Advanced Synthesis of 2-Bromo-1,3-Dimethoxybenzene for Commercial Scale-Up
Advanced Synthesis of 2-Bromo-1,3-Dimethoxybenzene for Commercial Scale-Up
In the rapidly evolving landscape of fine chemical synthesis, the demand for high-purity aromatic intermediates capable of supporting complex downstream applications continues to surge. Patent CN111269094A introduces a groundbreaking preparation method for 2-bromo-1,3-dimethoxybenzene, a critical building block extensively utilized in the development of novel organic luminescent materials and advanced pharmaceutical agents. This technical disclosure represents a significant leap forward from traditional halogenation techniques, addressing long-standing challenges regarding regioselectivity and environmental impact. By leveraging a sophisticated lithiation strategy followed by a controlled bromination using 1,2-dibromotetrafluoroethane, the process achieves exceptional purity levels exceeding 99.9% GC. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediates supplier, understanding the mechanistic advantages of this route is essential for securing a stable supply chain of high-value precursors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-bromo-1,3-dimethoxybenzene has relied heavily on direct electrophilic aromatic substitution using aggressive brominating agents such as elemental bromine, N-bromosuccinimide (NBS), or hydrobromic acid. These conventional pathways are plagued by inherent selectivity issues due to the activating nature of the methoxy groups, which often leads to poly-brominated byproducts and difficult-to-separate isomers. The resulting crude mixtures necessitate rigorous and costly purification protocols, frequently involving column chromatography or multiple recrystallization cycles that drastically reduce overall yield. Furthermore, the use of elemental bromine poses significant safety hazards and generates corrosive waste streams, creating substantial environmental compliance burdens for manufacturing facilities. The low atom economy and high E-factor associated with these legacy methods render them increasingly unsustainable for modern green chemistry initiatives and large-scale commercial production.
The Novel Approach
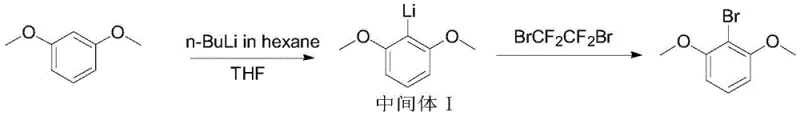
The innovative methodology outlined in the patent circumvents these pitfalls by employing a Directed Ortho-Metalation (DoM) strategy coupled with a unique bromine source. Instead of direct substitution, the process first converts m-phenyl dimethyl ether into a lithiated intermediate using n-butyllithium under strictly controlled thermal conditions. This organometallic species is then reacted with 1,2-dibromotetrafluoroethane, a reagent that offers superior control over the bromination event compared to traditional sources. This two-step sequence effectively bypasses the formation of poly-halogenated impurities, streamlining the downstream workup. The result is a robust synthetic route that not only enhances the total yield to over 88% but also simplifies the isolation of the target molecule to a single recrystallization step. This paradigm shift enables cost reduction in pharmaceutical intermediates manufacturing by minimizing solvent consumption and waste disposal costs while maximizing throughput.
Mechanistic Insights into Lithiation-Bromination Strategy
The core of this synthesis lies in the precise generation of the aryl lithium species at the 2-position of the benzene ring. The methoxy groups at the 1 and 3 positions act as powerful directing groups, coordinating with the lithium cation to facilitate deprotonation at the sterically accessible ortho-position. Maintaining the reaction temperature between 5-10°C during the addition of n-butyllithium is critical to prevent competing side reactions such as nucleophilic attack on the methoxy carbon or Wurtz-type coupling. Once the intermediate I is formed, the system is cooled to cryogenic temperatures (-60 to -70°C) before the introduction of the electrophile. This extreme cold is necessary to manage the exothermic nature of the subsequent bromination and to suppress any potential decomposition of the sensitive organolithium intermediate. The use of 1,2-dibromotetrafluoroethane serves as an efficient bromine transfer agent, likely proceeding through a radical or concerted mechanism that ensures mono-bromination with high fidelity.
Following the bromination event, the reaction mixture is carefully quenched with dilute hydrochloric acid to neutralize residual base and decompose any remaining organometallic species. The choice of quenching conditions is vital; adding the acid at 0-5°C prevents thermal shock and ensures the stability of the newly formed carbon-bromine bond. The subsequent extraction with ethyl acetate and washing with saturated brine effectively removes inorganic salts and polar impurities. Finally, the recrystallization from n-hexane exploits the differential solubility of the product versus trace impurities, yielding needle-shaped crystals of exceptional quality. This meticulous control over every unit operation—from lithiation to crystallization—demonstrates a deep understanding of process chemistry required for producing high-purity pharmaceutical intermediates suitable for sensitive catalytic couplings in drug discovery.
How to Synthesize 2-Bromo-1,3-Dimethoxybenzene Efficiently
Implementing this synthesis requires strict adherence to the thermal profiles and stoichiometric ratios defined in the patent to ensure reproducibility and safety. The process begins with the dissolution of the starting material in tetrahydrofuran, followed by the controlled addition of the lithiating agent. Operators must monitor the internal temperature closely to maintain the narrow window required for optimal lithiation. Subsequent cooling to cryogenic temperatures prior to bromination is non-negotiable for safety and yield optimization. The detailed standardized synthesis steps, including specific addition rates and quenching protocols, are outlined below to guide process engineers in scaling this technology.
- Dissolve m-phenyl dimethyl ether in THF, cool to 5-10°C, and add n-BuLi to form Intermediate I.
- Cool Intermediate I to -60 to -70°C and add 1,2-dibromotetrafluoroethane solution, then warm to 20-25°C.
- Quench with dilute HCl, extract with ethyl acetate, and recrystallize the crude product from n-hexane to achieve >99.9% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers tangible strategic benefits beyond mere technical superiority. The reliance on readily available industrial commodities such as m-phenyl dimethyl ether and n-butyllithium mitigates the risk of raw material shortages that often plague specialty chemical supply chains. Unlike processes dependent on scarce or highly regulated reagents, this method utilizes bulk chemicals that can be sourced from multiple global suppliers, ensuring business continuity. Furthermore, the simplification of the purification process eliminates the need for expensive chromatographic resins or extensive solvent swaps, directly translating to reduced operational expenditures. The ability to achieve >99.9% purity through simple recrystallization significantly shortens the production cycle time, allowing for faster turnaround on customer orders and improved inventory turnover rates.
- Cost Reduction in Manufacturing: The elimination of hazardous elemental bromine and the reduction in side-product formation lead to substantial savings in waste treatment and raw material costs. By avoiding complex purification trains, the process lowers energy consumption and solvent usage, driving down the overall cost of goods sold without compromising quality standards.
- Enhanced Supply Chain Reliability: Utilizing common industrial solvents like THF and n-hexane, along with stable starting materials, reduces dependency on niche suppliers. This diversification of the supply base enhances resilience against market volatility and logistical disruptions, ensuring a consistent flow of high-purity intermediates to downstream customers.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of heavy metal catalysts simplify the scale-up from pilot plant to multi-ton production. The process aligns with green chemistry principles by minimizing toxic waste generation, thereby reducing the regulatory burden and facilitating smoother environmental permitting for manufacturing sites.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 2-bromo-1,3-dimethoxybenzene. These insights are derived directly from the patented methodology and are intended to clarify the feasibility of adopting this route for large-scale operations. Understanding these nuances is crucial for technical teams evaluating vendor capabilities and process robustness.
Q: Why is 1,2-dibromotetrafluoroethane preferred over elemental bromine?
A: Using 1,2-dibromotetrafluoroethane significantly improves regioselectivity and reduces side reactions compared to traditional brominating agents like Br2 or NBS, leading to higher purity and easier purification.
Q: What is the expected purity of the final product?
A: The patented process achieves a GC purity of over 99.9% after a single recrystallization step using n-hexane, meeting stringent requirements for pharmaceutical and electronic applications.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the method utilizes readily available industrial raw materials and avoids hazardous reagents like elemental bromine, making it environmentally friendly and highly scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Bromo-1,3-Dimethoxybenzene Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced materials depends on the availability of ultra-high purity precursors. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate thermal controls required for this lithiation-bromination sequence are maintained with precision. We operate stringent purity specifications and utilize rigorous QC labs equipped with state-of-the-art GC and HPLC systems to verify that every batch meets the >99.9% purity benchmark essential for OLED and pharmaceutical applications. Our commitment to quality assurance guarantees that our clients receive materials that perform consistently in their most sensitive downstream reactions.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clearer picture of the economic advantages of switching to this greener, more efficient route. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your volume needs, ensuring a seamless integration of our high-quality intermediates into your supply chain.
