Scalable Production of High-Purity 3-Bromocarbazole via Novel Phase Transfer Catalysis
Introduction to Advanced 3-Bromocarbazole Manufacturing
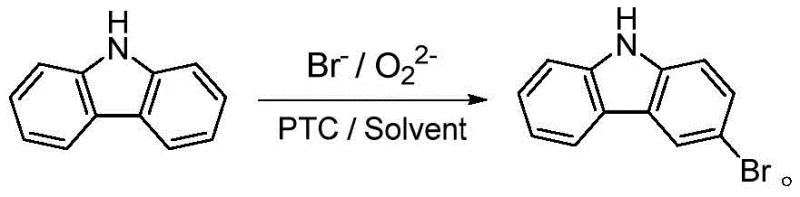
The global demand for high-performance organic photoelectric materials and specialized pharmaceutical intermediates has necessitated a paradigm shift in synthetic methodologies, particularly for key building blocks like 3-bromocarbazole. Patent CN112521328A, published in March 2021, introduces a groundbreaking preparation method that addresses longstanding inefficiencies in carbazole functionalization. This technology leverages a Br⁻/O₂²⁻ oxidative bromination system facilitated by phase transfer catalysis (PTC) to achieve exceptional purity and yield. Unlike traditional electrophilic substitutions that struggle with regioselectivity and environmental impact, this novel approach utilizes mild conditions and inexpensive reagents to produce 3-bromocarbazole with a purity exceeding 99%. For R&D directors and procurement specialists, this patent represents a viable pathway to cost reduction in pharmaceutical intermediate manufacturing while adhering to stringent green chemistry principles. The method effectively overcomes the poor solubility of carbazole, a common bottleneck, ensuring complete conversion and simplifying downstream purification processes significantly.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-bromocarbazole has relied heavily on direct bromination using elemental bromine or N-bromosuccinimide (NBS), methods fraught with significant operational and economic drawbacks. The use of elemental bromine poses severe safety hazards due to its high toxicity and corrosivity, requiring specialized containment infrastructure that drives up capital expenditure. Furthermore, these traditional reagents often suffer from poor atom economy, generating substantial amounts of hazardous waste that complicate disposal and increase environmental compliance costs. A critical technical challenge lies in the regioselectivity; the electron cloud density at the C3 and C6 positions of carbazole is similar, leading to the frequent formation of 3,6-dibromocarbazole impurities which are difficult to separate. Existing methods often require multi-step protection and deprotection strategies, such as acetylation at the 9-position, which elongates the synthetic route, reduces overall yield, and escalates production costs, making them unsuitable for large-scale industrial applications where efficiency is paramount.
The Novel Approach
The methodology disclosed in CN112521328A revolutionizes this landscape by employing an in-situ generation of active bromine species within a Br⁻/O₂²⁻ system, eliminating the need for hazardous molecular bromine. By integrating a phase transfer catalyst, the process effectively manages the solubility disparity between the organic carbazole substrate and the aqueous bromide/oxidant phases, ensuring a homogeneous reaction environment that accelerates kinetics. This approach allows for precise control over the bromine source equivalent, typically maintained between 1.05:1 and 1.15:1 relative to carbazole, which drastically suppresses the formation of dibrominated byproducts. The reaction proceeds under mild temperatures ranging from 0°C to 20°C, reducing energy consumption and thermal stress on the equipment. Consequently, this novel route offers a streamlined, single-step transformation that delivers high-purity 3-bromocarbazole with yields reaching up to 83%, providing a robust foundation for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into PTC-Mediated Oxidative Bromination
The core innovation of this synthesis lies in the synergistic interaction between the phase transfer catalyst and the oxidative bromination cycle. In the absence of a PTC, carbazole exhibits poor solubility in the biphasic mixture, leading to incomplete conversion and the physical entrapment of starting material within the product matrix. The addition of catalysts such as 18-crown-6 or tetrabutylammonium bromide facilitates the transport of bromide anions into the organic phase, where they interact with the oxidant (e.g., hydrogen peroxide) to generate the active electrophilic bromine species locally. This localized generation ensures that the bromination occurs selectively at the C3 position before the electron density is sufficiently depleted to prevent further substitution at C6. The strict control of reaction parameters, including the dropwise addition of oxidants and maintenance of low temperatures, kinetically favors the mono-brominated product, thereby enhancing the selectivity profile significantly compared to thermodynamic control methods.
Furthermore, the inclusion of 2,6-di-tert-butyl-p-cresol (BHT) as a stabilizer plays a pivotal role in maintaining solvent integrity and reaction safety. Organic solvents like tetrahydrofuran are susceptible to acidic hydrolysis and oxidative decomposition under the reaction conditions, which can lead to the formation of peroxides and other hazardous byproducts. BHT acts as a radical scavenger, mitigating these degradation pathways and ensuring that the solvent remains stable throughout the 6 to 10-hour reaction window. This stabilization not only protects the equipment from corrosion but also prevents the introduction of solvent-derived impurities into the final product stream. The combination of precise stoichiometric control, phase transfer enhancement, and radical stabilization creates a highly reproducible process capable of delivering consistent quality, which is essential for a reliable pharmaceutical intermediate supplier aiming to meet global regulatory standards.
How to Synthesize 3-Bromocarbazole Efficiently
Implementing this synthesis requires careful attention to the sequential addition of reagents and temperature management to maximize yield and purity. The process begins with the dissolution of carbazole in a selected organic solvent, such as dichloromethane or tetrahydrofuran, which must contain a specific mass fraction of BHT stabilizer ranging from 0.01% to 1.00%. Once the phase transfer catalyst is introduced, the system is cooled to 0°C to prepare for the exothermic bromination event. The detailed standardized synthesis steps, including specific molar ratios and workup procedures, are outlined below to guide process engineers in replicating this high-efficiency protocol.
- Dissolve carbazole in an organic solvent containing BHT stabilizer and add a phase transfer catalyst under stirring at room temperature.
- Cool the system to 0°C, add brominating agent (e.g., HBr or NaBr) dropwise, followed by the oxidant (e.g., H2O2) in batches while maintaining 0-20°C.
- Stir for 6-10 hours until conversion exceeds 99.5%, then neutralize, extract, wash, and recrystallize the crude product to obtain high-purity 3-bromocarbazole.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the adoption of this oxidative bromination technology offers transformative benefits that extend beyond mere technical feasibility. The replacement of expensive and hazardous reagents like NBS or liquid bromine with commodity chemicals such as sodium bromide and hydrogen peroxide results in a drastic reduction in raw material costs. This shift not only lowers the direct cost of goods sold but also mitigates the risks associated with the storage and handling of dangerous substances, thereby reducing insurance premiums and safety compliance overheads. The simplified post-treatment process, which involves standard extraction and recrystallization rather than complex chromatographic separations, significantly shortens the production cycle time. This efficiency gain allows manufacturers to respond more agilely to market fluctuations, ensuring a steady flow of high-purity pharmaceutical intermediates to downstream clients without the bottlenecks typical of legacy synthetic routes.
- Cost Reduction in Manufacturing: The elimination of noble metal catalysts and expensive brominating agents like NBS fundamentally alters the cost structure of production. By utilizing cheap, bulk-available bromide salts and hydrogen peroxide, the process achieves high atom economy where the only byproduct is water, minimizing waste disposal fees. The high conversion rate (>99.5%) ensures that raw material utilization is maximized, reducing the need for recycling unreacted starting materials. Furthermore, the mild reaction conditions reduce energy consumption for heating and cooling, contributing to substantial operational expenditure savings that can be passed down the supply chain.
- Enhanced Supply Chain Reliability: The reliance on widely available, non-controlled reagents enhances the resilience of the supply chain against geopolitical or logistical disruptions. Unlike specialized reagents that may have long lead times or single-source dependencies, sodium bromide and hydrogen peroxide are commoditized chemicals with robust global availability. The robustness of the reaction against minor variations in conditions ensures consistent batch-to-batch quality, reducing the risk of production failures or out-of-specification batches that could delay shipments. This reliability is critical for maintaining continuous operations in the fast-paced electronics and pharmaceutical sectors.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, having been validated from gram to multi-kilogram scales with consistent results. The absence of heavy metal contaminants simplifies the purification process, ensuring the final product meets stringent specifications for electronic and medical applications without requiring additional metal scavenging steps. Additionally, the green nature of the reagents and the aqueous byproduct stream align with increasingly strict environmental regulations, facilitating easier permitting and reducing the carbon footprint of the manufacturing facility, which is a key metric for modern sustainable procurement strategies.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis method. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on critical process parameters. Understanding these nuances is essential for process chemists aiming to adapt this technology for their specific production environments.
Q: How does the new method improve selectivity over traditional bromination?
A: By strictly controlling the bromine source equivalent (1.05-1.15:1) and temperature (0-20°C), the method minimizes the formation of 3,6-dibromocarbazole, ensuring high mono-bromination selectivity.
Q: What is the role of the phase transfer catalyst in this reaction?
A: The phase transfer catalyst enhances the solubility of carbazole in the organic-aqueous phase system, accelerating reaction speed and preventing product inclusion, which leads to higher conversion rates.
Q: Why is BHT added to the reaction solvent?
A: BHT acts as a stabilizer to prevent acidic hydrolysis and oxidative decomposition of the organic solvent, particularly tetrahydrofuran, ensuring reaction safety and solvent integrity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Bromocarbazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of next-generation OLED materials and pharmaceutical compounds. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to maintaining stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify that every batch of 3-bromocarbazole meets the exacting standards required for sensitive optoelectronic applications. Our capability to implement green chemistry protocols like the oxidative bromination method demonstrates our dedication to sustainable manufacturing practices that align with our clients' corporate responsibility goals.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific volume and purity requirements. By leveraging our expertise, you can secure a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this greener, more efficient process. We encourage you to request specific COA data and route feasibility assessments to validate the suitability of our 3-bromocarbazole for your supply chain, ensuring a partnership built on transparency, quality, and mutual growth in the competitive global chemical market.
