Advanced Synthesis of Axially Chiral Bipyridine Ligands for Asymmetric Catalysis
The landscape of asymmetric catalysis is continually evolving, driven by the demand for highly efficient and selective chiral ligands that can orchestrate complex molecular transformations with precision. A significant breakthrough in this domain is documented in patent CN113336764B, which details a novel and practical synthetic route for a class of axially chiral bipyridine ligands. Unlike traditional approaches that often struggle with low stereocontrol or require tedious resolution processes, this invention leverages a strategic combination of Mitsunobu etherification and metal-promoted Ullmann coupling. By utilizing readily available 3-hydroxy-2-halopyridines and chiral diols as the foundational building blocks, the methodology effectively transfers central chirality from the diol backbone to the axial chirality of the resulting bipyridine system. This approach not only streamlines the synthetic pathway but also ensures high optical purity, addressing a critical bottleneck for reliable pharmaceutical intermediate suppliers seeking to provide high-quality chiral auxiliaries for drug discovery and process chemistry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of axially chiral bipyridine ligands has been fraught with significant challenges that hinder their widespread adoption in industrial settings. Conventional strategies often rely on the resolution of racemic mixtures, a process that is inherently inefficient as it theoretically discards up to 50% of the synthesized material, leading to substantial waste and increased raw material costs. Furthermore, existing methods for constructing the biaryl axis frequently involve harsh reaction conditions or expensive transition metal catalysts that are difficult to remove from the final product, posing risks for downstream applications in sensitive high-purity OLED material or pharmaceutical synthesis. The lack of generalizable methods to induce axial chirality directly from chiral pools has meant that many potential ligand structures remain inaccessible or economically unviable for large-scale cost reduction in electronic chemical manufacturing.
The Novel Approach
The methodology outlined in the patent presents a transformative solution by decoupling the formation of the chiral skeleton from the construction of the biaryl axis. As illustrated in the general reaction scheme below, the process begins with a highly stereospecific Mitsunobu reaction, attaching the pyridine units to a chiral diol scaffold with inversion of configuration, followed by a robust Ullmann-type coupling to close the ring system. This two-step sequence avoids the thermodynamic equilibration issues common in direct oxidative couplings and allows for the precise tuning of steric bulk via the selection of the diol spacer. By enabling the use of inexpensive copper powder or in-situ generated nickel species, this route offers a pragmatic alternative that significantly simplifies the supply chain for complex chiral ligands.
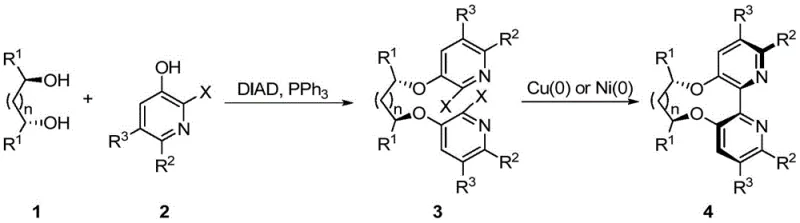
Mechanistic Insights into Copper and Nickel Promoted Ullmann Coupling
The core of this synthetic innovation lies in the meticulous control of the Ullmann coupling step, which is responsible for forging the carbon-carbon bond between the two pyridine rings. In the copper-promoted variant, elemental copper powder acts as both the reductant and the source of the active catalytic species, facilitating the oxidative addition and reductive elimination cycle necessary for biaryl formation at elevated temperatures ranging from 140°C to 180°C. Alternatively, the nickel-catalyzed pathway utilizes a divalent nickel salt reduced in situ by zinc powder in the presence of triphenylphosphine, generating a highly reactive Ni(0) species that operates under milder conditions (0°C to 80°C). This dual-option flexibility allows process chemists to optimize the reaction based on substrate sensitivity, ensuring that the delicate chiral information encoded in the diol backbone is preserved throughout the rigorous coupling process without epimerization.
Impurity control is paramount in the production of ligands intended for asymmetric catalysis, where trace contaminants can poison downstream catalytic cycles. The patent describes a rigorous workup procedure involving acid-base extraction to remove residual metal species and phosphine oxides. Specifically, treating the crude reaction mixture with strong acids like hydrochloric acid protonates the basic pyridine nitrogens, allowing for the separation of organic impurities, followed by basification to recover the free base ligand. This purification strategy, combined with the inherent selectivity of the Mitsunobu step which minimizes side reactions, results in a product profile with exceptional chemical and optical purity, meeting the stringent specifications required for commercial scale-up of complex polymer additives and fine chemical intermediates.
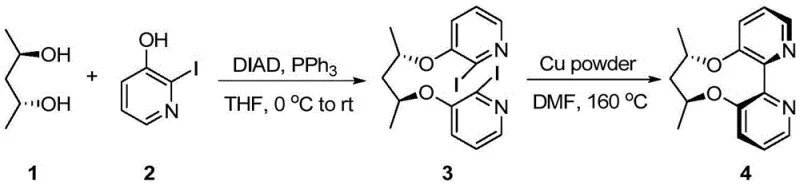
How to Synthesize Axially Chiral Bipyridine Ligands Efficiently
The synthesis protocol described in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing operational simplicity and reproducibility. The process initiates with the activation of the chiral diol and the halopyridine using standard Mitsunobu reagents (DIAD and PPh3) in tetrahydrofuran, followed by the critical cyclization step using either copper or nickel promoters in polar aprotic solvents like DMF. The detailed standardized synthesis steps, including specific molar ratios, temperature gradients, and workup procedures, are outlined in the guide below to ensure consistent batch-to-batch quality for research and development teams aiming to implement this technology.
- Perform Mitsunobu reaction between a chiral diol and 3-hydroxy-2-halopyridine using DIAD and triphenylphosphine to form the bis-ether intermediate.
- Execute Ullmann coupling using either copper powder at high temperature or in-situ generated nickel(0) catalyst to close the biaryl bond.
- Purify the crude product through acid-base extraction and column chromatography to isolate the optically pure bipyridine ligand.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this synthetic route offers compelling advantages that directly address the pain points of sourcing high-value chiral intermediates. By eliminating the need for chiral resolution, the process inherently doubles the theoretical yield compared to racemic synthesis followed by separation, leading to a drastic reduction in the consumption of starting materials and solvents. The reliance on commodity chemicals such as copper powder, zinc dust, and common organic solvents ensures that the supply chain is resilient against fluctuations in the availability of exotic catalysts, thereby enhancing the overall reliability of supply for long-term manufacturing contracts.
- Cost Reduction in Manufacturing: The economic viability of this method is underpinned by the use of stoichiometric copper powder or inexpensive nickel salts instead of precious metal catalysts like palladium, which significantly lowers the direct material cost per kilogram of product. Furthermore, the avoidance of resolution steps means that no valuable chiral material is discarded as waste, effectively maximizing the atom economy and reducing the burden on waste treatment facilities. The simplified purification protocol, which relies on standard acid-base extractions rather than preparative HPLC or complex crystallizations, further drives down processing costs and energy consumption.
- Enhanced Supply Chain Reliability: The starting materials, specifically 3-hydroxy-2-halopyridines and simple chiral diols like pentanediol, are commercially available in bulk quantities from multiple global suppliers, mitigating the risk of single-source dependency. The robustness of the reaction conditions, which tolerate a range of temperatures and solvent qualities, ensures that production schedules are not easily disrupted by minor variations in raw material specifications. This stability is crucial for maintaining continuous production lines and meeting the tight delivery windows demanded by downstream pharmaceutical and agrochemical clients.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction vessels and conditions that are easily transferable from gram-scale laboratory experiments to multi-kilogram pilot plants. The use of DMF and THF, while requiring careful handling, allows for established recycling protocols that align with modern environmental, health, and safety (EHS) standards. Additionally, the generation of solid waste is minimized primarily to spent copper or zinc salts, which can be managed through standard hazardous waste disposal channels, ensuring compliance with increasingly strict regulatory frameworks governing chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthetic pathway, derived directly from the experimental data and scope defined in the patent documentation. These insights are intended to assist technical teams in evaluating the feasibility of adopting this method for their specific ligand requirements and process constraints.
Q: What are the advantages of this synthesis method over traditional resolution?
A: This method utilizes chiral pool starting materials (chiral diols) to induce axial chirality directly, eliminating the need for costly and wasteful resolution steps typically required for racemic bipyridines.
Q: Can this process be scaled for industrial production?
A: Yes, the protocol employs robust conditions using common solvents like THF and DMF, and utilizes inexpensive metal promoters like copper powder or nickel salts, making it highly suitable for commercial scale-up.
Q: What represents the key innovation in the coupling step?
A: The innovation lies in the successful application of Ullmann coupling on the sterically hindered bis-ether intermediate, achieving biaryl bond formation without racemization of the chiral backbone.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Axially Chiral Bipyridine Ligands Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance chiral ligands play in advancing the frontiers of asymmetric synthesis and materials science. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory curiosity to industrial reality is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of axially chiral bipyridine ligands performs consistently in your catalytic applications.
We invite you to collaborate with us to leverage this innovative synthetic technology for your next project. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can support your supply chain goals and drive value for your organization.
