Advanced Synthesis of Beta-Trifluoromethyl Thiophosphates for Commercial Pharmaceutical Manufacturing
The landscape of organophosphorus chemistry is undergoing a significant transformation driven by the demand for fluorinated building blocks in modern drug discovery. Patent CN113264957A discloses a groundbreaking preparation method for beta-trifluoromethyl thiophosphate compounds, addressing critical limitations in current synthetic methodologies. This innovation leverages a copper-catalyzed multicomponent reaction involving olefins, elemental sulfur, hydrogen phosphites, and hypervalent iodine trifluoromethylating reagents. By enabling the simultaneous introduction of trifluoromethyl and thiophosphoryl groups under mild conditions, this technology opens new avenues for designing bioactive molecules with enhanced metabolic stability and lipophilicity. The strategic integration of these functional motifs is particularly valuable for developing next-generation pharmaceuticals and agrochemicals where precise modulation of physicochemical properties is paramount.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional strategies for synthesizing trifluoromethyl-substituted organophosphorus compounds have long been plagued by severe operational constraints and economic inefficiencies. Conventional routes often rely on pre-functionalized starting materials that are expensive, unstable, or difficult to source in bulk quantities, creating bottlenecks in the supply chain for key intermediates. Furthermore, many existing protocols require harsh reaction conditions, such as extreme temperatures or the use of strong bases, which can lead to poor chemoselectivity and the formation of complex impurity profiles that are costly to remove. The reliance on precious metal catalysts in some legacy methods further exacerbates cost issues and introduces regulatory hurdles regarding heavy metal residues in final active pharmaceutical ingredients. These factors collectively limit the structural diversity accessible to medicinal chemists and hinder the rapid scale-up required for commercial viability.
The Novel Approach
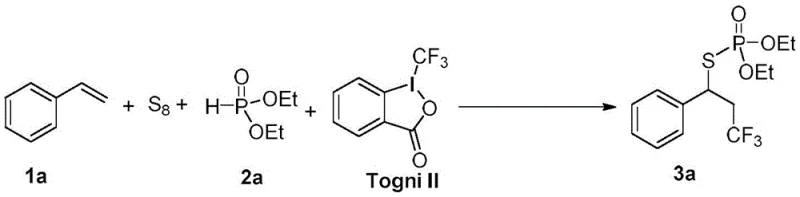
In stark contrast, the methodology described in the patent utilizes a robust and atom-economical strategy that directly functionalizes readily available olefins. By employing elemental sulfur powder and Togni reagents as key substrates, the process bypasses the need for complex pre-functionalization steps, significantly streamlining the synthetic sequence. The reaction proceeds efficiently at temperatures ranging from 0 to 100 degrees Celsius, often achieving optimal results at room temperature, which drastically reduces energy consumption and operational complexity. This approach not only expands the scope of compatible substrates to include a wide array of linear, branched, and cyclic olefins but also ensures high regioselectivity and yield. The use of earth-abundant copper catalysts further enhances the sustainability profile of the process, making it an attractive option for green chemistry initiatives in fine chemical manufacturing.
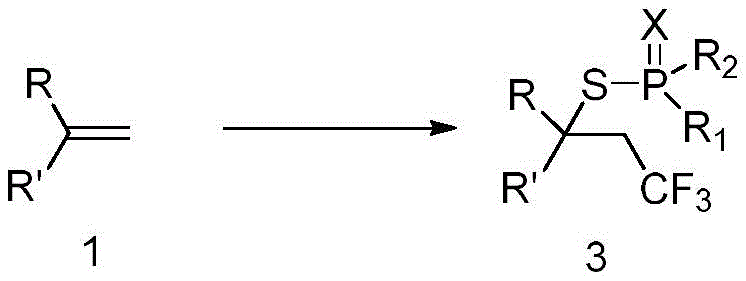
Mechanistic Insights into Copper-Catalyzed Trifluoromethylthiophosphorylation
The catalytic cycle likely initiates with the activation of the trifluoromethylating reagent by the copper catalyst, generating a reactive trifluoromethyl radical species that adds across the double bond of the olefin substrate. This radical addition step is crucial for establishing the carbon-carbon bond and setting the stereochemistry of the final product. Subsequent interception of the resulting carbon-centered radical by elemental sulfur leads to the formation of a thiyl radical or sulfide intermediate, which is then trapped by the phosphorus nucleophile. The presence of a base facilitates the deprotonation of the hydrogen phosphite, enhancing its nucleophilicity and driving the formation of the P-S bond. This cascade sequence effectively constructs two new bonds and two new stereocenters in a single operation, demonstrating remarkable efficiency and step economy compared to stepwise approaches.
Impurity control is inherently managed through the high selectivity of the radical propagation steps and the mild nature of the reaction conditions. The use of specific ligands, such as 1,10-phenanthroline or bipyridine derivatives, helps stabilize the copper center and modulate its redox potential, thereby suppressing side reactions like homocoupling or over-oxidation. The protocol specifies simple workup procedures involving extraction with ethyl acetate and drying over anhydrous sodium sulfate, which effectively removes inorganic salts and polar byproducts. Final purification via column chromatography using petroleum ether and ethyl acetate mixtures ensures the isolation of the target beta-trifluoromethyl thiophosphate with high purity, meeting the stringent specifications required for downstream applications in drug development.
How to Synthesize Beta-Trifluoromethyl Thiophosphate Efficiently
The practical implementation of this synthesis involves a straightforward one-pot procedure that minimizes handling risks and maximizes throughput. Operators begin by charging a reaction vessel with the copper catalyst, ligand, and sulfur powder in an organic solvent such as dichloromethane or acetonitrile under an inert atmosphere. After stirring to ensure homogeneity, the hydrogen phosphite and base are added, followed by the sequential introduction of the olefin and the Togni reagent. The reaction mixture is then allowed to stir at ambient temperature for a defined period, typically between 2 to 8 hours, before being quenched and processed. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction mixture by adding copper catalyst, ligand, sulfur powder, and solvent to a flask under inert atmosphere.
- Add hydrogen phosphite and base, stir at room temperature, then introduce the olefin substrate and Togni II reagent.
- React for 2-8 hours, extract with ethyl acetate, dry, and purify via column chromatography to obtain the target product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this technology offers substantial opportunities for cost optimization and supply chain resilience by utilizing commodity chemicals as primary feedstocks. The reliance on elemental sulfur and common olefins eliminates dependence on exotic or monopolized reagents, ensuring a stable and competitive pricing structure for raw materials. Moreover, the elimination of precious metal catalysts removes the need for expensive scavenging processes and reduces the regulatory burden associated with heavy metal limits in pharmaceutical products. This shift towards base metal catalysis aligns with global sustainability goals and reduces the overall environmental footprint of the manufacturing process.
- Cost Reduction in Manufacturing: The process achieves significant cost savings by replacing expensive transition metal catalysts with affordable copper salts and utilizing inexpensive sulfur powder as a sulfur source. The mild reaction conditions reduce energy costs associated with heating or cooling, while the high atom economy minimizes waste generation and disposal fees. Simplified purification protocols further lower operational expenditures by reducing solvent consumption and processing time, leading to a more economical production model for high-value intermediates.
- Enhanced Supply Chain Reliability: Sourcing risk is mitigated through the use of widely available starting materials like styrenes and diethyl phosphite, which are produced at scale by multiple global suppliers. The robustness of the reaction against moisture and air, facilitated by the inert gas protection, ensures consistent batch-to-batch quality and reduces the likelihood of production delays due to sensitive reagent degradation. This reliability is critical for maintaining continuous supply lines for long-term commercial projects and meeting just-in-time delivery requirements.
- Scalability and Environmental Compliance: The methodology is inherently scalable, having been demonstrated in multi-gram scales with consistent yields, indicating readiness for kilogram to tonne-level production. The use of less hazardous solvents and the generation of minimal toxic byproducts simplify waste management and ensure compliance with increasingly strict environmental regulations. The ability to tune the reaction parameters allows for easy adaptation to different reactor configurations, supporting flexible manufacturing strategies that can respond quickly to market demand fluctuations.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the scope, safety, and applicability of this novel trifluoromethylthiophosphorylation method. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation to assist technical teams in evaluating feasibility.
Q: What are the primary advantages of this new synthesis method over traditional organophosphorus routes?
A: This method utilizes mild reaction conditions (0-100°C) and inexpensive reagents like sulfur powder and Togni reagents, avoiding the harsh conditions and limited substrate scope often found in conventional phosphorylation techniques.
Q: Is this process suitable for large-scale industrial production of agrochemical intermediates?
A: Yes, the process features simple post-treatment procedures involving standard extraction and chromatography, along with high atom economy and selectivity, making it highly scalable for industrial manufacturing.
Q: What types of olefin substrates are compatible with this trifluoromethylthiophosphorylation reaction?
A: The reaction demonstrates broad substrate tolerance, successfully accommodating linear olefins, branched olefins, cyclic olefins, and various substituted styrenes including those with electron-withdrawing or donating groups.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Trifluoromethyl Thiophosphate Supplier
As a leader in custom synthesis and contract development, NINGBO INNO PHARMCHEM is uniquely positioned to leverage this advanced technology for your specific project needs. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to pilot plant is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee the quality of every batch delivered. Our commitment to excellence extends beyond mere synthesis; we provide comprehensive support to optimize your supply chain and accelerate your time to market.
We invite you to engage with our technical procurement team to discuss how this innovative route can benefit your pipeline. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic advantages tailored to your volume requirements. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing us to demonstrate our capability to deliver high-purity beta-trifluoromethyl thiophosphates that meet your exacting standards.
