Advanced Asymmetric Synthesis of Brivaracetam Methyl Ester Impurity for Global Quality Control
Advanced Asymmetric Synthesis of Brivaracetam Methyl Ester Impurity for Global Quality Control
The pharmaceutical industry faces increasing regulatory pressure to identify and quantify trace impurities in active pharmaceutical ingredients (APIs), particularly for potent antiepileptic drugs like Brivaracetam. Patent CN115557873A introduces a groundbreaking synthetic methodology for producing the specific (S,R)-configured methyl ester impurity of Brivaracetam, a critical reference standard required for rigorous quality control. Unlike traditional methods that rely on inefficient chiral separation techniques, this novel approach utilizes a direct asymmetric synthesis strategy starting from commercially available chiral building blocks. By employing a sequence of ring-opening, acylation, condensation-cyclization, and esterification reactions, the process achieves high stereochemical fidelity and exceptional purity levels exceeding 99%. This technological advancement represents a significant leap forward for reliable pharmaceutical intermediate supplier networks, enabling the consistent production of hard-to-find impurity standards essential for validating the safety and efficacy of generic and branded Brivaracetam formulations globally.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of specific stereoisomeric impurities such as the (S,R)-Brivaracetam methyl ester has been fraught with significant technical and economic challenges. The prevailing industry standard involved the use of preparative chiral high-performance liquid chromatography (HPLC) to separate the desired impurity from a racemic mixture or crude reaction byproducts. This technique is inherently inefficient for large-scale production due to its low throughput, massive solvent consumption, and the exorbitant cost of chiral stationary phases. Furthermore, chiral separation often results in substantial material loss, yielding only small quantities of the target compound, which drives up the unit cost dramatically. For procurement managers and supply chain directors, relying on such methods creates a bottleneck, leading to long lead times and potential supply discontinuities for critical quality control materials. The inability to scale these separation processes effectively means that manufacturers often struggle to secure sufficient quantities of high-purity impurity standards needed for comprehensive method validation and stability studies.
The Novel Approach
The methodology disclosed in patent CN115557873A fundamentally disrupts this status quo by shifting from a separation-based paradigm to a constructive asymmetric synthesis model. This innovative route leverages the inherent chirality of affordable starting materials, specifically (R)-4-propyldihydrofuran-2-one and (S)-2-aminobutyramide, to construct the target molecule with precise stereocontrol. By designing a linear synthesis that preserves the chiral integrity of the precursors through mild reaction conditions, the process eliminates the need for expensive chiral resolution steps entirely. The reaction sequence is optimized for operational simplicity, utilizing common reagents like thionyl chloride and potassium hydroxide in standard solvents such as dichloromethane. This strategic shift not only drastically simplifies the downstream processing but also aligns perfectly with the goals of cost reduction in API manufacturing support sectors. The result is a robust, scalable protocol that delivers the target impurity in high yields and purity, making it an ideal solution for industrial-scale production of reference standards.
Mechanistic Insights into Asymmetric Coupling and Cyclization
The core chemical innovation of this synthesis lies in the tandem condensation and cyclization step, which constructs the pivotal pyrrolidinone ring while establishing the critical nitrogen-carbon bond. The process begins with the activation of the chiral acid intermediate into a highly reactive acid chloride using thionyl chloride. This activated species is then introduced to a reaction vessel containing (S)-2-aminobutyramide and powdered potassium hydroxide at cryogenic temperatures ranging from -25°C to -15°C. The use of solid KOH acts as both a base to scavenge the generated HCl and a heterogeneous catalyst surface that may facilitate the nucleophilic attack of the amine on the acid chloride. This low-temperature environment is crucial for suppressing racemization at the alpha-carbon of the butyramide moiety, ensuring the retention of the (S)-configuration. Following the initial amide bond formation, the proximal amide nitrogen attacks the ketone or activated carbonyl within the chain (depending on the specific tautomeric state or intermediate activation), triggering an intramolecular cyclization that closes the five-membered lactam ring. This elegant cascade reaction efficiently builds the complex bicyclic-like topology of the Brivaracetam scaffold in a single operational phase.
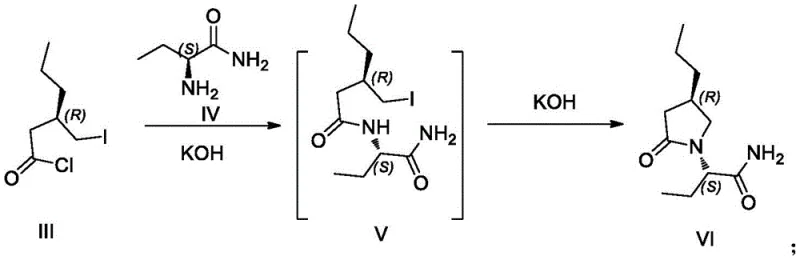
Following the formation of the cyclic lactam intermediate, the synthesis proceeds through a controlled hydrolysis and re-esterification sequence to install the methyl ester functionality characteristic of the target impurity. The hydrolysis step utilizes aqueous hydrochloric acid under heated conditions to cleave the primary amide group of the side chain, converting it into a carboxylic acid without disturbing the sensitive lactam ring. This selectivity is achieved by carefully tuning the pH and temperature, exploiting the difference in hydrolytic stability between the acyclic amide and the cyclic imide structure. The resulting free acid is then subjected to a second activation with thionyl chloride, regenerating the acid chloride transiently, which is immediately quenched with methanol. This final esterification step is rapid and high-yielding, driven by the irreversible formation of the methyl ester. The entire mechanistic pathway demonstrates a sophisticated understanding of chemoselectivity, allowing for the manipulation of multiple functional groups in the presence of sensitive chiral centers. This level of control is paramount for producing high-purity pharmaceutical intermediates that meet the stringent specifications required by global regulatory bodies for impurity profiling.
How to Synthesize Brivaracetam Methyl Ester Impurity Efficiently
The practical execution of this synthesis requires careful attention to temperature control and reagent stoichiometry to maximize yield and optical purity. The process is divided into five distinct operational stages, beginning with the ring-opening of the starting lactone and concluding with the final methyl ester purification. Each step has been optimized to minimize side reactions and facilitate easy workup procedures, such as simple phase separations and crystallizations. The detailed protocol below outlines the critical parameters for each transformation, providing a roadmap for laboratory and pilot-scale implementation. For a complete breakdown of the standardized operating procedures, please refer to the structured guide below.
- Perform ring-opening of 4-propyldihydrofuran-2-one using trimethyliodosilane (TMSI) in dichloromethane at 0-5°C to generate the iodo-acid intermediate.
- Convert the acid intermediate to its acid chloride using thionyl chloride, then couple with (S)-2-aminobutyramide under basic conditions (KOH) at low temperature (-25°C) to induce cyclization.
- Hydrolyze the resulting lactam amide to the free acid using aqueous HCl, followed by re-esterification via acid chloride formation and methanol treatment to yield the final methyl ester impurity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers transformative benefits that extend far beyond simple technical feasibility. By replacing resource-intensive chiral separation with efficient chemical synthesis, the overall cost structure of producing this critical impurity standard is significantly optimized. The reliance on bulk commodity chemicals like thionyl chloride, methanol, and dichloromethane, combined with readily available chiral pool starting materials, insulates the supply chain from the volatility associated with specialized chromatographic media. This shift ensures a more predictable and stable supply of quality control materials, which is essential for maintaining uninterrupted manufacturing operations of the parent drug. Furthermore, the simplified downstream processing reduces the environmental footprint and waste generation, aligning with modern sustainability goals in chemical manufacturing.
- Cost Reduction in Manufacturing: The elimination of preparative chiral HPLC removes one of the most expensive unit operations from the production workflow. Chiral columns have limited lifespans and require costly mobile phases, whereas this chemical synthesis utilizes standard reactors and inexpensive reagents. The qualitative reduction in operational complexity translates directly into lower production costs per gram of the final impurity standard. Additionally, the high yield reported in the patent examples suggests minimal raw material waste, further enhancing the economic efficiency of the process. This cost structure makes it feasible to produce larger batches of the impurity, allowing for better inventory management and economies of scale for suppliers.
- Enhanced Supply Chain Reliability: Sourcing chiral starting materials like 4-propyldihydrofuran-2-one is generally more reliable than securing capacity on specialized chiral separation equipment, which is often a bottleneck in the industry. The synthetic route described is robust and uses common chemical transformations that can be performed in a wide range of multipurpose chemical facilities. This flexibility reduces the risk of supply disruptions caused by equipment maintenance or capacity constraints at specialized CDMOs. By decentralizing the production capability to standard synthetic labs, the supply chain becomes more resilient and responsive to fluctuating demand for Brivaracetam quality control standards.
- Scalability and Environmental Compliance: The process is designed with commercial scale-up of complex organic molecules in mind, utilizing reaction conditions that are easily transferable from kilogram to multi-ton scales. The use of dichloromethane, while requiring proper handling, is well-understood in industrial settings with established recovery and recycling protocols. The avoidance of heavy metal catalysts or exotic reagents simplifies waste treatment and regulatory compliance regarding residual impurities. The straightforward crystallization and filtration steps described in the patent facilitate the isolation of high-purity solids without the need for complex chromatographic purification, making the process inherently greener and more suitable for large-scale industrial application.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of Brivaracetam impurities. These insights are derived directly from the technical specifications and experimental data provided in the patent literature, ensuring accuracy and relevance for industry professionals. Understanding these details is crucial for integrating this new supply source into your quality assurance workflows.
Q: Why is the asymmetric synthesis of Brivaracetam impurities preferred over chiral separation?
A: Conventional chiral column separation is costly and difficult to scale due to low throughput and high solvent consumption. Asymmetric synthesis from chiral starting materials offers a more direct, scalable, and cost-effective route for producing high-purity reference standards required for regulatory compliance.
Q: What are the critical reaction conditions for the coupling step in this synthesis?
A: The coupling between the acid chloride intermediate and (S)-2-aminobutyramide requires strict temperature control between -25°C and -15°C using powdered potassium hydroxide in dichloromethane. This ensures high stereoselectivity and prevents racemization or side reactions during the simultaneous amide formation and intramolecular cyclization.
Q: How does this method impact the supply chain for antiepileptic drug manufacturers?
A: By utilizing readily available and inexpensive starting materials like 4-propyldihydrofuran-2-one and avoiding expensive chiral chromatography, this method significantly reduces production costs and lead times. This enhances supply chain reliability for manufacturers needing consistent batches of impurity standards for quality assurance testing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Brivaracetam Methyl Ester Impurity Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality impurity standards play in the development and regulation of life-saving medications like Brivaracetam. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We adhere to stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to verify the identity and stereochemical purity of every batch. Our commitment to technical excellence ensures that the reference materials we provide are fully compliant with international pharmacopoeial standards, giving you confidence in your analytical results.
We invite you to collaborate with us to optimize your supply chain for Brivaracetam impurity standards. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume needs, demonstrating how our streamlined synthesis can reduce your overall procurement expenses. Contact us today to request specific COA data and route feasibility assessments, and let us partner with you to ensure the highest quality and reliability in your pharmaceutical manufacturing operations.
