Advanced Synthetic Route for Tolvaptan Degradation Derivative Manufacturing and Scale-Up
Advanced Synthetic Route for Tolvaptan Degradation Derivative Manufacturing and Scale-Up
The pharmaceutical industry's relentless pursuit of quality control has elevated the importance of precise impurity profiling, particularly for complex small molecules like Tolvaptan. As detailed in patent CN113264885A, a groundbreaking synthetic methodology has been established for the preparation of a specific Tolvaptan degradation derivative, serving as a critical reference standard for analytical validation. This novel approach transforms a readily available benzazepine precursor into the target molecule through a meticulously designed five-step cascade, addressing the historical challenges of obtaining high-purity degradation products. By leveraging robust organic transformations including acid-catalyzed dehydration, selective epoxidation, and reductive desulfonylation, this route offers a reliable solution for pharmaceutical manufacturers seeking to enhance their quality assurance protocols. The strategic design not only ensures structural fidelity but also optimizes the economic viability of producing these essential analytical standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the acquisition of specific drug degradation products for analytical method development has been fraught with significant logistical and technical hurdles. Conventional methods often rely on forced degradation studies of the active pharmaceutical ingredient (API) itself, a process that frequently yields complex mixtures of impurities that are difficult to separate and characterize. This lack of specificity can lead to ambiguous analytical results, complicating the regulatory approval process and potentially delaying market entry for generic formulations. Furthermore, isolating trace degradation products from bulk API batches is inherently inefficient, often resulting in negligible yields that are insufficient for comprehensive toxicological assessment or method validation. The reliance on unpredictable degradation pathways also introduces variability, making it difficult to establish consistent quality control benchmarks across different production batches.
The Novel Approach
In stark contrast, the synthetic route disclosed in the patent data presents a deterministic and highly efficient pathway to the target degradation derivative. By starting from a defined intermediate (Compound I) and proceeding through discrete, high-yielding chemical transformations, this method bypasses the randomness of spontaneous degradation. The process utilizes a logical sequence of functional group manipulations—dehydration, oxidation, reduction, methylation, and cleavage—that allows for precise control over the molecular architecture at every stage. This level of control ensures that the final product is chemically identical to the degradation impurity found in the drug substance, providing a gold-standard reference for HPLC and MS analysis. The ability to synthesize this derivative on demand eliminates supply chain bottlenecks associated with sourcing rare impurities and empowers quality control laboratories to validate their methods with confidence.
Mechanistic Insights into the Multi-Step Cascade Synthesis
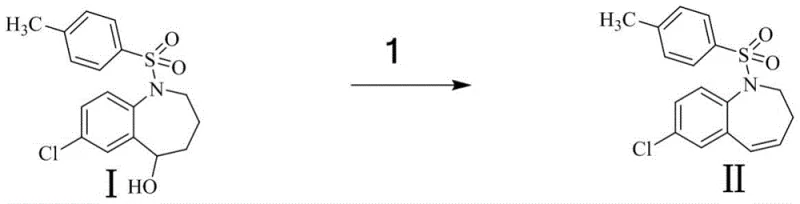
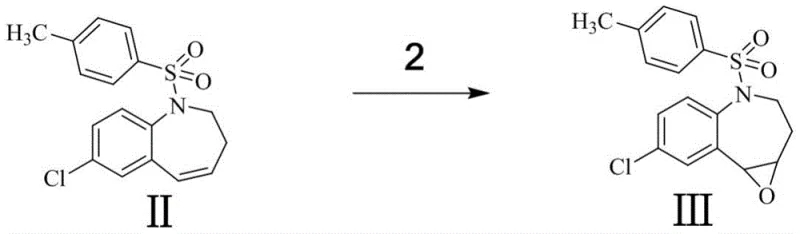
The core of this synthetic innovation lies in its elegant manipulation of the benzazepine scaffold. The journey begins with an acid-catalyzed dehydration, where the hydroxyl group of the starting material is protonated, facilitating the elimination of water to generate a conjugated alkene system (Compound II). This step is critical for setting up the subsequent oxidation, as the electron-rich double bond serves as the nucleophile for the epoxidation reaction. Following this, the alkene undergoes a Prilezhaev-type oxidation using hydrogen peroxide, where the peracid species transfers an oxygen atom to the double bond to form the strained epoxide ring (Compound III). This transformation is highly regioselective, driven by the electronic environment of the seven-membered ring, ensuring that the oxygen is installed at the precise location required for the final structure.
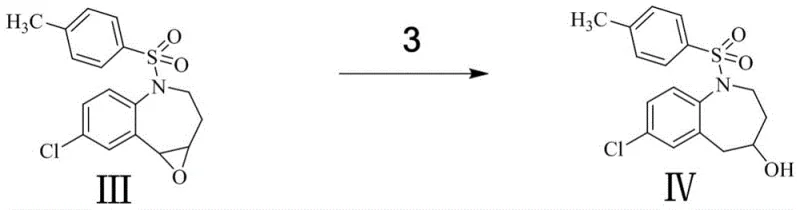
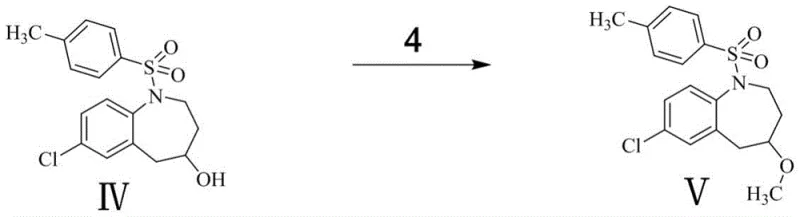
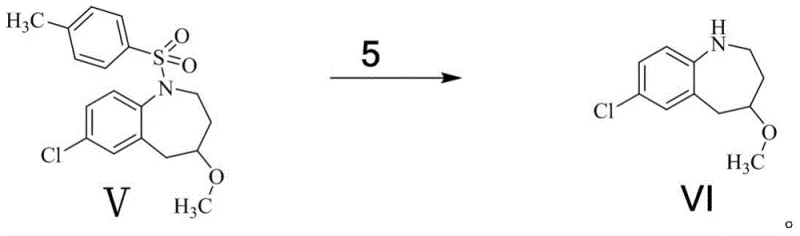
Subsequent steps involve the careful opening of the epoxide ring and functionalization of the resulting alcohol. The reduction of the epoxide with sodium borohydride proceeds via a hydride attack, regenerating the hydroxyl functionality but with altered stereochemistry or positioning relative to the nitrogen atom (Compound IV). This alcohol is then subjected to Williamson ether synthesis conditions, where deprotonation by a strong base like potassium tert-butoxide creates an alkoxide nucleophile that attacks iodomethane to install the methyl ether group (Compound V). The final and perhaps most chemically interesting step is the reductive cleavage of the p-toluenesulfonyl (tosyl) protecting group. Using magnesium chips in methanol, a single-electron transfer mechanism likely facilitates the breaking of the nitrogen-sulfur bond, liberating the secondary amine and yielding the final target molecule (Compound VI) without affecting the newly formed ether linkage.
How to Synthesize Tolvaptan Degradation Derivative Efficiently
Executing this synthesis requires strict adherence to the optimized reaction parameters identified during the patent's development phase to ensure maximum yield and purity. The process is designed to be operationally simple, utilizing standard laboratory equipment and common organic solvents, which facilitates easy translation from bench scale to pilot plant operations. Each step has been fine-tuned regarding temperature, molar ratios, and workup procedures to minimize side reactions and simplify purification. For R&D teams looking to implement this route, the following guide outlines the critical operational phases derived from the patent examples, ensuring reproducibility and safety throughout the manufacturing campaign.
- Dehydrate starting material Compound I using concentrated sulfuric acid in toluene at 110°C to form alkene Compound II.
- Oxidize Compound II with hydrogen peroxide in methanol to generate epoxy intermediate Compound III.
- Reduce the epoxide ring of Compound III using sodium borohydride in THF to yield alcohol Compound IV.
- Methylate Compound IV with iodomethane and potassium tert-butoxide to form ether Compound V.
- Perform reductive cleavage of the tosyl group in Compound V using magnesium chips in methanol to obtain target Compound VI.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this synthetic route offers compelling advantages that directly impact the bottom line and operational resilience. The strategic selection of reagents avoids the use of precious metal catalysts or exotic ligands, which are often subject to volatile pricing and geopolitical supply constraints. Instead, the process relies on bulk commodity chemicals such as concentrated sulfuric acid, hydrogen peroxide, and magnesium chips, ensuring a stable and cost-effective supply chain. This fundamental shift in reagent selection significantly reduces the raw material cost profile, making the production of this high-value reference standard economically sustainable even at smaller batch sizes typically required for analytical standards.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of straightforward aqueous workups drastically lower the operational expenditure (OPEX) associated with production. By avoiding complex chromatographic separations in favor of crystallization and simple extractions where possible, the process reduces solvent consumption and waste disposal costs. Furthermore, the high yields reported in the patent examples (often exceeding 85-90% per step) minimize material loss, ensuring that the theoretical cost per gram of the final product remains highly competitive compared to traditional isolation methods.
- Enhanced Supply Chain Reliability: The reliance on universally available solvents like toluene, methanol, and THF mitigates the risk of production stoppages due to solvent shortages. Since the reaction conditions do not require specialized high-pressure reactors or cryogenic temperatures (beyond standard ice baths), the process can be manufactured in a wide range of multipurpose chemical facilities. This flexibility allows for diversified sourcing strategies, enabling procurement managers to qualify multiple contract manufacturing organizations (CMOs) without requiring significant capital investment in specialized infrastructure.
- Scalability and Environmental Compliance: The synthetic pathway is inherently scalable, with linear progression from gram to kilogram scales demonstrated through the optimization of molar ratios. The waste stream is primarily composed of benign inorganic salts and recoverable organic solvents, simplifying environmental compliance and wastewater treatment. The absence of heavy metal residues in the final product also streamlines the quality control release process, as extensive testing for residual catalysts is rendered unnecessary, accelerating the time-to-market for the reference standard.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of this tolvaptan degradation derivative. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity for stakeholders evaluating this technology for adoption. Understanding these nuances is crucial for integrating this intermediate into your broader quality control and supply chain frameworks.
Q: What is the primary advantage of this synthetic route for Tolvaptan derivatives?
A: The primary advantage is the use of inexpensive, commodity reagents like sulfuric acid and magnesium chips, avoiding expensive transition metal catalysts while achieving yields over 87% in key steps.
Q: How is the purity of the final degradation derivative controlled?
A: Purity is controlled through optimized reaction conditions, such as specific molar ratios of oxidants and reducing agents, followed by rigorous purification via column chromatography and crystallization, ensuring >99% purity.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process utilizes common solvents like toluene, methanol, and THF, and avoids hazardous high-pressure conditions, making it highly scalable for industrial manufacturing of pharmaceutical reference standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tolvaptan Degradation Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity reference standards play in ensuring patient safety and regulatory compliance. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency. We are committed to delivering materials that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify identity and potency.
We invite you to collaborate with us to optimize your supply chain for this critical intermediate. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can support your long-term strategic goals in pharmaceutical development.
