Scalable Synthesis of 3-(Benzyloxy)-1-cyclobutanone via Hunsdiecker Reaction for Pharmaceutical Applications
Introduction to Advanced Cyclobutanone Synthesis
The pharmaceutical industry continuously demands robust and scalable synthetic routes for complex heterocyclic intermediates, particularly those serving as core scaffolds for antiviral and oncology therapeutics. Patent CN111320535B, published in April 2022, introduces a groundbreaking preparation method for 3-(benzyloxy)-1-cyclobutanone, a critical building block for HIV-1 reverse transcriptase inhibitors and PLK inhibitors. This innovation addresses the longstanding challenges associated with the commercial manufacturing of strained four-membered ring ketones, offering a pathway that bypasses the severe safety and environmental liabilities of previous methodologies. By leveraging a strategic combination of nucleophilic substitution and the classic Hunsdiecker reaction, this technology enables the production of high-purity intermediates under significantly milder conditions. For global procurement teams and R&D directors, this represents a pivotal shift towards safer, more sustainable, and economically viable supply chains for essential pharmaceutical intermediates.
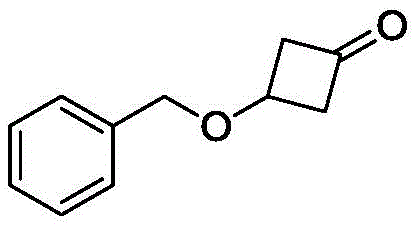
Furthermore, the structural integrity of the cyclobutanone ring is preserved throughout the synthesis, ensuring that the final product meets the rigorous purity specifications required for downstream API synthesis. The ability to source such complex motifs reliably is often a bottleneck in drug development timelines. This patent not only solves the synthetic challenge but also optimizes the economic profile of the molecule by utilizing inexpensive, commodity-grade starting materials. As we delve deeper into the technical specifics, it becomes evident that this methodology is designed with industrial scalability in mind, providing a reliable foundation for the commercial scale-up of complex pharmaceutical intermediates without compromising on safety or yield.
The Limitations of Conventional Methods vs. The Novel Approach
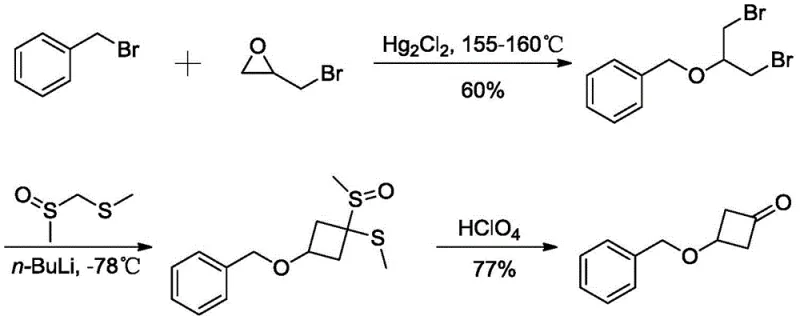
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of 3-(benzyloxy)-1-cyclobutanone was plagued by hazardous reagents and extreme reaction conditions that posed significant barriers to industrial adoption. One prominent prior art method, documented in WO2013011115A1, relied on the use of mercuric chloride as a catalyst at dangerously high temperatures ranging from 155-160°C. This approach not only introduced toxic heavy metal contamination risks requiring extensive purification but also utilized perchloric acid in the final oxidation step, a highly regulated and dangerous "pipe product" that complicates logistics and storage. Another alternative route employed zinc-copper reagents and zinc powder for dechlorination, which introduced the risk of dust explosions and required stoichiometric amounts of expensive metals. These conventional pathways resulted in lower total yields and generated substantial hazardous waste, making them unsuitable for modern green chemistry standards and cost reduction in API manufacturing.
The Novel Approach
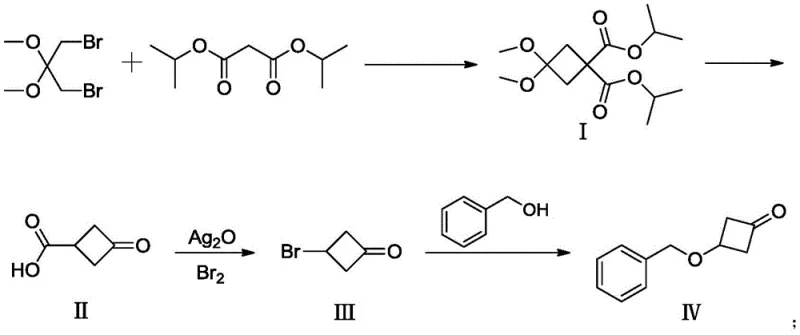
In stark contrast, the novel approach detailed in CN111320535B utilizes a clever four-step sequence that begins with the nucleophilic substitution of 3-dibromo-2,2-dimethoxypropane and diisopropyl malonate. This initial cyclization builds the four-membered ring under controlled basic conditions, avoiding the need for high-energy thermal inputs. The subsequent steps involve acid-mediated deprotection and a highly efficient Hunsdiecker reaction to install the necessary functionality, culminating in a mild etherification with benzyl alcohol. This route completely eliminates the need for mercury, perchloric acid, or explosive zinc powders, replacing them with manageable reagents like silver oxide and elemental bromine. The result is a streamlined process that operates at moderate temperatures (40-70°C for key steps), drastically simplifying the engineering requirements for reactor design and thermal control. This shift not only enhances operator safety but also significantly lowers the barrier to entry for reliable pharmaceutical intermediate suppliers looking to offer this material at competitive price points.
Mechanistic Insights into the Hunsdiecker Bromination Strategy
The cornerstone of this synthetic success lies in the third step, which employs the Hunsdiecker reaction to convert the carboxylic acid intermediate (II) into the corresponding bromide (III). Mechanistically, this involves the formation of a silver carboxylate salt in situ using silver oxide, followed by the addition of elemental bromine. The reaction proceeds through a radical mechanism where the silver-carboxylate bond undergoes homolytic cleavage upon interaction with bromine, releasing carbon dioxide and generating an alkyl radical. This radical immediately captures a bromine atom to form the desired C-Br bond. The elegance of this transformation in the context of a cyclobutanone system is that it occurs under relatively mild thermal conditions (40-70°C), preventing the ring-opening or decomposition that often plagues strained ring systems under harsher radical conditions. The patent data indicates that this step achieves yields as high as 97%, demonstrating exceptional chemoselectivity and minimal formation of side products.
From an impurity control perspective, the choice of solvent (DCM, chloroform, or carbon tetrachloride) and the precise control of the bromine addition rate are critical. The protocol specifies heating to reflux to ensure complete conversion of the silver salt, followed by careful cooling and filtration to remove silver bromide precipitate. This solid-liquid separation is straightforward and scalable, ensuring that the resulting organic phase contains the bromo-intermediate with high purity. By avoiding strong oxidants like perchloric acid used in older methods, the risk of over-oxidation or chlorination of the cyclobutane ring is entirely mitigated. This mechanistic precision ensures that the final product profile is clean, reducing the burden on downstream purification processes and facilitating the production of high-purity pharmaceutical intermediates that meet strict regulatory guidelines for residual metals and genotoxic impurities.
How to Synthesize 3-(Benzyloxy)-1-cyclobutanone Efficiently
The execution of this synthesis requires careful attention to reaction parameters, particularly during the base-mediated cyclization and the final etherification steps. The process begins with the generation of an enolate from diisopropyl malonate using sodium hydride in DMF, followed by the addition of the dibromo precursor to close the ring. Subsequent acid hydrolysis removes the protecting groups to reveal the keto-acid, which is then subjected to the silver-mediated bromination. The final step involves the nucleophilic attack of benzyl alkoxide on the bromo-cyclobutanone. Each step has been optimized in the patent examples to maximize yield, with reported efficiencies of 90%, 98%, 97%, and 96% respectively. For process chemists looking to implement this route, maintaining anhydrous conditions during the base-mediated steps and controlling the exotherm during bromine addition are paramount. The detailed standardized synthesis steps see the guide below.
- Perform nucleophilic substitution between 3-dibromo-2,2-dimethoxypropane and diisopropyl malonate using NaH in DMF to form the cyclobutane intermediate (I).
- Deprotect and hydrolyze compound (I) under acidic conditions (e.g., HCl reflux) to yield 3-oxocyclobutanecarboxylic acid (II).
- Convert compound (II) to its silver salt using Ag2O, then react with elemental bromine (Hunsdiecker reaction) to obtain the bromo-intermediate (III).
- Execute nucleophilic substitution between compound (III) and benzyl alcohol using a base like NaH in THF to finalize 3-(benzyloxy)-1-cyclobutanone (IV).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers profound strategic advantages that extend beyond simple unit cost savings. The elimination of highly regulated and dangerous reagents such as perchloric acid and mercuric chloride removes significant logistical hurdles and compliance costs associated with the storage, transport, and disposal of hazardous materials. Furthermore, the reliance on commodity chemicals like diisopropyl malonate and acetone derivatives ensures a stable and resilient supply base, insulating the production schedule from the volatility often seen with specialized organometallic reagents. This stability is crucial for maintaining continuous manufacturing operations and meeting the just-in-time delivery expectations of major pharmaceutical clients. By adopting this method, organizations can achieve cost reduction in pharmaceutical intermediate manufacturing through simplified waste treatment protocols and reduced insurance premiums related to hazardous material handling.
- Cost Reduction in Manufacturing: The new process eliminates the need for expensive and toxic heavy metal catalysts like mercury and zinc-copper couples, which not only reduces raw material costs but also removes the expensive downstream processing steps required to strip trace metals from the final product. The high yields observed in each step (consistently above 90%) minimize material loss and solvent usage, leading to a substantially improved overall mass balance. Additionally, the avoidance of cryogenic conditions (-78°C) required in some prior art routes significantly lowers energy consumption, as the reactions proceed efficiently at reflux or moderate temperatures. These factors combine to create a leaner, more cost-effective production model that enhances margin potential without sacrificing quality.
- Enhanced Supply Chain Reliability: By utilizing starting materials that are readily available on the global chemical market, this synthesis route mitigates the risk of supply disruptions caused by the scarcity of exotic reagents. The process does not rely on "pipe products" or strictly controlled substances that require special permits for purchase and transport, thereby streamlining the procurement workflow and reducing lead times. The robustness of the reaction conditions means that production can be easily transferred between different manufacturing sites or scaled up without the need for specialized, corrosion-resistant equipment required for handling perchloric acid. This flexibility ensures a consistent and reliable supply of high-purity 3-(benzyloxy)-1-cyclobutanone, safeguarding the downstream API production schedules of our partners.
- Scalability and Environmental Compliance: The mild reaction temperatures and the absence of explosive reagents make this process inherently safer and easier to scale from pilot plant to multi-ton commercial production. The waste stream is significantly cleaner, devoid of heavy metal sludge, which simplifies effluent treatment and ensures compliance with increasingly stringent environmental regulations. The use of common solvents like DCM, THF, and DMF allows for established recovery and recycling protocols, further enhancing the sustainability profile of the manufacturing process. This alignment with green chemistry principles not only reduces the environmental footprint but also future-proofs the supply chain against evolving regulatory pressures regarding chemical safety and waste management.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in patent CN111320535B, offering clarity on yield expectations, safety profiles, and scalability. Understanding these nuances is essential for R&D teams evaluating this route for technology transfer and for procurement specialists assessing the long-term viability of the supply source. We encourage stakeholders to review these details to fully appreciate the operational improvements this method brings to the production of cyclobutanone derivatives.
Q: What are the primary safety advantages of the new synthesis route compared to prior art?
A: The new route eliminates the use of mercuric chloride and perchloric acid, both of which are highly hazardous and strictly controlled substances. It also avoids the use of explosive zinc powder, significantly reducing operational risk.
Q: How does the Hunsdiecker reaction step impact the overall yield?
A: The Hunsdiecker reaction step (Step 3) in this patent demonstrates exceptional efficiency, with reported yields reaching up to 97%. This high conversion rate minimizes waste and maximizes the throughput of the valuable bromo-intermediate.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process utilizes mild reaction temperatures (40-70°C for key steps) and commercially available raw materials like diisopropyl malonate and acetone derivatives, making it highly scalable and cost-effective for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-(Benzyloxy)-1-cyclobutanone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex pharmaceutical intermediates requires more than just a patent; it demands deep process expertise and a commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of 3-(benzyloxy)-1-cyclobutanone meets the exacting standards required for HIV inhibitor and oncology drug synthesis. Our capability to implement the safe, high-yield route described in CN111320535B positions us as a strategic partner capable of delivering both technical excellence and supply security.
We invite global pharmaceutical companies and contract manufacturers to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific project needs. By leveraging our manufacturing capabilities, you can access a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this safer, more efficient process. We encourage you to contact us today to request specific COA data, route feasibility assessments, and sample availability, allowing you to validate the superior quality and reliability of our supply chain for your critical drug development programs.
