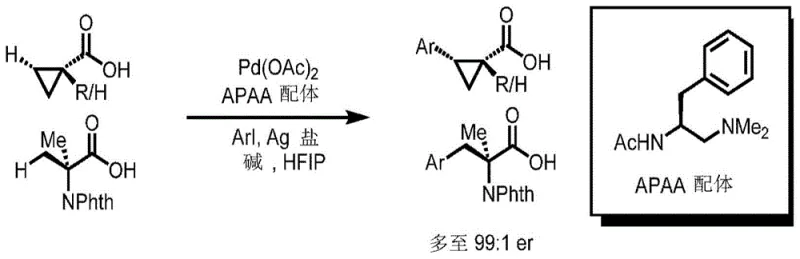
Impurity control is inherently managed through the high specificity of the ligand-substrate interaction. Unlike non-selective radical processes that can lead to a myriad of side products, this organometallic pathway is highly regulated by the steric and electronic properties of the APAA ligand. The screening data indicates that specific substituents on the ligand, such as the benzyl group in L1, provide optimal balance between reactivity and enantioselectivity. Deviations from this optimal structure result in diminished yields or poor stereocontrol, highlighting the precision engineering involved. For quality assurance teams, this means the process is less prone to generating difficult-to-separate diastereomers, thereby simplifying downstream purification and ensuring consistent batch-to-batch purity profiles essential for regulatory compliance.
How to Synthesize Chiral Cyclopropane Derivatives Efficiently
Implementing this synthesis route requires careful attention to reagent quality and reaction parameters to maximize the benefits of the catalytic system. The standard protocol involves charging a reaction vessel with the cyclopropanecarboxylic acid substrate, an aryl iodide coupling partner, and the specific APAA ligand in HFIP solvent. The addition of silver carbonate and sodium carbonate serves dual purposes: acting as oxidants to maintain the palladium cycle and as bases to neutralize the acid byproduct. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction mixture by combining cyclopropanecarboxylic acid substrate, aryl iodide, Pd(OAc)2 catalyst, and APAA ligand in HFIP solvent.
- Add stoichiometric amounts of Ag(I) salt and carbonate base to facilitate the catalytic cycle and neutralize acid byproducts.
- Heat the reaction mixture to 80°C for 16 hours, then purify the resulting chiral product via preparative TLC or chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the elimination of directing group manipulation offers substantial cost savings and supply chain resilience. By removing the need for separate installation and cleavage steps, manufacturers can significantly reduce the consumption of raw materials, solvents, and energy. This streamlined workflow directly correlates to lower operational expenditures and a reduced environmental burden, aligning with modern green chemistry initiatives. For procurement managers, this efficiency translates into more competitive pricing structures for key chiral intermediates without compromising on quality standards. The ability to source complex building blocks through a shorter synthetic route mitigates risks associated with supply chain disruptions and long lead times.
- Cost Reduction in Manufacturing: The direct utilization of free carboxylic acids eliminates the expense associated with purchasing or synthesizing specialized directing group reagents. Furthermore, the reduction in unit operations decreases labor costs and equipment occupancy time, leading to a more economical overall process. The high atom economy of the reaction ensures that a greater proportion of starting materials end up in the final product, minimizing waste disposal fees. These cumulative efficiencies allow for significant cost optimization in pharmaceutical intermediate manufacturing, making high-value chiral compounds more accessible for large-scale production.
- Enhanced Supply Chain Reliability: The reagents required for this transformation, such as aryl iodides and palladium salts, are commercially available from multiple global suppliers, reducing dependency on single-source vendors. The robustness of the reaction conditions allows for flexibility in sourcing raw materials without stringent purity requirements that might otherwise bottleneck production. This diversity in the supply base enhances continuity and reduces the risk of shortages affecting downstream drug development timelines. Additionally, the scalability of the process from milligram to kilogram scale ensures that supply can grow in tandem with clinical demand.
- Scalability and Environmental Compliance: Operating at moderate temperatures and using established solvent systems facilitates easy scale-up in standard glass-lined or stainless steel reactors. The absence of toxic heavy metal scavenging steps, often required when transition metals are used in excess, simplifies the workup and reduces hazardous waste generation. This aligns well with increasingly strict environmental regulations regarding chemical discharge and solvent emissions. The process design inherently supports sustainable manufacturing practices, which is a key criterion for modern supply chain audits and vendor selection processes in the global pharmaceutical industry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this C-H activation technology. These answers are derived directly from the experimental data and scope defined within the patent documentation to ensure accuracy and reliability. Understanding these details helps stakeholders assess the feasibility of integrating this method into their existing development pipelines.
Q: Does this method require pre-installation of directing groups?
A: No, the patented technology utilizes free carboxylic acids directly, eliminating the need for exogenous directing groups and subsequent removal steps.
Q: What level of enantioselectivity can be achieved?
A: The process demonstrates high stereocontrol, achieving enantiomeric ratios up to 99:1 er for various cyclopropane and amino acid substrates.
Q: Is this process suitable for late-stage functionalization?
A: Yes, the mild conditions and broad substrate tolerance allow for the modification of complex drug candidates like Itanapraced without protecting group manipulation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Cyclopropane Supplier
NINGBO INNO PHARMCHEM stands at the forefront of adopting advanced synthetic methodologies to deliver superior chemical solutions. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative lab-scale discoveries like this Pd-catalyzed arylation can be successfully translated to industrial manufacturing. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of chiral intermediate meets the exacting standards required by global regulatory bodies. Our commitment to technical excellence ensures that complex molecular architectures are delivered with consistency and reliability.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific molecule. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how this direct C-H activation strategy can optimize your supply chain and accelerate your time to market.
