Advanced Synthesis of Optically Pure Chiral Y6 Acceptors for High-Efficiency Organic Solar Cells
Advanced Synthesis of Optically Pure Chiral Y6 Acceptors for High-Efficiency Organic Solar Cells
The rapid evolution of organic photovoltaics (OPV) demands materials that push the boundaries of power conversion efficiency (PCE) while maintaining manufacturability. Patent CN113173937A introduces a groundbreaking advancement in this field by disclosing a method for synthesizing non-fullerene acceptor materials based on chiral alkane chains. Unlike conventional Y6 molecules which exist as a statistical mixture of stereoisomers, this innovation enables the production of optically pure variants, specifically the (S,S)-Y6 configuration. This structural precision addresses the inherent disorder found in racemic mixtures, resulting in superior film morphology and device performance. The patent details a comprehensive synthetic pathway that transforms simple precursors into high-performance electron acceptors, offering a viable route for next-generation solar energy applications.
For procurement specialists and supply chain managers seeking a reliable non-fullerene acceptor supplier, this technology represents a significant opportunity. By controlling the stereochemistry of the alkyl side chains, manufacturers can achieve consistent batch-to-batch quality, a critical factor in the commercialization of organic solar modules. The ability to synthesize these materials without resorting to expensive post-synthesis chiral separation processes implies a streamlined production workflow. This report analyzes the technical depth of patent CN113173937A, evaluating its potential for cost reduction in organic solar cell manufacturing and its readiness for commercial scale-up of complex photovoltaic materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
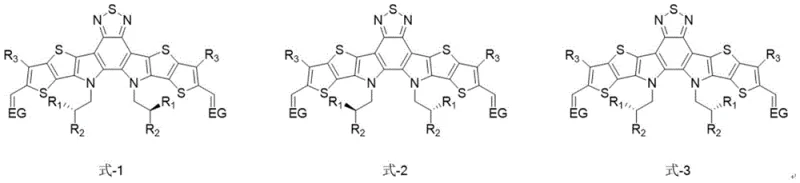
Traditional synthesis of the benchmark Y6 acceptor typically utilizes 2-ethylhexyl chains, which possess a chiral center. In standard chemical synthesis without chiral induction, this results in a product that is a complex mixture of three distinct stereoisomers: the (R,R) enantiomer, the (S,S) enantiomer, and the meso (R,S) compound. This stereochemical heterogeneity introduces disorder into the solid-state packing of the material. Such disorder can act as a trap for charge carriers, limiting the short-circuit current density (Jsc) and overall fill factor of the resulting solar cell devices. Furthermore, the presence of multiple isomers complicates the structure-property relationship studies, making it difficult for R&D teams to optimize device architecture precisely. The inability to isolate a single, pure isomer efficiently has historically capped the efficiency potential of these materials, with standard Y6 mixtures typically hovering around 16% PCE in optimized systems.
The Novel Approach
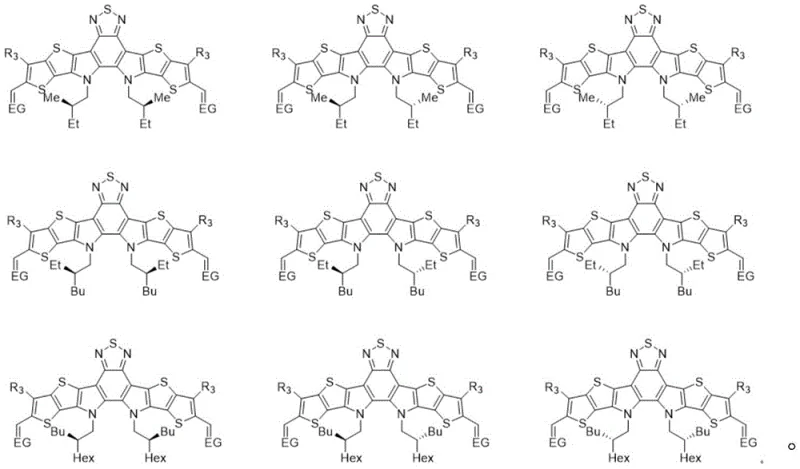
The methodology outlined in CN113173937A circumvents these issues by employing optically pure chiral bromoalkanes as the alkylating agents. Instead of accepting a mixture, the synthesis deliberately installs specific chiral configurations, such as the (S,S) configuration, onto the fused ring core. This approach ensures that every molecule in the final batch possesses the exact same three-dimensional geometry. The result is a highly ordered molecular arrangement in the thin film, which facilitates more efficient charge transport and reduces recombination losses. As demonstrated in the patent examples, the (S,S)-Y6 variant achieves a PCE of 16.5%, surpassing the performance of the racemic mixture. This improvement is not merely incremental; it validates the hypothesis that stereochemical purity is a critical lever for unlocking higher efficiencies in organic electronics.
Mechanistic Insights into Chiral Alkylation and Fused Ring Construction
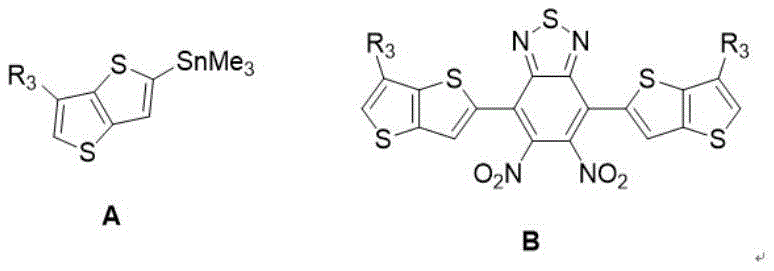
The synthetic route described in the patent is a masterpiece of modern organic synthesis, relying on a sequence of robust carbon-carbon and carbon-heteroatom bond-forming reactions. The process begins with the construction of the electron-deficient core. A Stille coupling reaction is employed to attach thienothiophene units to a 4,7-dibromo-5,6-dinitrobenzothiadiazole central scaffold. This step is crucial for establishing the conjugated backbone that will eventually facilitate electron delocalization. Following this, a thermal cyclization reaction using triphenylphosphine in o-dichlorobenzene closes the rings to form the rigid, planar fused structure characteristic of Y6 derivatives. This core formation sets the stage for the critical differentiation step: the introduction of chirality.
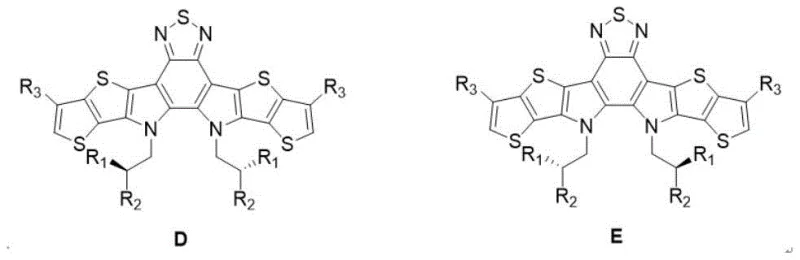
The defining feature of this patent is the nucleophilic substitution step where the nitrogen atoms of the fused core are alkylated. Rather than using racemic 2-bromo-octane or similar mixtures, the process utilizes optically pure halogenated alkanes. Under alkaline conditions, typically using potassium carbonate in dimethyl sulfoxide (DMSO), the chiral alkyl group is transferred to the nitrogen with retention of configuration. This step effectively locks the stereochemistry into the molecule early in the synthesis. Subsequent steps involve the Vilsmeier-Haack reaction to install aldehyde functionalities at the terminal positions, followed by a Knoevenagel condensation with strong electron-withdrawing end groups (such as IC-F). This final condensation extends the conjugation and lowers the LUMO energy level, optimizing the material for electron acceptance.
Impurity control is inherently built into this design. By starting with high-purity chiral alkylating agents, the formation of diastereomeric impurities is prevented at the source. In traditional routes, separating the (R,R), (S,S), and (R,S) isomers of the final large molecule would require preparative chiral HPLC, a technique that is prohibitively expensive and low-yielding on an industrial scale. By contrast, this method shifts the burden of purity to the smaller, more manageable alkyl halide starting materials, which are easier to purify or source commercially. This strategic shift in synthetic design significantly simplifies the downstream purification of the final acceptor material, ensuring a cleaner product profile with fewer batch-to-batch variations.
How to Synthesize Optically Pure Chiral Y6 Efficiently
The synthesis of these advanced materials requires precise control over reaction conditions to maintain the integrity of the chiral centers and the conjugated system. The patent provides a detailed roadmap that balances yield with stereochemical fidelity. From the initial coupling to the final condensation, each step has been optimized to minimize side reactions and maximize throughput. For R&D teams looking to replicate or license this technology, understanding the nuances of the alkylation and condensation steps is paramount. The following guide outlines the standardized operational framework derived from the patent disclosure, serving as a foundation for process development.
- Perform Stille coupling between 4,7-dibromo-5,6-dinitrobenzothiadiazole and stannyl-thienothiophene to form the nitro-intermediate.
- Execute condensation ring-closing reaction using triphenylphosphine in o-dichlorobenzene to form the fused benzothiadiazole core.
- Conduct nucleophilic substitution with optically pure chiral bromoalkane (e.g., S-bromoisooctane) under alkaline conditions to introduce chirality.
- Apply Vilsmeier-Haack reaction to introduce aldehyde groups at the terminal positions of the fused ring system.
- Finalize synthesis via Knoevenagel condensation with electron-withdrawing end groups (e.g., IC-F) to yield the final acceptor material.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders focused on the bottom line and supply security, the transition to this chiral synthesis route offers compelling economic arguments. The primary driver for cost reduction in organic solar cell manufacturing lies in the elimination of complex separation processes. Traditional methods that produce isomeric mixtures often suffer from yield penalties when attempting to enrich specific isomers or deal with inconsistent material properties. By adopting a route that delivers a single, defined isomer directly, manufacturers can streamline their purification workflows. This reduction in processing steps translates directly to lower solvent consumption, reduced energy usage, and decreased labor costs per kilogram of produced material.
- Cost Reduction in Manufacturing: The synthetic strategy leverages readily available precursors and avoids the need for expensive chiral catalysts or resolution agents in the final stages. The use of standard reagents like potassium carbonate and common solvents such as DMSO and chloroform ensures that raw material costs remain stable and predictable. Furthermore, the high yields reported for intermediate steps (e.g., 90% for the Stille coupling and 85% for the alkylation) indicate a material-efficient process. This efficiency minimizes waste generation, which is a significant cost factor in the disposal of chemical byproducts in compliant facilities.
- Enhanced Supply Chain Reliability: Sourcing optically pure alkyl halides is generally more straightforward than sourcing custom-synthesized chiral large molecules. This decouples the supply risk from the complexity of the final API-like structure. By modularizing the synthesis, supply chain managers can secure the chiral building blocks from multiple vendors, reducing the risk of single-source bottlenecks. Additionally, the robustness of the reaction conditions—such as the moderate temperatures used in the final Knoevenagel condensation (60-70°C)—suggests that the process is tolerant of minor fluctuations, enhancing the reliability of production schedules and reducing lead time for high-purity organic semiconductors.
- Scalability and Environmental Compliance: The patent emphasizes mild reaction conditions which are inherently safer and easier to scale. The avoidance of extreme cryogenic temperatures or high-pressure reactors simplifies the engineering requirements for scaling from gram to ton quantities. Moreover, the improved performance of the resulting material means that less active material may be required per watt of generated power, indirectly reducing the environmental footprint of the solar module manufacturing. The process aligns well with green chemistry principles by maximizing atom economy in the coupling steps and minimizing the need for resource-intensive purification techniques.
Frequently Asked Questions (FAQ)
Understanding the technical specifics of this new material class is essential for making informed procurement decisions. The following questions address common inquiries regarding the performance, stability, and integration of these chiral acceptors into existing manufacturing lines. The answers are derived directly from the experimental data and technical disclosures within patent CN113173937A, providing a factual basis for evaluation.
Q: What is the primary advantage of using optically pure chiral alkane chains in Y6 acceptors compared to traditional mixtures?
A: Traditional Y6 contains a mixture of (R,R), (S,S), and meso (R,S) isomers due to the chiral center in the 2-ethylhexyl chain. Using optically pure chains, specifically the (S,S) configuration, eliminates structural disorder, leading to improved molecular packing, higher short-circuit current, and a photoelectric conversion efficiency increase from ~16% to 16.5%.
Q: Is the synthesis of chiral Y6 scalable for industrial production?
A: Yes, the patent describes a robust five-step route using standard organic reactions like Stille coupling and Knoevenagel condensation. The conditions are mild (e.g., 60-70°C for the final step), and intermediate yields are high (80-90%), indicating strong potential for commercial scale-up without requiring complex chiral separation techniques on the final product.
Q: How does this method reduce manufacturing costs for organic solar cell materials?
A: Cost reduction is achieved by introducing chirality at the alkylation stage using commercially available or easily synthesized optically pure bromoalkanes, rather than attempting to separate enantiomers of the large, complex final Y6 molecule. This simplifies purification and avoids the significant yield losses associated with chiral chromatography of the final acceptor.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Y6 Acceptor Supplier
The transition from laboratory curiosity to commercial reality requires a partner with deep technical expertise and proven scaling capabilities. NINGBO INNO PHARMCHEM stands at the forefront of fine chemical manufacturing, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with rigorous QC labs capable of verifying the optical purity and electronic properties of complex photovoltaic materials like SS-Y6. We understand that stringent purity specifications are non-negotiable for high-efficiency OPV devices, and our quality management systems are designed to meet these exacting standards consistently.
We invite industry leaders to collaborate with us to unlock the full potential of chiral non-fullerene acceptors. Whether you require custom synthesis of specific chiral variants or large-scale supply of standardized materials, our team is ready to assist. Contact our technical procurement team today to request a Customized Cost-Saving Analysis. We encourage you to reach out for specific COA data and route feasibility assessments tailored to your unique device architecture and production requirements.
