Advanced Homogeneous Oxidation Process for High-Purity Nopinic Acid Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to transform abundant natural feedstocks into high-value active intermediates. Patent CN1331837C introduces a groundbreaking advancement in the synthesis of Nopinic acid, a critical intermediate utilized in the treatment of digestive tract ulcers. This technology addresses long-standing inefficiencies in terpene oxidation by shifting from traditional heterogeneous systems to a highly efficient homogeneous phase oxidation process. By leveraging potassium permanganate in a specialized tert-butyl alcohol and water solvent matrix, the invention achieves a transformative yield improvement, pushing production efficiency beyond the 70.0% threshold. This represents a paradigm shift for manufacturers aiming to secure a reliable pharmaceutical intermediates supplier capable of delivering consistent quality at scale. The process not only enhances economic viability but also aligns with modern green chemistry principles by simplifying downstream processing and reducing waste generation associated with low-yield legacy methods.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of Nopinic acid from beta-pinene has been plagued by significant technical bottlenecks that hindered its widespread commercial adoption. Traditional methodologies predominantly relied on heterogeneous reaction systems, which inherently suffer from poor mass transfer efficiency between the organic substrate and the aqueous oxidant. To mitigate these kinetic barriers, prior art processes were forced to employ expensive and often toxic phase-transfer catalysts, adding layers of complexity to both the reaction setup and the subsequent purification stages. Furthermore, these legacy techniques treated Nopinic acid merely as a minor by-product of nopinone synthesis, resulting in abysmal yields that typically hovered between a mere 20% and 30%. Such low efficiency not only inflated the cost of goods sold but also generated substantial chemical waste, creating a heavy burden on environmental compliance and waste treatment infrastructure. The reliance on multiphase systems also introduced variability in batch-to-batch consistency, making it difficult for procurement teams to guarantee supply continuity for sensitive pharmaceutical formulations.
The Novel Approach
In stark contrast to these outdated practices, the novel approach detailed in the patent data utilizes a sophisticated homogeneous phase oxidation strategy that fundamentally resolves the mass transfer limitations of the past. By employing a mixed solvent system comprising tert-butyl alcohol and water, the reaction medium becomes fully miscible with both the beta-pinene substrate and the potassium permanganate oxidant, creating a single-phase environment that maximizes molecular collision frequency. This ingenious solvent engineering eliminates the absolute necessity for phase-transfer catalysts, thereby stripping away a significant cost center and potential source of metallic contamination. The result is a streamlined operational workflow where the oxidation proceeds with exceptional selectivity and vigor, driving the yield of Nopinic acid to exceed 70.0%. This dramatic improvement in atom economy translates directly into cost reduction in pharma manufacturing, allowing producers to extract significantly more value from every kilogram of raw beta-pinene input. The simplicity of the workup procedure, involving straightforward hot filtration and crystallization, further underscores the commercial superiority of this method over the convoluted separation protocols required by heterogeneous alternatives.
Mechanistic Insights into KMnO4-Mediated Homogeneous Oxidation
The core of this technological breakthrough lies in the precise manipulation of reaction conditions to facilitate a controlled oxidative cleavage of the double bond in beta-pinene. The mechanism initiates with the formation of a cyclic manganate ester intermediate, which is stabilized by the alkaline environment provided by sodium or potassium hydroxide. The presence of tert-butyl alcohol is not merely incidental but acts as a crucial co-solvent that modulates the polarity of the reaction medium, ensuring that the hydrophobic terpene remains in solution throughout the oxidative attack. As the reaction progresses, the manganese species undergoes reduction, eventually precipitating as manganese dioxide, which is easily removed via the specified hot filtration step. This clean separation of the inorganic by-product is vital for maintaining the purity profile of the organic filtrate, preventing the carryover of heavy metal impurities that could compromise downstream pharmaceutical applications. The subsequent heating to 75-90°C serves to decompose any unstable intermediates and ensures the complete conversion of the substrate, locking in the high yield potential of the homogeneous system.
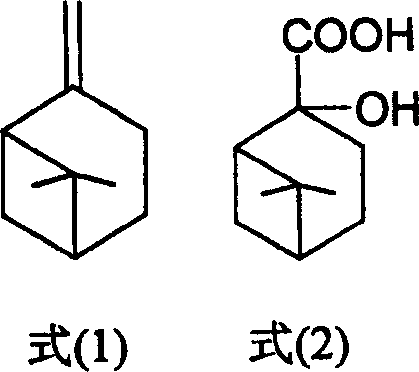
Controlling the impurity profile is paramount when producing intermediates for medical use, and this process incorporates several built-in safeguards to ensure high-purity Nopinic acid. The strict temperature control during the titration phase, maintained between 0-5°C, is critical for suppressing exothermic runaway reactions that could lead to over-oxidation or the formation of ring-opened degradation products. By carefully managing the stoichiometry of potassium permanganate to beta-pinene within the optimal range of 2.5:1 to 3.3:1, the process minimizes the presence of unreacted starting material while avoiding excessive oxidant that could attack the carboxylic acid functionality. The purification strategy, which involves converting the crude acid to its sodium salt followed by recrystallization, leverages the differential solubility of the target molecule versus potential isomers or oligomers. This multi-stage purification ensures that the final product meets the stringent purity specifications required for the synthesis of organic ammonium salts used in ulcer therapy, effectively mitigating the risk of toxicological issues arising from trace contaminants.
How to Synthesize Nopinic Acid Efficiently
Implementing this synthesis route requires careful attention to the sequential addition of reagents and thermal management to replicate the high yields reported in the patent examples. The process begins with the preparation of the oxidizing solution, followed by the controlled addition of the terpene substrate, and concludes with a robust isolation protocol involving acidification and recrystallization. Operators must adhere strictly to the temperature gradients specified, particularly during the exothermic addition phase, to ensure safety and reproducibility. The detailed standardized synthesis steps below outline the precise operational parameters required to achieve commercial-grade results, serving as a foundational guide for process engineers looking to adopt this technology.
- Mix potassium permanganate, alkali, tert-butyl alcohol, and water, then cool the mixture to 0-30°C before titrating beta-pinene while maintaining temperature between 0-5°C.
- Heat the reactants to 75-90°C after titration, filter immediately while hot, concentrate the filtrate, and purify the resulting solid to obtain sodium nopinate.
- Acidify the sodium nopinate solution with acid to pH 2-3, separate the precipitated Nopinic acid, and purify via recrystallization to obtain the final high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this homogeneous oxidation process offers a compelling value proposition that extends far beyond simple yield metrics. The elimination of phase-transfer catalysts represents a direct removal of a costly raw material line item, while simultaneously simplifying the supply chain by reducing the number of distinct chemical inputs required for production. The substantial increase in yield from roughly 30% to over 70% effectively more than doubles the output capacity of existing reactor infrastructure without the need for capital expenditure on new equipment. This intensification of production capability significantly reduces the lead time for high-purity carboxylic acids, allowing manufacturers to respond more agilely to market demand fluctuations. Furthermore, the use of beta-pinene, a readily available derivative of turpentine, ensures a stable and abundant raw material base, insulating the supply chain from the volatility often associated with synthetic petrochemical feedstocks.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the drastic simplification of the reaction system and the doubling of theoretical yield. By removing the need for expensive phase-transfer catalysts and their subsequent removal steps, the operational expenditure per kilogram of product is significantly lowered. The higher conversion rate means that less raw beta-pinene is wasted in the form of unreacted material or degraded by-products, maximizing the return on investment for every ton of feedstock purchased. Additionally, the simplified workup procedure reduces the consumption of solvents and energy required for extensive purification, contributing to a leaner and more cost-effective manufacturing model that enhances overall profit margins.
- Enhanced Supply Chain Reliability: Sourcing reliability is bolstered by the use of beta-pinene, a commodity chemical with a well-established global supply network derived from the pulp and paper industry. Unlike specialized synthetic precursors that may be subject to single-source bottlenecks, beta-pinene is produced in large volumes, ensuring that raw material availability remains robust even during periods of high market demand. The robustness of the reaction conditions, which tolerate a range of temperatures and stoichiometric ratios without catastrophic failure, further enhances process reliability, reducing the risk of batch failures that could disrupt delivery schedules. This stability allows supply chain planners to forecast production outputs with greater confidence, ensuring continuous availability for downstream pharmaceutical clients.
- Scalability and Environmental Compliance: The transition to a homogeneous system greatly facilitates the commercial scale-up of complex terpenes, as heat and mass transfer issues are minimized compared to slurry-based heterogeneous reactions. The process generates manganese dioxide as a solid by-product, which is easily filtered and can potentially be recycled or disposed of according to standard hazardous waste protocols, simplifying environmental compliance. The absence of chlorinated solvents in the reaction phase (using tert-butyl alcohol and water instead) aligns with increasingly strict environmental regulations regarding volatile organic compound (VOC) emissions. This eco-friendly profile not only reduces the regulatory burden on the manufacturing site but also appeals to end-users who are prioritizing sustainable sourcing in their supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Nopinic acid synthesis technology. These insights are derived directly from the patent specifications and are intended to clarify the operational advantages and chemical principles underpinning the process. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing production portfolios.
Q: What is the primary advantage of this homogeneous oxidation method over conventional techniques?
A: The primary advantage is the elimination of phase-transfer catalysts and the transition to a homogeneous reaction system, which drastically increases the yield from a historical 20-30% to over 70.0%, while simplifying the purification process.
Q: What are the critical temperature controls required during the beta-pinene titration?
A: Strict temperature control is essential; the initial mixture must be cooled to 0-30°C, and during the滴加 (titration) of beta-pinene, the temperature variation must be tightly controlled within the 0-5°C range to prevent side reactions and ensure safety.
Q: How is the final Nopinic acid purified to meet pharmaceutical standards?
A: The process involves converting the intermediate to sodium nopinate, followed by acidification to pH 2-3. The crude acid is then subjected to rigorous recrystallization, typically using benzene or similar solvents, to ensure high purity suitable for pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nopinic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of high-efficiency intermediates like Nopinic acid in the development of next-generation gastrointestinal therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and risk-mitigated. We are committed to maintaining stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify that every batch meets the exacting standards required for pharmaceutical synthesis. Our infrastructure is designed to handle the specific thermal and safety requirements of oxidation chemistries, providing a secure and compliant environment for the manufacture of sensitive fine chemicals.
We invite you to collaborate with us to leverage this advanced oxidation technology for your supply chain needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this high-yield process. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can drive value and efficiency for your organization.
