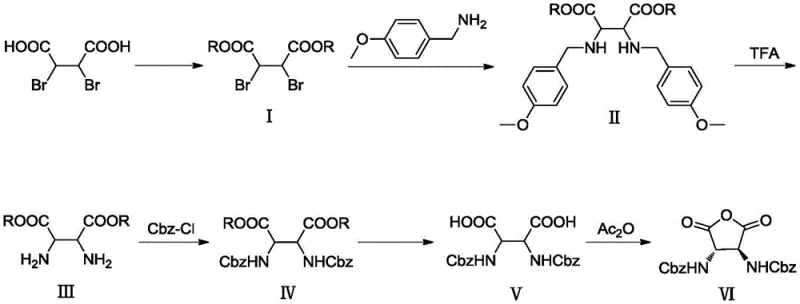
Advanced Manufacturing of Chiral Ligand Intermediates for High-Purity Catalyst Production
The landscape of asymmetric catalysis is constantly evolving, driven by the demand for higher enantiomeric purity and more sustainable manufacturing processes in the pharmaceutical and fine chemical sectors. A significant breakthrough in this domain is detailed in patent CN111205251B, which discloses a novel preparation method for the chiral transition metal catalyst intermediate known as (3S,4S)-2,5-dioxytetrahydrofuran-3,4-bis-benzyl carbamate. This compound serves as a critical ligand precursor for the stereoselective oxidation of olefins and sulfides, reactions that are foundational for synthesizing complex natural products and active pharmaceutical ingredients (APIs). The patent outlines a robust, six-step synthetic strategy that fundamentally reimagines the production workflow, moving away from problematic solubility issues and expensive noble metal catalysts towards a streamlined, ester-based methodology. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, understanding the nuances of this improved pathway is essential for securing a stable supply chain of high-value chiral building blocks.
The development of efficient routes for chiral ligands is not merely an academic exercise but a commercial imperative. Traditional methods often suffer from bottlenecks that hinder large-scale production, such as the inability to filter intermediates or the requirement for specialized high-pressure reactors. The technology described in CN111205251B addresses these pain points directly by introducing a sequence that maintains intermediates in solution phase for longer durations, thereby facilitating easier handling and purification. This shift represents a paradigm change in how we approach the commercial scale-up of complex pharmaceutical intermediates, offering a blueprint for reducing lead times and enhancing overall process reliability without compromising on the stringent stereochemical requirements demanded by modern drug synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the standard literature route for synthesizing similar succinic acid derivatives relied heavily on the use of benzylamine as the nitrogen source. As illustrated in the retrospective analysis of older methodologies, this approach presented severe logistical and economic challenges. The primary drawback was the physicochemical nature of the intermediates; specifically, the benzylamine-substituted succinic acids exhibited extremely poor solubility in common organic solvents. This lack of solubility made liquid-liquid extraction nearly impossible, forcing manufacturers to rely on difficult filtration processes that often resulted in significant product loss and low overall yields. Furthermore, the deprotection step in the conventional route necessitated the use of palladium on carbon (Pd/C) under a hydrogen atmosphere to remove the benzyl groups.
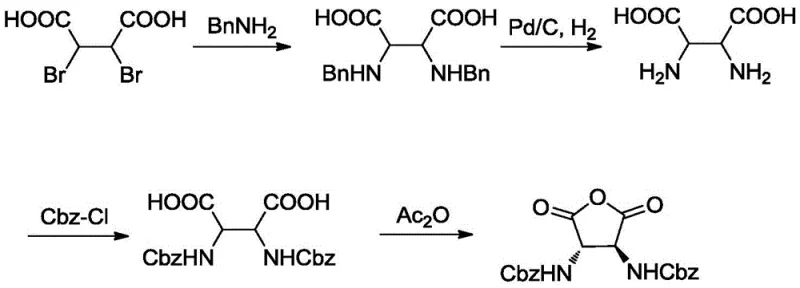
The reliance on palladium catalysts introduces multiple layers of complexity and cost. Not only is palladium a precious metal with fluctuating market prices, but its use also mandates rigorous heavy metal removal protocols to meet regulatory standards for pharmaceutical ingredients. The hydrogenation process itself requires specialized high-pressure equipment, increasing capital expenditure and safety risks associated with handling hydrogen gas. Additionally, the acetic acid-hydrochloric acid solvent systems often employed in these deprotection steps are corrosive and difficult to recycle, creating environmental burdens. These cumulative factors render the conventional method economically unviable for large-scale industrial applications, highlighting the urgent need for a more pragmatic and cost-effective synthetic strategy.
The Novel Approach
The inventive method disclosed in the patent overcomes these historical barriers through a clever modification of the amine component and the protection strategy. Instead of benzylamine, the process utilizes p-methoxybenzylamine (PMB) for the initial nucleophilic substitution. This seemingly small structural change has profound implications for process chemistry. The methoxy group enhances the solubility of the resulting intermediates in solvents like dichloromethane (DCM) and tetrahydrofuran (THF), allowing for smooth extraction and washing operations that were previously unattainable. More critically, the PMB group is acid-labile, meaning it can be removed using trifluoroacetic acid (TFA) rather than requiring catalytic hydrogenation. This substitution completely eliminates the need for expensive palladium reagents and high-pressure reactors, drastically simplifying the operational requirements.
Furthermore, the new route incorporates an early-stage esterification step, converting the starting dibromosuccinic acid into a diester before amination. This ensures that the molecule remains lipophilic and soluble throughout the early and middle stages of the synthesis. The subsequent steps—acylation with benzyl chloroformate, hydrolysis, and final cyclization—are optimized for mild conditions, typically operating between 0°C and 35°C. This gentle thermal profile minimizes the formation of thermal byproducts and racemization risks. By integrating these improvements, the novel approach achieves a synthesis that is not only chemically elegant but also industrially robust, offering a clear path toward cost reduction in chiral ligand manufacturing while maintaining the high purity standards required for downstream catalytic applications.
Mechanistic Insights into the Ester-Mediated Substitution and Cyclization
The core of this synthetic success lies in the mechanistic interplay between the ester functionality and the nucleophilic substitution dynamics. In the initial steps, racemic-2,3-dibromosuccinic acid is converted to its dimethyl or diethyl ester derivative (Compound I). This esterification serves a dual purpose: it activates the adjacent carbons for nucleophilic attack by increasing the electrophilicity of the alpha-carbons, and it masks the polar carboxylic acid groups that would otherwise hinder solubility. When p-methoxybenzylamine is introduced in the presence of a base like triethylamine, a double nucleophilic substitution occurs. The amine attacks the carbon-bromine bonds, displacing the bromide ions and forming the diamine diester (Compound II). The electron-donating methoxy group on the benzyl ring stabilizes the transition state and ensures that the resulting amine is sufficiently nucleophilic yet protected enough to prevent unwanted side reactions.
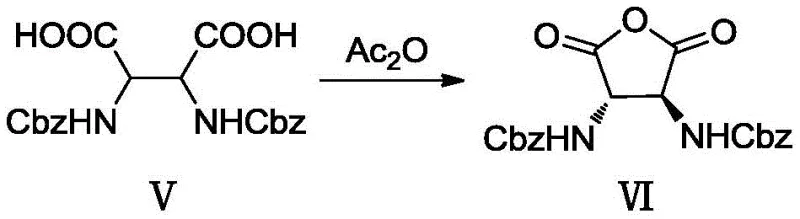
The subsequent deprotection and cyclization mechanisms are equally critical for ensuring product integrity. The removal of the PMB groups using trifluoroacetic acid proceeds via an acid-catalyzed cleavage mechanism, generating a stable carbocation intermediate that is trapped by the trifluoroacetate anion, releasing the free amine (Compound III) without affecting the ester groups. Following acylation with Cbz-Cl to install the final benzyl carbamate protections, the molecule undergoes hydrolysis to reveal the dicarboxylic acid (Compound V). The final step involves an intramolecular dehydration cyclization using acetic anhydride. Here, the two carboxylic acid groups react with the anhydride to form a mixed anhydride intermediate, which then undergoes nucleophilic attack by the adjacent oxygen or through a concerted mechanism to close the five-membered tetrahydrofuran ring, yielding the final anhydride product (Compound VI). This precise control over reaction pathways ensures high stereochemical fidelity and minimal impurity generation.
How to Synthesize (3S,4S)-2,5-dioxytetrahydrofuran-3,4-bis-benzyl carbamate Efficiently
Executing this synthesis requires careful attention to stoichiometry and temperature control, particularly during the exothermic esterification and acylation steps. The process is designed to be telescoped where possible, minimizing the isolation of unstable intermediates. The use of common solvents like DCM, methanol, and THF ensures that the process is compatible with standard glass-lined or stainless steel reactors found in most multipurpose chemical plants. Operators should monitor the reaction progress closely using TLC or HPLC, especially during the TFA deprotection step, to ensure complete removal of the PMB groups before proceeding to acylation. The detailed standardized synthesis steps, including specific molar ratios, addition rates, and workup procedures, are outlined below to guide process engineers in replicating this high-yielding route.
- Esterification of racemic-2,3-dibromosuccinic acid with alcohol under acidic catalysis to form diester compound (I).
- Nucleophilic substitution of compound (I) with p-methoxybenzylamine to yield diamine diester compound (II).
- Acid-mediated removal of the p-methoxybenzyl protecting groups using trifluoroacetic acid to obtain amino diester (III).
- Acylation of compound (III) with benzyl chloroformate to install Cbz protecting groups, forming compound (IV).
- Alkaline hydrolysis of the ester groups in compound (IV) to generate the dicarboxylic acid intermediate (V).
- Intramolecular dehydration cyclization of compound (V) using acetic anhydride to yield the final anhydride product (VI).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from the conventional palladium-based route to this novel ester-mediated pathway offers substantial strategic benefits. The most immediate impact is seen in the reduction of raw material costs and capital expenditure. By eliminating the requirement for palladium on carbon, companies can avoid the volatility associated with precious metal pricing and the complex logistics of catalyst recovery and recycling. Moreover, the removal of high-pressure hydrogenation steps reduces the need for specialized reactor infrastructure, allowing production to be shifted to more versatile, lower-cost manufacturing suites. This flexibility enhances supply chain resilience, as the process is less dependent on single-source equipment vendors or specialized hazardous operation permits.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven by the simplification of the workflow and the elimination of high-cost reagents. Without the need for palladium catalysts, the direct material cost per kilogram of the final product is significantly lowered. Additionally, the improved solubility of intermediates translates to higher recovery rates during extraction and crystallization, reducing waste and maximizing yield. The use of trifluoroacetic acid for deprotection, while a specialized reagent, is generally more cost-effective and easier to handle on a large scale than managing high-pressure hydrogen gas and spent metal catalysts. These factors combine to create a manufacturing process with a markedly lower cost of goods sold (COGS), providing a competitive edge in the marketplace.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the scarcity of specialized reagents or the bottleneck of complex purification steps. This new method mitigates those risks by relying on commodity chemicals such as dibromosuccinic acid, methanol, and p-methoxybenzylamine, all of which are widely available from multiple global suppliers. The robustness of the reaction conditions means that batch-to-batch variability is minimized, ensuring consistent quality and delivery schedules. Furthermore, the simplified workup procedures reduce the turnaround time between batches, allowing for faster throughput and the ability to respond more agilely to fluctuations in market demand for chiral ligands and their downstream derivatives.
- Scalability and Environmental Compliance: Scaling chemical processes often exposes hidden inefficiencies, but this route is inherently designed for expansion. The liquid-phase nature of the intermediates facilitates pumping and transfer in large-scale reactors, avoiding the clogging and filtration issues that plague solid-heavy processes. From an environmental perspective, the avoidance of heavy metals aligns with increasingly stringent green chemistry regulations and corporate sustainability goals. The waste streams generated are primarily organic salts and solvents, which are easier to treat and recycle compared to heavy metal-contaminated waste. This compliance readiness reduces the regulatory burden and potential liability for manufacturers, making the process not just chemically superior but also environmentally sustainable for long-term operation.
Frequently Asked Questions (FAQ)
Understanding the technical specifics of this synthesis is vital for stakeholders evaluating its adoption. The following questions address common inquiries regarding the practical implementation and benefits of this patented method. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this technology outperforms legacy methods in terms of efficiency, cost, and operational safety.
Q: Why is the new synthesis route superior to the conventional palladium-catalyzed method?
A: The conventional method relies on benzylamine which results in intermediates with poor solubility, complicating extraction and purification. Furthermore, it requires expensive palladium on carbon (Pd/C) for hydrogenolysis. The new route utilizes p-methoxybenzylamine, which offers superior solubility and allows for acid-mediated deprotection using trifluoroacetic acid, completely eliminating the need for precious metal catalysts and high-pressure hydrogenation equipment.
Q: What are the key raw materials required for this chiral ligand intermediate?
A: The synthesis begins with racemic-2,3-dibromosuccinic acid, a readily available commodity chemical. Other key reagents include methanol or ethanol for esterification, p-methoxybenzylamine for nucleophilic substitution, trifluoroacetic acid for deprotection, and benzyl chloroformate for the final protection steps. All materials are commercially accessible for industrial scale-up.
Q: How does this process address impurity control and yield optimization?
A: By converting the initial dibromo acid into a diester, the process significantly improves the solubility of intermediates in organic solvents like DCM and THF. This facilitates efficient liquid-liquid extraction and washing steps, removing inorganic salts and side products more effectively than the precipitation-heavy conventional route. The mild reaction conditions (e.g., 20-25°C for substitution) also minimize thermal degradation and side reactions, leading to consistently high yields across all six steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (3S,4S)-2,5-dioxytetrahydrofuran-3,4-bis-benzyl carbamate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced chiral ligands requires more than just a patent; it demands engineering excellence and unwavering commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial plant is seamless. We understand the critical nature of stringent purity specifications in the pharmaceutical sector, which is why our facilities are equipped with rigorous QC labs capable of detecting trace impurities and verifying stereochemical integrity at every stage of the manufacturing process. Our expertise allows us to optimize the esterification and cyclization steps described in CN111205251B to achieve maximum efficiency and yield.
We invite potential partners to engage with us to explore how this innovative synthesis route can be integrated into your supply chain. Whether you require custom synthesis services or bulk supply of this critical intermediate, our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can drive value and reliability for your chiral catalyst projects.
