Advanced Synthetic Route for Canagliflozin Intermediate: Enhancing Purity and Scalability
The pharmaceutical landscape for Type 2 diabetes management has been significantly reshaped by SGLT-2 inhibitors, with Canagliflozin standing out as a pivotal therapeutic agent. At the heart of manufacturing this critical medication lies the efficient production of its key building block, 2-(4-fluorophenyl)-5-[(5-bromo-2-methylphenyl)methyl]thiophene. Patent CN110724123A, published in early 2020, introduces a transformative synthetic methodology that addresses long-standing bottlenecks in the supply chain of this high-value intermediate. By shifting away from complex thiophene precursors and utilizing a linear construction strategy starting from commodity aromatics, this technology offers a robust pathway for reliable pharmaceutical intermediate supplier networks to enhance their production capabilities. The innovation focuses on simplifying the molecular assembly while maintaining rigorous quality standards essential for regulatory compliance.
![Chemical structure of 2-(4-fluorophenyl)-5-[(5-bromo-2-methylphenyl)methyl]thiophene](/insights/img/canagliflozin-intermediate-synthesis-pharma-supplier-20260305040034-01.png)
This structural motif is not merely a chemical curiosity but a fundamental component determining the efficacy of the final drug substance. The patent details a sequence that leverages classic organic transformations—specifically Friedel-Crafts chemistry and heterocyclic cyclization—but optimizes them for modern manufacturing constraints. For R&D directors evaluating process feasibility, the shift towards using 4-bromotoluene and ethyl 5-bromolevulinate as primary feedstocks represents a strategic move towards cost-effective and logistically simpler sourcing. This approach mitigates the risks associated with sourcing specialized, low-volume heterocycles that often plague the early stages of API synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this Canagliflozin intermediate has relied on pathways that introduce significant operational friction and cost volatility. As illustrated in prior art such as WO2012160218 and CN104892566A, the traditional route necessitates the prior preparation of 2-(4-fluorophenyl)thiophene. This prerequisite step is chemically demanding, often requiring specific coupling conditions that limit throughput and increase the cumulative impurity profile before the main skeleton is even assembled. Furthermore, alternative cyclization methods described in patents like CN102115468A utilize unstable reagents such as 3-chloro-3-(4-fluorophenyl)-2-propenal.
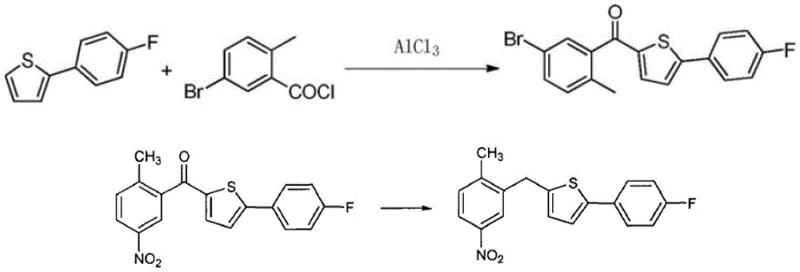
The instability of these aldehyde intermediates poses severe challenges for commercial scale-up of complex pharmaceutical intermediates. They are prone to oxidation upon exposure to air, necessitating inert atmosphere handling and immediate consumption, which disrupts continuous processing workflows. Additionally, the yields associated with these older methodologies are often suboptimal, leading to excessive solvent usage and waste generation. For procurement managers, these factors translate into unpredictable lead times and inflated raw material costs, as suppliers must price in the risk of reagent degradation and lower process efficiency. The reliance on such fragile intermediates creates a bottleneck that hinders the ability to respond rapidly to market demand surges.
The Novel Approach
The methodology disclosed in CN110724123A fundamentally re-engineers the synthesis by constructing the thiophene ring at the final stage rather than introducing it as a pre-formed unit. This "build-up" strategy begins with a Friedel-Crafts alkylation between 4-bromotoluene and ethyl 5-bromolevulinate, catalyzed by aluminum trichloride. This initial step effectively installs the carbon backbone with high regioselectivity, leveraging the activating effects of the methyl group on the toluene ring. Subsequent hydrolysis converts the ester to a carboxylic acid, setting the stage for a second Friedel-Crafts acylation with fluorobenzene.
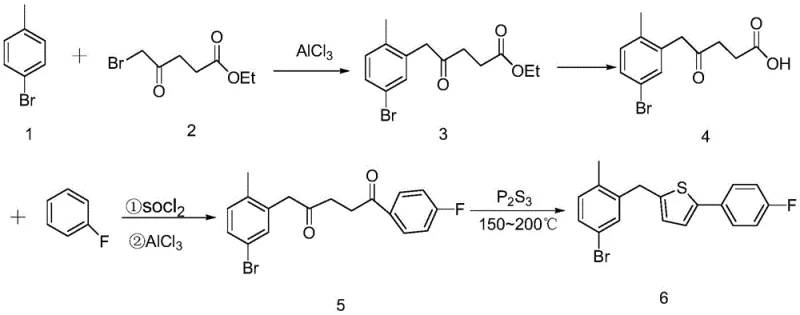
The culmination of this sequence is a thermal cyclization using phosphorus trisulfide (P2S3) in diphenyl ether at elevated temperatures (150-200°C). This thionation-cyclization cascade is remarkably efficient, closing the ring to form the target thiophene structure in a single pot operation. By deferring the formation of the sensitive heterocycle until the end, the process avoids the handling issues associated with unstable thiophene precursors. This results in a streamlined workflow that is inherently safer and more amenable to automation. For supply chain heads, this translates to reducing lead time for high-purity pharmaceutical intermediates by eliminating the need to procure and quality-control multiple complex starting materials, consolidating the value chain into a more manageable and resilient operation.
Mechanistic Insights into AlCl3-Catalyzed Friedel-Crafts and P2S3 Cyclization
The core of this synthetic innovation lies in the precise control of electrophilic aromatic substitutions and the subsequent heterocyclization. In the first step, aluminum trichloride (AlCl3) acts as a potent Lewis acid, coordinating with the carbonyl oxygen of the bromolevulinate to generate a highly reactive acylium-like species. This electrophile attacks the para-position of the 4-bromotoluene relative to the methyl group, driven by steric and electronic factors. The reaction conditions, maintained between -10°C and room temperature, are critical for suppressing poly-alkylation side reactions, ensuring that the mono-substituted keto-ester (Compound 3) is formed with high selectivity. This control is vital for minimizing downstream purification loads.
Following hydrolysis to the acid (Compound 4), the molecule undergoes activation via thionyl chloride to form an acid chloride in situ. This activated species then participates in a second Friedel-Crafts acylation with fluorobenzene. The choice of fluorobenzene is strategic; the fluorine atom deactivates the ring slightly but directs the incoming acyl group to the para-position, yielding the 1,4-diketone precursor (Compound 5). The final transformation involves the reaction of this 1,4-diketone with P2S3. Mechanistically, P2S3 serves as a sulfur donor, converting the carbonyl groups into thiocarbonyl intermediates which spontaneously cyclize with the loss of water (or H2S equivalents) to aromatize into the thiophene ring. This high-temperature step (150-200°C) drives the equilibrium towards the stable aromatic product, effectively locking in the structural integrity of the final intermediate.
Impurity control is embedded within the mechanistic design of this route. By avoiding the use of pre-formed thiophenes, the process eliminates impurities related to thiophene ring substitution patterns (e.g., 2,5- vs 2,4-disubstitution) that are common in direct functionalization approaches. Furthermore, the distinct polarity differences between the linear diketone precursor and the final aromatic thiophene product facilitate easy separation via crystallization or extraction. The patent reports an HPLC purity of 99.6% for the final product, demonstrating the efficacy of this mechanistic pathway in delivering high-purity pharmaceutical intermediates suitable for direct use in subsequent API synthesis steps without extensive chromatographic purification.
How to Synthesize 2-(4-Fluorophenyl)-5-[(5-bromo-2-methylphenyl)methyl]thiophene Efficiently
The operational protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale execution. The process is divided into four distinct operational units: alkylation, hydrolysis, acylation, and cyclization. Each step utilizes standard chemical engineering unit operations such as stirred tank reactors, phase separation, and distillation. The use of common solvents like dichloromethane, methanol, and diphenyl ether ensures that the process can be implemented in existing multipurpose facilities without requiring specialized corrosion-resistant equipment beyond standard glass-lined or stainless steel reactors. The detailed standardized synthesis steps below outline the specific molar ratios and temperature profiles required to replicate the high yields reported in the intellectual property.
- Perform Friedel-Crafts alkylation of 4-bromotoluene with ethyl 5-bromolevulinate using AlCl3 catalyst to form the keto-ester intermediate.
- Hydrolyze the ester group under basic conditions (NaOH/MeOH) to yield the corresponding carboxylic acid derivative.
- Convert the acid to acid chloride using thionyl chloride, followed by Friedel-Crafts acylation with fluorobenzene to generate the diketone precursor.
- Execute thermal cyclization using phosphorus trisulfide (P2S3) in diphenyl ether at elevated temperatures to form the final thiophene ring.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers compelling advantages that directly impact the bottom line and supply security. The shift from specialized heterocyclic starting materials to commodity aromatics like 4-bromotoluene drastically simplifies the procurement landscape. These raw materials are produced on a massive scale globally, ensuring consistent availability and shielding the supply chain from the volatility often seen in niche fine chemical markets. This stability is crucial for maintaining continuous API production schedules and meeting the rigorous Just-In-Time delivery requirements of major pharmaceutical clients.
- Cost Reduction in Manufacturing: The elimination of the pre-synthesis step for 2-(4-fluorophenyl)thiophene removes an entire layer of processing costs, including solvent recovery, catalyst consumption, and labor associated with that isolated step. Furthermore, the avoidance of unstable aldehyde reagents reduces waste disposal costs associated with degraded materials. The overall atom economy is improved by constructing the molecule from simpler blocks, leading to substantial cost savings in raw material expenditure and overhead allocation per kilogram of finished intermediate.
- Enhanced Supply Chain Reliability: By relying on robust, shelf-stable starting materials, the risk of production stoppages due to reagent spoilage is virtually eliminated. The process tolerances described in the patent (e.g., reaction temperatures ranging from 0°C to 200°C) are well within the capabilities of standard industrial infrastructure, reducing the dependency on specialized cryogenic or high-pressure equipment. This flexibility allows for broader supplier qualification, enabling procurement teams to diversify their vendor base and mitigate single-source risks effectively.
- Scalability and Environmental Compliance: The synthetic sequence generates fewer byproducts compared to multi-step coupling routes, simplifying effluent treatment. The final cyclization step, while requiring heat, does not produce heavy metal waste, aligning with increasingly stringent environmental regulations. The high purity of the crude product reduces the need for energy-intensive recrystallization cycles, further lowering the carbon footprint of the manufacturing process and supporting sustainability goals within the pharmaceutical supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and process descriptions provided in CN110724123A, offering clarity on how this method compares to legacy processes in terms of operational parameters and output quality. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer.
Q: What are the key advantages of this new synthetic route over conventional methods?
A: This route eliminates the need for pre-synthesizing complex thiophene derivatives like 2-(4-fluorophenyl)thiophene. It utilizes readily available commodity chemicals (4-bromotoluene) and avoids unstable intermediates like chloro-propenals found in older patents, resulting in higher overall stability and easier scale-up.
Q: How does the process ensure high purity for the final API intermediate?
A: The process employs robust purification steps including acid-base extractions and recrystallization. The final cyclization step yields a product with HPLC purity exceeding 99.6%, minimizing downstream purification burdens for the active pharmaceutical ingredient manufacturer.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method is designed for industrial viability. It avoids cryogenic conditions (operating mostly between 0°C and 200°C), uses standard solvents like dichloromethane and diphenyl ether, and relies on common Lewis acid catalysts, making it highly adaptable for multi-ton manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(4-Fluorophenyl)-5-[(5-bromo-2-methylphenyl)methyl]thiophene Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires more than just chemical knowledge; it demands engineering excellence and unwavering commitment to quality. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped to handle the specific thermal and chemical requirements of this Friedel-Crafts and cyclization sequence, ensuring that the stringent purity specifications demanded by global regulatory bodies are consistently met. Our rigorous QC labs employ state-of-the-art analytical techniques to monitor every batch, guaranteeing that the impurity profile remains well within acceptable limits for downstream API synthesis.
We invite pharmaceutical manufacturers and procurement leaders to collaborate with us to leverage this advanced synthetic route for their Canagliflozin supply chains. By partnering with our technical team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. We encourage you to contact our technical procurement team today to request specific COA data from our pilot batches and discuss route feasibility assessments for your upcoming projects. Let us help you secure a competitive advantage through superior chemistry and reliable supply.
