Advanced Palladium-Catalyzed Synthesis of Chiral 1,2-Diol Esters for Commercial Scale-up
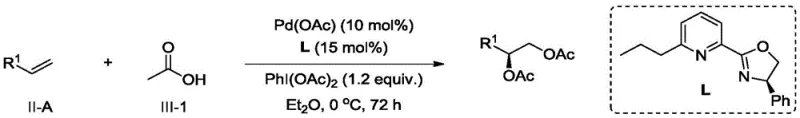
The pharmaceutical and fine chemical industries are constantly seeking more efficient and sustainable pathways to access chiral building blocks, particularly 1,2-diol esters which serve as critical precursors for complex active pharmaceutical ingredients. Patent CN114249682B introduces a groundbreaking preparation method that leverages a palladium-catalyzed asymmetric double oxidation strategy to transform inexpensive olefins into high-value chiral diol esters. This technology represents a significant paradigm shift away from traditional stoichiometric oxidants, offering a catalytic system that combines high enantioselectivity with exceptional functional group tolerance. By utilizing a specialized oxazoline ligand system in conjunction with hypervalent iodine oxidants, this process achieves stereochemical control that was previously difficult to attain with non-activated terminal olefins. The implications for industrial synthesis are profound, as it opens new avenues for constructing chiral centers without the environmental and safety burdens associated with heavy metal waste streams.
For research and development teams focused on process chemistry, the ability to access these motifs through a direct oxidative functionalization of alkenes simplifies synthetic routes considerably. The patent data indicates that this method is not merely a laboratory curiosity but a robust protocol capable of handling a diverse array of substrate structures, including those with sensitive functional groups that would typically degrade under harsher oxidative conditions. This versatility makes it an attractive candidate for the late-stage functionalization of complex molecules, allowing medicinal chemists to rapidly generate analog libraries. Furthermore, the mild reaction conditions, often operating between -20°C and 30°C, reduce the energy footprint of the manufacturing process, aligning with modern green chemistry principles that are increasingly mandated by global regulatory bodies.

The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral 1,2-diols and their ester derivatives has relied heavily on the Sharpless asymmetric dihydroxylation, which utilizes osmium tetroxide as the key catalyst. While effective for certain substrate classes, this conventional approach suffers from significant drawbacks that hinder its widespread adoption in cost-sensitive commercial manufacturing. Osmium is an extremely rare and expensive precious metal, and its tetroxide form is highly toxic and volatile, requiring stringent safety protocols and specialized containment equipment that drive up capital expenditure. Moreover, the removal of trace osmium residues from the final active pharmaceutical ingredient is a challenging and costly purification step, often requiring additional scavenging resins or complex crystallization processes to meet regulatory limits. Additionally, the substrate scope of osmium-catalyzed reactions is often limited, particularly struggling with less sterically hindered mono-substituted terminal olefins which are common feedstocks in the petrochemical industry.
Beyond the safety and cost issues, the environmental impact of stoichiometric oxidants used in traditional methods cannot be overlooked. Many older protocols generate substantial amounts of heavy metal waste, creating disposal challenges and increasing the overall carbon footprint of the synthesis. The lack of atom economy in these processes means that a significant portion of the starting materials ends up as waste rather than incorporated into the desired product. For procurement managers and supply chain heads, these inefficiencies translate into higher raw material costs and greater supply chain volatility due to the reliance on scarce metals. The difficulty in scaling these reactions safely further complicates technology transfer from the lab to the pilot plant, often resulting in prolonged development timelines and delayed market entry for new drug candidates. These cumulative factors create a strong imperative for the industry to adopt alternative catalytic systems that mitigate these risks.
The Novel Approach
The methodology disclosed in CN114249682B addresses these historical pain points by introducing a palladium-catalyzed system that operates with high efficiency and selectivity. Unlike osmium, palladium is more abundant and generally less toxic, allowing for safer handling and easier integration into existing manufacturing infrastructure. The core innovation lies in the use of chiral oxazoline ligands which create a highly defined steric environment around the palladium center, enabling precise control over the facial selectivity of the oxidation event. This ligand design allows the catalyst to differentiate between the enantiotopic faces of the olefin substrate even when the steric differences are minimal, such as in linear terminal alkenes. The result is a process that delivers high enantiomeric excess values consistently across a broad range of substrates, eliminating the need for costly chiral resolution steps downstream.
Furthermore, this novel approach utilizes hypervalent iodine compounds as terminal oxidants, which are commercially available and generate benign byproducts upon reduction. This choice of oxidant significantly simplifies the workup procedure, as the reduced iodine species are typically soluble in aqueous phases and can be easily washed away, leaving the organic product in high purity. The reaction conditions are remarkably mild, often proceeding at or near room temperature, which reduces energy consumption and minimizes the risk of thermal runaway incidents during scale-up. For process engineers, this translates to a more robust and forgiving protocol that can be adapted to continuous flow reactors or large batch vessels with minimal modification. The combination of safety, efficiency, and selectivity makes this palladium-catalyzed double oxidation a superior alternative for the commercial production of chiral intermediates.
Mechanistic Insights into Pd-Catalyzed Asymmetric Double Oxidation
Understanding the mechanistic underpinnings of this transformation is crucial for R&D directors aiming to optimize the process for specific applications. The catalytic cycle begins with the coordination of the olefin substrate to the palladium center, which is pre-complexed with the chiral oxazoline ligand. This coordination step is reversible and establishes the initial orientation of the substrate within the chiral pocket of the catalyst. The hypervalent iodine oxidant then interacts with the palladium-olefin complex, facilitating the transfer of oxygen atoms to the double bond. The chiral ligand plays a pivotal role in this transition state, imposing steric constraints that favor the formation of one enantiomer over the other through a well-defined transition state geometry. Detailed kinetic studies suggest that the rate-determining step involves the nucleophilic attack of the carboxylate on the activated olefin-palladium species, a step that is highly sensitive to the electronic properties of both the ligand and the substrate.
The exceptional enantioselectivity observed in this system, often exceeding 90% ee, is a direct result of the rigid structure of the oxazoline ligand which prevents free rotation and maintains a consistent chiral environment throughout the catalytic cycle. This rigidity ensures that the differentiation between the pro-chiral faces of the olefin is maximized, leading to high optical purity in the final product. Impurity control is further enhanced by the chemoselectivity of the palladium catalyst, which tolerates a wide range of functional groups such as esters, ethers, and halides without causing side reactions like over-oxidation or polymerization. The patent data confirms that even substrates with electron-withdrawing groups or bulky substituents can be processed effectively, demonstrating the robustness of the catalytic system. This level of control is essential for pharmaceutical manufacturing where impurity profiles must be strictly managed to ensure patient safety and regulatory compliance.
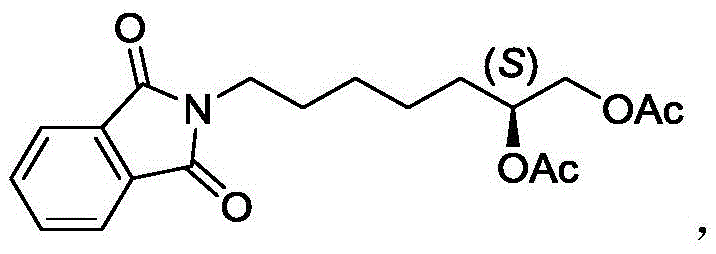
How to Synthesize Chiral 1,2-Diol Esters Efficiently
To implement this synthesis route effectively, operators must adhere to strict anhydrous conditions and precise stoichiometric ratios as outlined in the patent examples. The standard protocol involves dissolving the olefin substrate and carboxylic acid in a dry ether solvent such as diethyl ether or tetrahydrofuran under an inert atmosphere. The palladium catalyst and chiral ligand are added sequentially, followed by the slow addition of the hypervalent iodine oxidant to control the exotherm. The reaction mixture is then stirred at low temperatures, typically between 0°C and -10°C, for an extended period ranging from 24 to 96 hours to ensure complete conversion. Monitoring the reaction progress via thin-layer chromatography or HPLC is recommended to determine the optimal endpoint before quenching with aqueous sodium thiosulfate to reduce residual iodine species.
- Combine the olefin substrate with carboxylic acid, palladium catalyst, oxazoline ligand, and hypervalent iodine oxidant in an anhydrous solvent.
- Stir the reaction mixture under a protective gas atmosphere at controlled low temperatures ranging from -20°C to 30°C for extended periods.
- Quench the reaction, extract with organic solvents, and purify the crude product via flash column chromatography to isolate the high-purity diol ester.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this palladium-catalyzed technology offers substantial strategic advantages for procurement and supply chain management. The primary benefit lies in the drastic reduction of raw material costs associated with replacing osmium-based reagents with more abundant palladium salts and organic ligands. This substitution eliminates the need for expensive heavy metal scavengers and complex purification trains, thereby streamlining the manufacturing process and reducing the overall cost of goods sold. Additionally, the use of readily available terminal olefins as starting materials ensures a stable and diversified supply chain, reducing dependency on specialized and potentially volatile precursor markets. The robustness of the reaction conditions also minimizes the risk of batch failures, leading to higher overall yields and more predictable production schedules which are critical for meeting just-in-time delivery commitments.
Supply chain reliability is further enhanced by the scalability of this process, which has been demonstrated to work efficiently from gram scale to multi-kilogram batches without loss of selectivity or yield. The mild reaction temperatures reduce the energy load on manufacturing facilities, contributing to lower utility costs and a smaller environmental footprint. For sustainability officers, the reduced toxicity profile of the reagents simplifies waste management and lowers the costs associated with hazardous waste disposal. The high functional group tolerance means that fewer protection and deprotection steps are required in multi-step syntheses, shortening the overall production timeline and accelerating time-to-market for new products. These combined factors create a compelling economic case for integrating this technology into existing supply chains for fine chemical intermediates.
- Cost Reduction in Manufacturing: The elimination of toxic osmium reagents removes the need for costly safety containment systems and specialized waste treatment protocols, leading to significant operational savings. By utilizing a catalytic amount of palladium rather than stoichiometric amounts of heavy metals, the material cost per kilogram of product is substantially lowered. The simplified workup procedure reduces solvent consumption and labor hours required for purification, further driving down manufacturing expenses. These efficiencies allow for more competitive pricing strategies in the global market for chiral intermediates.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as simple olefins and carboxylic acids ensures that raw material sourcing is not a bottleneck for production. Unlike specialized chiral pool starting materials which may have limited suppliers, the feedstocks for this process are widely available from multiple global vendors. This diversification mitigates the risk of supply disruptions and price spikes, providing greater stability for long-term production planning. The robustness of the catalyst system also means that variations in raw material quality have minimal impact on the final product, ensuring consistent output quality.
- Scalability and Environmental Compliance: The process is inherently scalable due to its mild thermal profile and lack of hazardous gas evolution, making it suitable for large-scale reactor operations. The reduced environmental impact aligns with increasingly strict global regulations on heavy metal emissions and solvent usage, future-proofing the manufacturing process against regulatory changes. The high atom economy of the reaction minimizes waste generation, supporting corporate sustainability goals and reducing the carbon footprint of the supply chain. This compliance advantage facilitates smoother regulatory approvals and market access in environmentally sensitive regions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation to ensure accuracy and relevance for potential partners. Understanding these details is essential for evaluating the feasibility of adopting this technology for specific project requirements.
Q: What are the advantages of this Pd-catalyzed method over traditional Osmium oxidation?
A: This method avoids the use of toxic and expensive osmium tetroxide, utilizing a more sustainable palladium system that offers broader substrate compatibility and easier downstream processing.
Q: What level of enantioselectivity can be achieved with this process?
A: The patented process demonstrates excellent stereocontrol, achieving enantiomeric excess (ee) values frequently exceeding 90% and up to 98% for specific substrates.
Q: Is this synthesis method suitable for large-scale manufacturing?
A: Yes, the reaction operates under mild conditions with readily available reagents, making it highly amenable to scale-up for commercial production of fine chemical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2-Diol Ester Supplier
NINGBO INNO PHARMCHEM stands at the forefront of implementing advanced catalytic technologies like the one described in CN114249682B to deliver high-quality chiral intermediates to the global market. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition seamlessly from development to full-scale manufacturing. Our facilities are equipped with state-of-the-art rigorous QC labs and stringent purity specifications to guarantee that every batch meets the highest industry standards. We understand the critical nature of supply chain continuity and are committed to providing reliable volumes of high-purity 1,2-diol esters to support your drug development and commercialization efforts.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific molecular targets. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the economic benefits of switching to this palladium-catalyzed method for your specific application. We encourage potential partners to contact us to obtain specific COA data and route feasibility assessments that demonstrate our capability to deliver on both quality and cost metrics. Let us collaborate to optimize your supply chain and accelerate the delivery of life-saving medicines to patients worldwide.
