Advanced Manufacturing of Chiral Ligand Intermediates for Stereoselective Catalysis
Advanced Manufacturing of Chiral Ligand Intermediates for Stereoselective Catalysis
The landscape of asymmetric synthesis is constantly evolving, driven by the demand for highly enantiopure building blocks in the pharmaceutical and agrochemical sectors. A pivotal development in this field is detailed in patent CN111205251B, which discloses a robust and economically viable preparation method for (3S,4S)-2,5-dioxytetrahydrofuran-3,4-bis-carbamic acid benzyl ester. This compound serves as a critical chiral ligand intermediate for transition metal catalysts, enabling the stereoselective oxidation of olefins and sulfides. The patented technology represents a significant leap forward in process chemistry, addressing long-standing bottlenecks related to cost, solubility, and operational complexity. By re-engineering the synthetic pathway from racemic-2,3-dibromosuccinic acid, the inventors have created a route that is not only chemically elegant but also commercially superior, positioning it as a key asset for any organization seeking a reliable chiral ligand supplier.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
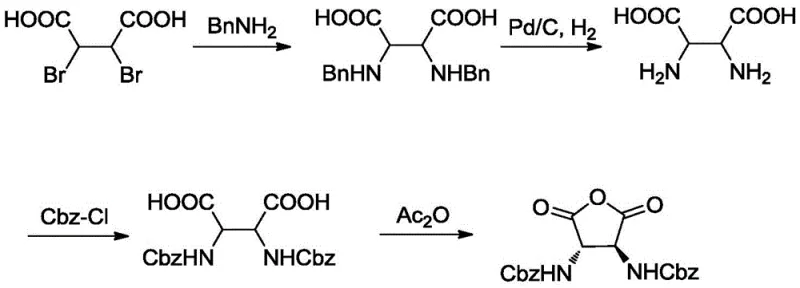
Historically, the synthesis of similar chiral succinic acid derivatives has relied on pathways that are fraught with inefficiencies and high operational costs. As illustrated in prior art literature, such as the methods reported in the Journal of the Chinese Chemical Society, traditional routes often involve the nucleophilic substitution of dibromosuccinic acid followed by hydrogenolysis to remove benzyl protecting groups. This conventional approach suffers from severe drawbacks, primarily the poor solubility of intermediate benzylamine acids and free amino acids in common organic solvents. This insolubility complicates extraction and purification, leading to significant product loss and extended processing times. Furthermore, the reliance on palladium-carbon (Pd/C) for deprotection introduces a dependency on expensive precious metal catalysts and requires specialized high-pressure hydrogenation equipment, which poses safety risks and increases capital expenditure for manufacturing facilities.
The Novel Approach
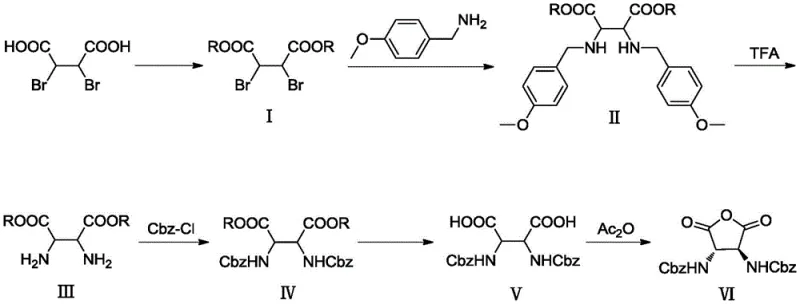
In stark contrast, the methodology outlined in CN111205251B introduces a strategic modification that circumvents these historical challenges. The core innovation lies in the substitution of the standard benzyl amine with p-methoxybenzylamine during the nucleophilic substitution step. This specific choice of protecting group allows for its subsequent removal via acidolysis using trifluoroacetic acid (TFA), completely eliminating the need for catalytic hydrogenation. Additionally, the process maintains the carboxylic acid functionalities as esters throughout the early and middle stages of the synthesis. This esterification strategy dramatically improves the solubility of the intermediates in solvents like dichloromethane and ethanol, facilitating smooth reaction kinetics and straightforward work-up procedures. The result is a streamlined process that enhances overall yield and purity while drastically simplifying the operational workflow for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into the Six-Step Synthetic Cascade
The chemical elegance of this process is rooted in its precise control over functional group transformations and stereochemistry. The synthesis initiates with the esterification of racemic-2,3-dibromosuccinic acid, converting the polar carboxylic acids into lipophilic esters (Compound I). This is followed by a double nucleophilic substitution where p-methoxybenzylamine displaces the bromide atoms. Crucially, this step establishes the nitrogen framework while retaining the carbon backbone integrity. The subsequent deprotection step utilizes the electron-donating methoxy group on the benzyl ring to facilitate cleavage under acidic conditions (TFA), generating the free diamine diester (Compound III) without affecting the ester moieties. This orthogonality is vital for maintaining the structural fidelity of the molecule before the introduction of the final Cbz protecting groups via acylation with benzyl chloroformate.
The final stages of the synthesis focus on constructing the rigid anhydride core essential for the ligand's chelating properties. Following the acylation, the ester groups are hydrolyzed under mild alkaline conditions to reveal the dicarboxylic acid (Compound V). The concluding step involves an intramolecular dehydration cyclization using acetic anhydride. This reaction closes the five-membered ring to form the 2,5-dioxytetrahydrofuran core. The mechanism here is driven by the proximity of the two carboxylic acid groups and the thermodynamic stability of the resulting cyclic anhydride. Throughout this cascade, the process avoids harsh conditions that could lead to racemization or epimerization, ensuring that the final product retains the high optical purity required for effective stereoselective catalysis in downstream applications.
How to Synthesize (3S,4S)-2,5-dioxytetrahydrofuran-3,4-bis-carbamic acid benzyl ester Efficiently
Executing this synthesis requires careful attention to reaction stoichiometry and temperature control, particularly during the exothermic esterification and acylation steps. The protocol is designed to be robust, utilizing readily available reagents such as thionyl chloride, triethylamine, and acetic anhydride. The transition from the open-chain dibromo precursor to the cyclic anhydride is achieved through a logical sequence of protection, deprotection, and cyclization events that maximize atom economy. For process chemists looking to implement this route, the key lies in managing the solubility transitions between the ester and acid forms of the intermediates. The detailed standardized operating procedures for each transformation, including specific molar ratios and quenching protocols, are essential for reproducing the high yields reported in the patent data.
- Esterification of racemic-2,3-dibromosuccinic acid with alcohol under acidic catalysis to form diester compound (I).
- Nucleophilic substitution of compound (I) with p-methoxybenzylamine to generate diamine diester compound (II).
- Removal of the p-methoxybenzyl protecting group using trifluoroacetic acid (TFA) to yield the free amino diester (III).
- Acylation of compound (III) with benzyl chloroformate (Cbz-Cl) to protect the amines as carbamates, forming compound (IV).
- Hydrolysis of the ester groups in compound (IV) under alkaline conditions to obtain the dicarboxylic acid derivative (V).
- Intramolecular dehydration of compound (V) using acetic anhydride to cyclize and form the final anhydride product (VI).
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented process offers compelling advantages that directly address the pain points of procurement managers and supply chain directors. The elimination of palladium catalysts is perhaps the most significant value driver. Not only does this remove the cost of the metal itself, but it also eradicates the need for expensive and time-consuming heavy metal scavenging steps to meet regulatory limits for residual metals in pharmaceutical ingredients. This simplification translates directly into lower production costs and a reduced environmental footprint. Furthermore, the improved solubility profile of the intermediates means that reactors can be operated at higher concentrations, increasing throughput per batch and reducing solvent consumption. These factors combine to create a manufacturing process that is inherently more cost-effective and sustainable than legacy methods.
- Cost Reduction in Manufacturing: The economic benefits of this route are substantial and multifaceted. By replacing the hydrogenation step with a simple acid treatment, the process removes the requirement for high-pressure reactors and the associated safety infrastructure. The avoidance of palladium on carbon eliminates a major variable cost and the logistical complexity of handling and disposing of spent precious metal catalysts. Additionally, the high yields reported in the patent examples suggest minimal waste generation, further driving down the cost of goods sold (COGS). The use of commodity chemicals like p-methoxybenzylamine and acetic anhydride ensures that raw material costs remain stable and predictable, shielding the supply chain from volatility associated with specialty reagents.
- Enhanced Supply Chain Reliability: Supply continuity is critical for the production of high-purity pharmaceutical intermediates. This synthesis route enhances reliability by relying on a broad base of commercially available starting materials rather than bespoke or scarce reagents. The robustness of the chemical steps, characterized by mild reaction temperatures and ambient pressure conditions, reduces the risk of batch failures due to equipment malfunction or operator error. The simplified work-up procedures, which rely on standard extractions and filtrations rather than complex chromatographic separations, ensure that production timelines are consistent. This predictability allows for better inventory planning and significantly reduces lead time for high-purity chiral ligands, ensuring that downstream customers receive their materials on schedule.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to the plant floor often reveals hidden challenges, but this route is designed with scalability in mind. The absence of gaseous reagents like hydrogen simplifies the engineering requirements for large-scale reactors. Moreover, the process generates less hazardous waste; the byproducts of the acidolysis and hydrolysis steps are generally easier to treat and dispose of compared to the heavy metal waste streams from hydrogenation. This alignment with green chemistry principles facilitates easier regulatory approval and environmental compliance. The ability to run reactions in common solvents like methanol and DCM, which have well-established recovery and recycling protocols, further supports the goal of sustainable commercial scale-up of complex fine chemicals.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is crucial for stakeholders evaluating its potential for integration into their supply chains. The following questions address common inquiries regarding the practical implementation and quality aspects of this technology. The answers are derived directly from the technical disclosures and experimental data provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers.
Q: How does this new synthesis route improve upon traditional methods for chiral ligand production?
A: The novel route described in patent CN111205251B eliminates the need for expensive palladium-on-carbon (Pd/C) catalysts and hydrogenation steps required in conventional literature methods. By utilizing p-methoxybenzylamine protection followed by acidolytic deprotection with trifluoroacetic acid, the process avoids the solubility issues associated with free amino acid intermediates and significantly reduces raw material costs while simplifying purification.
Q: What are the critical quality control parameters for this chiral transition metal catalyst intermediate?
A: Critical parameters include the stereochemical purity (ensuring the specific 3S,4S configuration), the absence of residual halides from the dibromo-succinic acid starting material, and the complete removal of protecting group byproducts. The final cyclization step must be monitored to ensure high conversion to the anhydride form without polymerization or degradation, which is achieved through controlled temperature and stoichiometry of the dehydrating agent.
Q: Is this manufacturing process suitable for large-scale industrial production?
A: Yes, the process is specifically designed for industrial scalability. It utilizes common solvents like DCM, methanol, and THF, and avoids hazardous high-pressure hydrogenation equipment. The intermediate compounds maintain good solubility throughout the sequence, facilitating efficient filtration and extraction operations, which are essential for reducing lead time and ensuring supply continuity in commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (3S,4S)-2,5-dioxytetrahydrofuran-3,4-bis-carbamic acid benzyl ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality chiral ligands play in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of global pharmaceutical clients. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our expertise in process optimization allows us to adapt patented methodologies like CN111205251B to fit specific customer requirements, guaranteeing a supply of materials that consistently exceed industry standards for optical purity and chemical integrity.
We invite you to collaborate with us to leverage this advanced synthetic technology for your projects. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to make informed decisions about integrating this efficient and cost-effective chiral ligand intermediate into your manufacturing pipeline. Together, we can drive innovation and efficiency in the field of asymmetric synthesis.
