Advanced Synthesis of Rhodium Caprylate Dimer for Industrial Homogeneous Catalysis Applications
The pharmaceutical and fine chemical industries rely heavily on the precision and efficiency of homogeneous catalysts, particularly those based on transition metals like rhodium. A significant breakthrough in this domain is documented in patent CN111718253B, which details a novel preparation method for rhodium caprylate dimer, a critical catalyst used in C-H insertion reactions and cyclopropanation processes essential for synthesizing beta-lactams and other complex pharmaceutical intermediates. This patent addresses long-standing inefficiencies in catalyst production by introducing a streamlined two-step synthesis that utilizes readily available rhodium trichloride (RhCl3) and alkali metal caprylates. By shifting away from expensive precursors and energy-intensive separation techniques, this technology offers a robust pathway for producing high-purity catalytic materials that meet the stringent demands of modern industrial manufacturing. For R&D directors and procurement specialists, understanding the nuances of this synthesis route is vital for optimizing supply chains and reducing the overall cost of goods sold in catalyst-dependent production lines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of rhodium (II) octanoate has been plagued by significant operational hurdles and economic inefficiencies that hinder large-scale adoption. Traditional methods, such as the ligand exchange reaction between rhodium acetate and caprylic acid, suffer from the high boiling point of caprylic acid (approximately 237°C), which necessitates extreme thermal conditions that increase energy consumption and pose risks of product pyrolysis. Furthermore, the separation of the target rhodium dimer from the excess high-boiling acid is notoriously difficult, often resulting in mediocre yields hovering around 72 percent. Alternative approaches like Soxhlet extraction, while capable of achieving higher yields near 97.5 percent, are operationally complex and rely on rhodium acetate dimer, a precursor that is both expensive and difficult to source reliably in bulk quantities. These legacy processes create bottlenecks in the supply chain, leading to inconsistent batch quality and inflated production costs that ultimately impact the profitability of downstream pharmaceutical applications.
The Novel Approach
In stark contrast to these cumbersome legacy techniques, the method disclosed in CN111718253B introduces a simplified yet highly effective strategy that leverages inexpensive rhodium trichloride as the starting metal source. This innovative route bypasses the need for high-boiling carboxylic acids by employing an alkali metal caprylate, such as sodium caprylate, to facilitate a smooth ligand replacement under mild conditions. The process eliminates the complex distillation steps required in older methods, replacing them with a straightforward precipitation and filtration workflow that is inherently easier to scale. By utilizing common alcohols as reducing agents instead of specialized reagents, the novel approach drastically simplifies the raw material procurement process, ensuring a more stable and cost-effective supply chain. This shift not only enhances the economic viability of producing rhodium caprylate dimer but also aligns with green chemistry principles by reducing energy usage and waste generation associated with high-temperature separations.
Mechanistic Insights into RhCl3-Mediated Reduction and Ligand Exchange
The core of this technological advancement lies in a carefully orchestrated two-step mechanism that ensures the selective formation of the Rh(II) dimer without over-reduction to metallic rhodium. Initially, rhodium trichloride undergoes a ligand exchange reaction with alkali metal caprylate to form an intermediate rhodium (III) caprylate species in solution. This intermediate is then subjected to a controlled reduction process using weak reducing agents such as ethanol, isopropanol, or n-butanol under reflux conditions at temperatures between 70°C and 80°C. The choice of a weak reducing agent is mechanistically critical; it provides just enough reducing power to convert the trivalent rhodium center to the divalent state required for the lantern-like dimer structure, while preventing the thermodynamic drive toward zero-valent metal formation. Comparative data within the patent highlights that stronger reducing agents like hydrazine hydrate fail catastrophically in this context, instantly producing black metallic rhodium precipitates that render the catalyst useless for homogeneous applications.
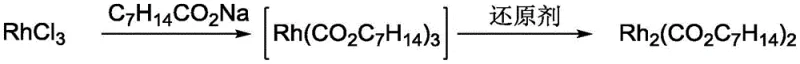
Following the reduction phase, the reaction mixture contains the target rhodium caprylate dimer alongside soluble by-products such as sodium chloride and residual alkali metal salts. The purification mechanism exploits the distinct solubility profile of the rhodium dimer, which exhibits extremely low solubility in water. By pouring the hot reaction solution into deionized water, the target product precipitates immediately as a green solid, allowing for efficient isolation via filtration. Subsequent washing steps are designed to target specific impurities: a wash with 10wt% sodium bicarbonate solution effectively removes water-soluble inorganic salts and unreacted caprylate, while a secondary wash with diethyl ether dissolves and removes organic by-products like acetaldehyde generated during the alcohol oxidation. This multi-stage purification protocol ensures that the final product achieves a purity level exceeding 98 percent, making it suitable for sensitive pharmaceutical syntheses where trace metal impurities can compromise reaction selectivity.
How to Synthesize Rhodium Caprylate Dimer Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires strict adherence to the stoichiometric ratios and temperature controls defined in the patent to maximize yield and purity. The process begins with the uniform mixing of RhCl3 and sodium caprylate in a molar ratio ranging from 1:2.9 to 1:4.5 to ensure complete ligand saturation before the reduction step commences. Operators must maintain the reflux temperature precisely between 70°C and 80°C for a duration of 4 to 12 hours, depending on the specific alcohol reducing agent employed, to guarantee complete conversion of the Rh(III) intermediate. The detailed standardized synthetic steps, including specific volumes for washing and drying parameters, are outlined below to assist technical teams in replicating this high-efficiency protocol.
- Mix RhCl3 solution with alkali metal caprylate (e.g., sodium caprylate) to form a first solution containing the Rh(III) intermediate.
- Add a weak reducing agent such as ethanol or isopropanol and heat to 70-80°C for reflux reaction to reduce Rh(III) to Rh(II).
- Precipitate the product by adding the hot reaction mixture to deionized water, followed by washing with NaHCO3 and ether to remove impurities.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis method represents a strategic opportunity to optimize operational expenditures and mitigate supply risks associated with precious metal catalysts. By transitioning from expensive rhodium acetate precursors to commodity-grade rhodium trichloride and sodium caprylate, manufacturers can achieve substantial cost savings in raw material acquisition without compromising on the quality of the final catalyst. The elimination of high-energy distillation steps further contributes to cost reduction in homogeneous catalyst manufacturing by lowering utility consumption and reducing the thermal load on production equipment. Additionally, the use of common alcohols as reducing agents simplifies inventory management, as these solvents are widely available and do not require specialized handling or storage protocols compared to more hazardous or exotic reagents.
- Cost Reduction in Manufacturing: The substitution of high-cost starting materials with economically viable alternatives like RhCl3 and sodium salts directly lowers the bill of materials, while the simplified workup procedure reduces labor and processing time. The avoidance of high-boiling point solvents means that energy costs for heating and separation are significantly diminished, leading to a more lean and efficient production model. Furthermore, the high yield of over 80 percent ensures that precious rhodium metal is utilized with maximum atom economy, minimizing waste and maximizing the return on investment for every kilogram of metal processed.
- Enhanced Supply Chain Reliability: Sourcing rhodium acetate dimer can often be a bottleneck due to its limited availability and high market volatility, whereas rhodium trichloride and alkali metal caprylates are standard industrial chemicals with robust supply networks. This shift in raw material dependency enhances supply chain resilience, ensuring that production schedules are not disrupted by shortages of niche precursors. The simplicity of the process also means that it can be easily replicated across multiple manufacturing sites, providing redundancy and flexibility in the global supply network for critical catalytic materials.
- Scalability and Environmental Compliance: The aqueous precipitation method described in the patent is inherently scalable, moving seamlessly from gram-scale laboratory synthesis to multi-ton industrial production without the need for complex engineering modifications. The washing steps utilize benign reagents like sodium bicarbonate and allow for the easy recovery of solvents, aligning with increasingly strict environmental regulations regarding waste disposal and solvent emissions. This environmental compatibility reduces the regulatory burden on manufacturers and supports sustainability goals by minimizing the generation of hazardous waste streams associated with traditional high-temperature organic synthesis.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this rhodium caprylate dimer synthesis method, drawing directly from the experimental data and comparative examples provided in the patent documentation. These insights are intended to clarify the operational parameters and troubleshooting strategies for technical teams evaluating this process for commercial adoption. Understanding these specifics is crucial for ensuring consistent product quality and process safety during scale-up activities.
Q: Why are weak reducing agents preferred over strong ones like hydrazine hydrate?
A: Strong reducing agents like hydrazine hydrate cause over-reduction, converting trivalent rhodium directly into zero-valent metal rhodium (black precipitate), whereas weak alcohols selectively reduce Rh(III) to the desired Rh(II) dimer state.
Q: What is the purity and yield advantage of this new method compared to ligand exchange?
A: This method achieves a total yield of over 80% and purity exceeding 98%, significantly outperforming the traditional ligand exchange method which often struggles with yields around 72% due to separation difficulties.
Q: How are by-products like NaCl and acetaldehyde removed?
A: Water-soluble by-products like NaCl and excess sodium caprylate are removed via washing with 10wt% NaHCO3 solution, while organic impurities like acetaldehyde are eliminated through a secondary washing step using diethyl ether.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Rhodium Caprylate Dimer Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance catalysts play in the success of complex pharmaceutical and fine chemical syntheses. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that employ advanced analytical techniques to verify every batch against the highest industry standards. Our capability to manufacture rhodium caprylate dimer using the optimized methods described in CN111718253B allows us to offer a reliable rhodium catalyst supplier partnership that balances technical excellence with commercial viability.
We invite potential partners to engage with our technical procurement team to discuss how our advanced manufacturing capabilities can support your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized synthesis routes can reduce your overall production costs while maintaining superior catalyst performance. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your application needs, ensuring that your supply chain is built on a foundation of quality, reliability, and innovation.
