Scalable Manufacturing of 1-Aminomethyl-1-Cyclopropanol via Novel Titanium-Mediated Route
Scalable Manufacturing of 1-Aminomethyl-1-Cyclopropanol via Novel Titanium-Mediated Route
The pharmaceutical industry constantly seeks robust synthetic pathways for complex intermediates that balance cost efficiency with structural integrity. Patent CN111454163A introduces a groundbreaking methodology for synthesizing 1-aminomethyl-1-cyclopropanol, a critical building block in modern drug discovery. This innovative approach fundamentally shifts the paradigm from traditional noble metal catalysis to a more economical titanium-mediated cycle. By leveraging accessible starting materials like 2,5-hexanedione and glycine ethyl ester hydrochloride, the process establishes a reliable foundation for high-volume production. The strategic elimination of palladium hydroxide not only addresses supply chain vulnerabilities associated with precious metals but also significantly lowers the barrier to entry for manufacturing this valuable scaffold. For R&D teams evaluating new routes, this patent offers a compelling alternative that aligns with green chemistry principles while delivering the requisite purity for downstream applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1-aminomethyl-1-cyclopropanol has been hindered by reliance on palladium hydroxide as a primary catalyst, creating substantial economic and operational bottlenecks. The traditional protocols often require stoichiometric or near-stoichiometric amounts of this noble metal, driving raw material costs to prohibitive levels ranging from 20,000 to 30,000 RMB per kilogram in some market contexts. Beyond the direct financial burden, the use of palladium introduces complex purification challenges, necessitating rigorous metal scavenging steps to meet stringent pharmaceutical impurity specifications. Furthermore, the sensitivity of palladium catalysts to poisoning and the requirement for specialized handling equipment complicate the scale-up process for contract development and manufacturing organizations. These factors collectively restrict the widespread application of the intermediate, limiting its potential in cost-sensitive therapeutic areas where margin compression is a constant concern for procurement managers.
The Novel Approach
In stark contrast, the methodology disclosed in CN111454163A utilizes a sophisticated sequence of condensation, cyclopropanation, and deprotection reactions that bypass the need for precious metals entirely. The process initiates with a Paal-Knorr pyrrole synthesis under mild aqueous conditions, followed by a titanium-mediated transformation that constructs the cyclopropane ring with high fidelity. This shift to base metal catalysis, specifically utilizing tetraisopropyl titanate and Grignard reagents, results in a drastic reduction in synthesis costs, potentially lowering expenses to a fraction of the traditional route. The operational simplicity is further enhanced by the use of common solvents like tetrahydrofuran and ethanol, which are easily recovered and recycled in an industrial setting. For supply chain leaders, this represents a transition to a more resilient manufacturing model that is less susceptible to fluctuations in the global precious metals market.
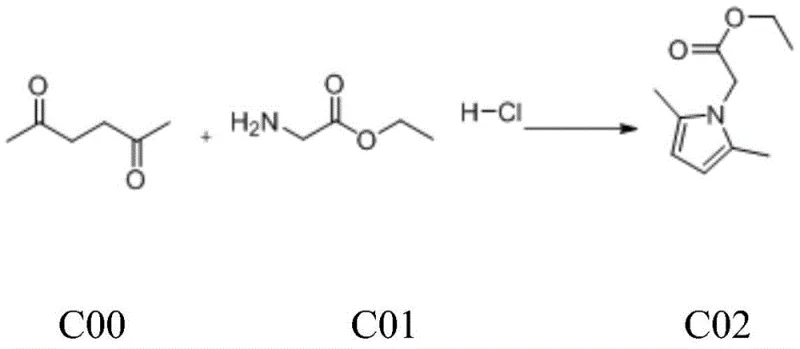
Mechanistic Insights into Titanium-Mediated Cyclopropanation and Deprotection
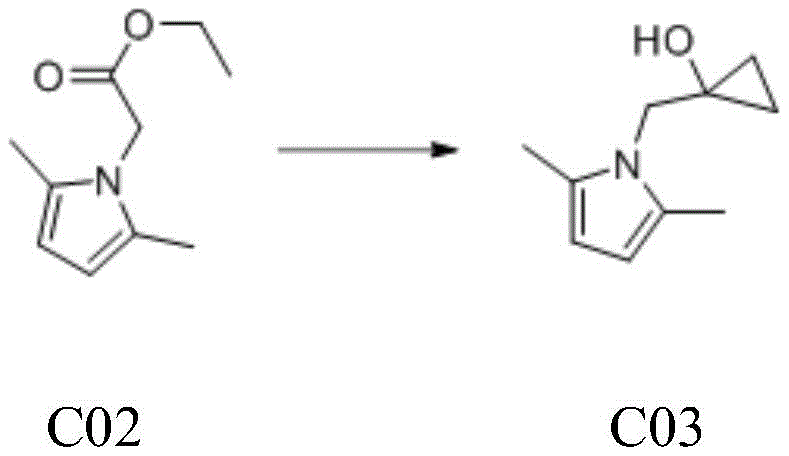
The core innovation of this synthetic route lies in the second step, which employs a variation of the Kulinkovich reaction to convert the ester functionality into a cyclopropanol moiety. In this mechanism, tetraisopropyl titanate reacts with ethyl magnesium bromide to generate a reactive titanacyclopropane species in situ, which subsequently attacks the carbonyl group of the pyrrole-attached ester. This intricate organometallic transformation proceeds through a cyclic transition state that ensures the formation of the strained three-membered ring with excellent regioselectivity. The reaction conditions are meticulously controlled, with the Grignard reagent added dropwise over a period exceeding four hours to manage the violent exotherm and maintain temperatures below 20°C. Such precise thermal management is critical for preventing side reactions and ensuring the structural integrity of the sensitive pyrrole ring during the cyclopropanation event.
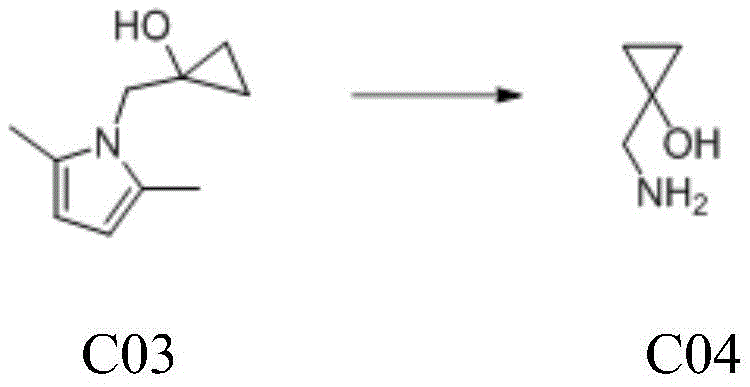
Following the construction of the cyclopropane core, the final stage involves the strategic removal of the pyrrole protecting group to reveal the primary amine. This deprotection is achieved using hydroxylamine hydrochloride under reflux conditions in absolute ethanol, a method chosen for its chemoselectivity and mildness. The mechanism likely involves nucleophilic attack by hydroxylamine on the pyrrole ring, leading to ring opening and the release of the free aminomethyl group without disturbing the fragile cyclopropanol structure. The process includes a careful pH adjustment to neutrality using sodium hydroxide, which facilitates the separation of organic impurities and ensures the final product is isolated as a stable free base or salt. This step underscores the importance of orthogonal protecting group strategies in multi-step synthesis, allowing for the unmasking of functional groups late in the sequence to maximize overall yield.
How to Synthesize 1-Aminomethyl-1-Cyclopropanol Efficiently
Executing this synthesis requires strict adherence to the specified reaction parameters to ensure safety and reproducibility, particularly during the exothermic Grignard addition. The process begins with the formation of the pyrrole intermediate in an aqueous potassium carbonate medium, followed by extraction and concentration to yield the crude ester. Operators must then dissolve this intermediate in dry tetrahydrofuran and cool the solution to between 8°C and 10°C before introducing the titanium catalyst. The subsequent addition of ethyl magnesium bromide demands vigilant temperature monitoring and slow addition rates to prevent thermal runaway, followed by a quench with ammonium chloride to decompose titanium complexes. Detailed standardized synthetic steps see the guide below.
- Condense 2,5-hexanedione with glycine ethyl ester hydrochloride in aqueous potassium carbonate to form the pyrrole intermediate C02.
- React the pyrrole ester C02 with ethyl magnesium bromide and tetraisopropyl titanate in THF to generate the cyclopropanol derivative C03.
- Cleave the pyrrole protecting group using hydroxylamine hydrochloride in ethanol under reflux to yield the final product 1-aminomethyl-1-cyclopropanol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patent-covered methodology offers transformative benefits that extend far beyond simple unit price reductions. By removing the dependency on palladium hydroxide, manufacturers can insulate their production costs from the volatility of the precious metals market, ensuring more predictable budgeting and long-term pricing stability. The use of commodity chemicals such as 2,5-hexanedione and glycine esters guarantees a robust supply of starting materials, mitigating the risk of shortages that often plague specialized catalyst supply chains. Furthermore, the simplified purification workflow, which relies on standard extractions and distillations rather than complex chromatography or metal scavenging, significantly reduces processing time and solvent consumption. These operational efficiencies translate directly into improved margins and a more competitive position in the global marketplace for pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The elimination of noble metal catalysts removes one of the most significant cost drivers in the traditional synthesis of this intermediate. Without the need for expensive palladium salts and the associated recovery or disposal costs, the overall cost of goods sold is substantially decreased. Additionally, the high atom economy of the cyclopropanation step minimizes waste generation, further contributing to lower operational expenditures and a more sustainable manufacturing footprint.
- Enhanced Supply Chain Reliability: Sourcing raw materials for this process is straightforward, as reagents like tetraisopropyl titanate and ethyl magnesium bromide are widely available from multiple global suppliers. This diversification of the supply base reduces the risk of single-source dependency and ensures continuity of supply even during market disruptions. The robustness of the reaction conditions also allows for flexible manufacturing scheduling, enabling producers to respond quickly to fluctuating demand from downstream drug developers.
- Scalability and Environmental Compliance: The process is inherently scalable, having been designed with industrial production in mind, utilizing standard reactor configurations and common solvent systems. The absence of heavy metal residues simplifies environmental compliance and waste treatment, as effluent streams do not require specialized processing to remove toxic palladium traces. This alignment with green chemistry principles not only reduces regulatory burdens but also enhances the corporate sustainability profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route, derived directly from the patent specifications and practical manufacturing considerations. Understanding these details is crucial for technical teams assessing the feasibility of integrating this intermediate into their own supply chains. The answers provided reflect the specific advantages of the titanium-mediated approach over legacy methods, focusing on purity, cost, and operational safety.
Q: How does this new synthesis method reduce costs compared to traditional routes?
A: The novel method completely eliminates the need for expensive palladium hydroxide catalysts, which traditionally drive up material costs significantly. By utilizing common reagents like titanium tetraisopropoxide and Grignard reagents, the raw material expense is drastically lowered while maintaining high purity standards.
Q: What are the critical control points for ensuring high purity in the cyclopropanation step?
A: Temperature control is paramount during the addition of ethyl magnesium bromide, which must be kept below 20°C to manage exothermic heat release. Additionally, the use of diatomaceous earth during filtration and precise pH adjustment during workup ensures the removal of titanium salts and side products.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process avoids hazardous high-pressure hydrogenation and uses standard solvent systems like THF and ethanol, making it highly adaptable for multi-kilogram to ton-scale production. The simplified workup procedures, such as direct distillation and extraction, further enhance its viability for industrial supply chains.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Aminomethyl-1-Cyclopropanol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in accelerating drug development timelines and ensuring commercial success. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to pilot plant is seamless and efficient. We adhere to stringent purity specifications and utilize rigorous QC labs to verify that every batch of 1-aminomethyl-1-cyclopropanol meets the exacting standards required by top-tier pharmaceutical companies. Our commitment to quality assurance means that clients can rely on us for consistent supply without compromising on the chemical integrity of their final active pharmaceutical ingredients.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this palladium-free methodology. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your volume needs, ensuring a partnership built on transparency, technical excellence, and mutual growth.
