Advanced Organocatalytic Synthesis of Chiral Indole Derivatives for Commercial Pharmaceutical Manufacturing
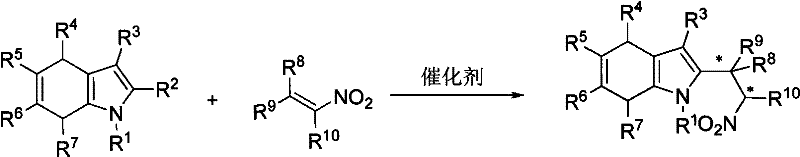
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and environmentally benign synthetic routes for complex heterocyclic intermediates. A significant breakthrough in this domain is documented in patent CN101456838B, which discloses a highly efficient method for synthesizing 2-(2-nitroethyl)-4,7-dihydroindole compounds. This technology leverages asymmetric organocatalysis, specifically utilizing chiral phosphoric acids to drive the Friedel-Crafts alkylation of 4,7-dihydroindoles with nitroalkenes. Unlike traditional methods that often rely on expensive and toxic transition metals, this approach offers a metal-free alternative that delivers exceptional enantioselectivity and yield. For R&D directors and procurement managers, this represents a pivotal shift towards greener chemistry that does not compromise on performance, enabling the production of high-purity pharmaceutical intermediates with reduced regulatory burdens regarding heavy metal residues.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of chiral indole scaffolds has frequently depended on transition metal catalysis or stoichiometric chiral auxiliaries. These conventional pathways often suffer from significant drawbacks, including the requirement for harsh reaction conditions, the generation of toxic waste streams, and the persistent challenge of removing trace metal contaminants from the final API. Furthermore, many traditional catalytic systems exhibit limited substrate scope, failing to accommodate diverse electronic and steric environments without a drastic loss in stereoselectivity. The reliance on precious metals also introduces volatility into the supply chain, as prices for materials like palladium or rhodium can fluctuate wildly, impacting the cost stability of long-term manufacturing contracts. Additionally, the purification steps required to meet stringent pharmacopeial limits for heavy metals add considerable time and expense to the overall production cycle.
The Novel Approach
The methodology outlined in the patent data introduces a transformative solution by employing chiral phosphoric acid as a Brønsted acid catalyst. This organocatalytic system operates under remarkably mild conditions, typically between -50°C and 30°C, and achieves reaction completion within minutes to hours. The process demonstrates broad substrate tolerance, successfully accommodating various substituted 4,7-dihydroindoles and nitroalkenes with yields generally ranging from 85% to 98% and enantiomeric excess (ee) values between 87% and 97%. By eliminating the need for metal salts, this novel approach simplifies the downstream processing workflow, removing the necessity for specialized scavenging resins or complex extraction protocols. This efficiency translates directly into cost reduction in pharmaceutical intermediate manufacturing, as it streamlines the path from raw materials to high-value chiral building blocks while ensuring a cleaner impurity profile.
Mechanistic Insights into Chiral Phosphoric Acid Catalyzed Friedel-Crafts Alkylation
The core of this technological advancement lies in the unique activation mode of the chiral phosphoric acid catalyst. The catalyst functions through a dual hydrogen-bonding mechanism, simultaneously activating the electrophilic nitroalkene and organizing the nucleophilic 4,7-dihydroindole within a well-defined chiral pocket. This precise spatial arrangement dictates the facial selectivity of the attack, ensuring the formation of the desired enantiomer with high fidelity. The rigid binaphthyl backbone of the catalyst provides the necessary steric bulk to differentiate between the prochiral faces of the reacting species. This mechanistic elegance allows for the synthesis of complex molecular architectures with multiple stereocenters in a single operation, significantly reducing the step count compared to linear synthetic strategies.
Furthermore, the organocatalytic nature of this reaction inherently minimizes the formation of metal-associated impurities, which are often difficult to characterize and remove. The absence of metal coordination complexes means that side reactions typical of redox-active metals, such as unwanted oxidations or polymerizations, are largely suppressed. This results in a cleaner crude reaction mixture, facilitating easier purification via standard techniques like recrystallization or column chromatography. For quality control teams, this implies a more robust and predictable impurity profile, reducing the risk of batch failures during final release testing. The ability to tune the catalyst structure by modifying the substituents on the binaphthyl ring further allows for optimization of reactivity and selectivity for specific substrate classes, providing a versatile platform for process development.
How to Synthesize 2-(2-Nitroethyl)-4,7-dihydroindoles Efficiently
The practical implementation of this synthesis involves a straightforward protocol suitable for both laboratory and pilot-scale operations. The reaction is typically conducted in common organic solvents such as dichloromethane, benzene, or toluene, often in the presence of molecular sieves to maintain anhydrous conditions. The molar ratio of nitroalkene to 4,7-dihydroindole is optimized to ensure complete conversion while minimizing waste, with catalyst loading kept low to maximize economic efficiency. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction system by dissolving the chiral phosphoric acid catalyst and 4,7-dihydroindole substrate in an organic solvent such as dichloromethane or benzene under inert atmosphere.
- Slowly add the nitroalkene reactant to the mixture at controlled temperatures ranging from -50°C to 30°C while maintaining vigorous stirring to ensure proper mixing and heat dissipation.
- Monitor the reaction progress until completion, typically within minutes to hours, followed by standard workup procedures including washing, drying, and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this organocatalytic route offers substantial strategic benefits for supply chain management and cost engineering. The elimination of precious metal catalysts removes a major cost driver and supply risk factor, stabilizing the bill of materials for large-scale production campaigns. Moreover, the mild reaction conditions reduce energy consumption and equipment stress, contributing to lower operational expenditures and enhanced safety profiles in manufacturing facilities.
- Cost Reduction in Manufacturing: The removal of expensive transition metals and the associated purification steps leads to significant cost savings. Without the need for metal scavengers or rigorous heavy metal testing, the overall cost of goods sold is drastically simplified. The high yields reported (85%-98%) ensure maximum material throughput, minimizing waste disposal costs and maximizing the return on raw material investment. This efficiency is critical for maintaining competitive pricing in the global market for fine chemicals.
- Enhanced Supply Chain Reliability: The substrates required for this reaction, such as substituted indoles and nitroalkenes, are commercially available and chemically stable, ensuring a secure supply base. The robustness of the catalyst system means that production is less susceptible to variations in raw material quality or minor fluctuations in process parameters. This reliability reduces lead time for high-purity pharmaceutical intermediates, allowing manufacturers to respond more agilely to market demands and inventory requirements without compromising on delivery schedules.
- Scalability and Environmental Compliance: The metal-free nature of the process aligns perfectly with increasingly stringent environmental regulations and green chemistry initiatives. Scaling up this reaction does not introduce the complexities of handling toxic metal waste streams, simplifying environmental permitting and waste treatment protocols. The ability to operate at near-ambient temperatures further enhances scalability, as it reduces the need for extreme cooling or heating infrastructure, making the transition from gram-scale discovery to tonnage-scale commercial production seamless and economically viable.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral synthesis technology. These insights are derived directly from the experimental data and scope defined in the patent literature, providing a factual basis for decision-making.
Q: What are the advantages of using chiral phosphoric acid over traditional metal catalysts?
A: Chiral phosphoric acid catalysts eliminate the risk of heavy metal contamination, which is critical for pharmaceutical compliance. They also operate under milder conditions and often provide superior enantioselectivity compared to traditional transition metal complexes.
Q: Can this synthesis method be scaled for industrial production?
A: Yes, the process utilizes readily available substrates and mild reaction conditions (-50°C to 30°C), making it highly suitable for scale-up. The absence of sensitive metal catalysts simplifies the purification process and reduces operational costs.
Q: What downstream applications do these nitroethyl indole compounds support?
A: These intermediates are versatile precursors that can be oxidized to form optically active 2-(2-nitroethyl)indoles or reduced and cyclized to synthesize biologically active tetrahydro-gamma-carboline derivatives used in drug discovery.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(2-Nitroethyl)-4,7-dihydroindole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing advanced synthetic technologies to drive drug development forward. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in patent literature can be translated into reliable industrial reality. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of chiral intermediates meets the highest standards required by global regulatory agencies. Our commitment to quality ensures that your supply chain remains uninterrupted and compliant.
We invite you to collaborate with us to optimize your sourcing strategy for these valuable building blocks. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how this specific organocatalytic route can benefit your project economics. We encourage potential partners to contact our technical procurement team to obtain specific COA data and route feasibility assessments tailored to your unique molecular targets. Let us help you navigate the complexities of chiral synthesis with confidence and precision.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
