Advanced Synthesis of 9,10-Bis(chloromethyl)anthracene for High-Performance Optical Materials
The landscape of fine chemical synthesis for optoelectronic intermediates is undergoing a significant transformation driven by the need for safer, more efficient, and environmentally compliant processes. A pivotal advancement in this domain is documented in patent CN102108041B, which details a refined methodology for the production of 9,10-bis(chloromethyl)anthracene, a critical building block for fluorescent materials and organic semiconductors. This innovation addresses the longstanding industrial challenges associated with the chloromethylation of polycyclic aromatic hydrocarbons, specifically replacing hazardous gas-phase reagents with a controlled liquid-phase system. By utilizing a saturated solution of hydrochloric acid in dioxane, the process not only mitigates the severe corrosion issues inherent to traditional methods but also enhances the overall purity profile of the final product. For R&D directors and procurement specialists seeking a reliable electronic chemical supplier, understanding the nuances of this patented route is essential for securing a stable supply chain of high-performance precursors used in OLEDs and advanced diagnostic applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 9,10-disubstituted anthracene derivatives has relied heavily on the direct introduction of anhydrous hydrogen chloride gas into a reaction mixture containing anthracene and paraformaldehyde. While chemically feasible, this conventional gas-phase approach presents formidable engineering and economic obstacles that hinder scalable manufacturing. The continuous flow of corrosive HCl gas exerts extreme stress on reactor vessels, piping, and sealing components, leading to accelerated equipment degradation and frequent maintenance downtime. Furthermore, controlling the stoichiometry and reaction kinetics in a gas-liquid heterogeneous system is inherently difficult, often resulting in inconsistent conversion rates and the formation of undesirable by-products that complicate downstream purification. The inefficiency of gas absorption also means that a significant portion of the reagent may be lost to the exhaust, necessitating complex scrubbing systems to meet environmental regulations. Consequently, the operational costs associated with equipment replacement, safety monitoring, and waste treatment render the traditional method economically unsustainable for modern, high-volume production facilities aiming for cost reduction in electronic chemical manufacturing.
The Novel Approach
In stark contrast, the methodology disclosed in the patent introduces a paradigm shift by employing a homogeneous liquid phase reaction medium consisting of dioxane saturated with concentrated hydrochloric acid. This strategic modification fundamentally alters the reaction dynamics, allowing for precise control over the concentration of the chloromethylating agent throughout the process. By dissolving the HCl in dioxane prior to the addition of anthracene and paraformaldehyde, the system ensures uniform contact between reactants, which promotes consistent reaction rates and minimizes localized hot spots that can lead to decomposition. The liquid-phase nature of the reagent eliminates the need for complex gas handling infrastructure, thereby simplifying the operational workflow and reducing the risk of accidental leaks. Moreover, the use of dioxane as a solvent offers the distinct advantage of recyclability; post-reaction processing allows for the recovery and reuse of the solvent, which significantly lowers raw material consumption. This approach not only safeguards capital equipment from corrosive damage but also aligns with green chemistry principles by reducing waste generation, making it an ideal candidate for the commercial scale-up of complex polymer additives and optical intermediates.
Mechanistic Insights into Chloromethylation of Anthracene
The core chemical transformation driving this synthesis is an electrophilic aromatic substitution, specifically a chloromethylation reaction facilitated by the in situ generation of a chloromethyl cation equivalent. In the presence of the acidic dioxane medium, paraformaldehyde depolymerizes to release formaldehyde, which subsequently reacts with the hydrochloric acid to form the active electrophilic species, likely a protonated formaldehyde or a chloromethyl oxonium ion. The anthracene molecule, possessing high electron density at the 9 and 10 positions due to its resonance structure, acts as a potent nucleophile attacking this electrophile. This regioselectivity is thermodynamically favored because substitution at the central ring preserves the aromaticity of the two flanking benzene rings, whereas substitution at other positions would disrupt the conjugated system more severely. The reaction proceeds through a sigma-complex intermediate before losing a proton to restore aromaticity, ultimately installing the chloromethyl groups at the 9 and 10 positions simultaneously or sequentially depending on the local concentration of reagents.
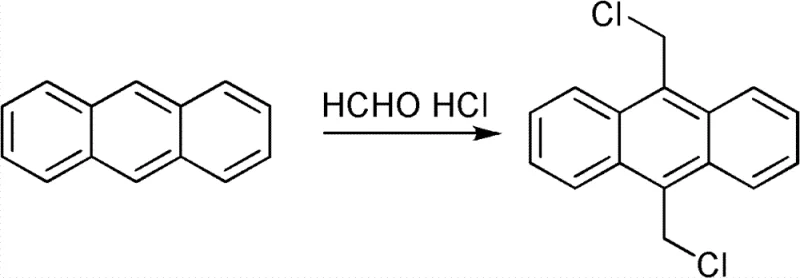
Controlling the impurity profile in this reaction is paramount for applications in high-purity OLED material manufacturing, where trace contaminants can act as quenching sites for excitons. The patented process achieves superior impurity control primarily through the moderation of reaction conditions afforded by the liquid solvent system. Unlike the aggressive environment created by bubbling dry HCl gas, the saturated solution provides a buffered acidity that prevents over-chlorination or the formation of polymeric tars often seen in harsher conditions. The specific mass ratio of concentrated hydrochloric acid to anthracene to paraformaldehyde, optimized between 2.5-3.0:1:0.8-1.2, ensures that the electrophile is generated at a rate that matches the nucleophilic attack, minimizing side reactions. Furthermore, the extended stirring period of 10 to 15 hours after the initial micro-reflux allows for the completion of the reaction and the equilibration of intermediates, ensuring that unreacted starting materials are minimized before the workup begins. This meticulous control results in a crude product that is already of high quality, requiring only a single recrystallization step from toluene to achieve purity levels exceeding 99.5%, which is critical for downstream electronic applications.
How to Synthesize 9,10-Bis(chloromethyl)anthracene Efficiently
Implementing this synthesis route requires careful attention to the preparation of the saturated acid solution and the thermal profile of the reaction to ensure reproducibility and safety. The process begins with the creation of the reactive medium by mixing dioxane and concentrated hydrochloric acid, followed by the simultaneous addition of the solid reactants to initiate the chloromethylation. Maintaining a micro-reflux state is crucial, as it provides the necessary activation energy for the depolymerization of paraformaldehyde and the subsequent electrophilic attack without causing thermal degradation of the sensitive anthracene core. Following the heating phase, a prolonged stirring period at lower temperatures allows the reaction to reach completion gently. For a comprehensive, step-by-step breakdown of the exact quantities, temperatures, and workup procedures validated by experimental data, please refer to the standardized synthesis guide below.
- Prepare a saturated solution of concentrated hydrochloric acid in dioxane within a reaction vessel equipped with stirring capabilities.
- Add anthracene and paraformaldehyde to the solution in a mass ratio ranging from 1: 0.8 to 1:1.2 relative to anthracene, ensuring complete dispersion.
- Heat the mixture to a micro-reflux state for 3 to 5 hours, then cease heating and continue stirring for an additional 10 to 15 hours to complete the reaction.
- Filter the resulting yellow solid, wash thoroughly with dioxane, dry under vacuum at 40°C, and recrystallize from toluene to achieve purity exceeding 99.5%.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this liquid-phase chloromethylation protocol represents a strategic opportunity to optimize both operational expenditures and supply continuity. The primary economic driver is the drastic reduction in equipment maintenance and replacement costs; by eliminating the direct use of corrosive HCl gas, the lifespan of reactors and associated piping is significantly extended, deferring capital investment in new hardware. Additionally, the ability to recover and recycle the dioxane solvent creates a closed-loop system that reduces the volume of hazardous waste requiring disposal, thereby lowering compliance costs and environmental fees. The simplicity of the operation, which avoids complex gas flow controllers and scrubbers, also reduces the training burden on operators and minimizes the risk of production stoppages due to technical failures. These factors combine to create a more resilient manufacturing process that is less susceptible to the volatility of maintenance schedules and regulatory inspections.
- Cost Reduction in Manufacturing: The elimination of expensive gas-handling infrastructure and the reduction in equipment corrosion directly translate to lower fixed costs per unit of production. By utilizing a saturated solution instead of gaseous reagents, the process avoids the inefficiencies associated with gas absorption, ensuring that nearly all purchased hydrochloric acid participates in the reaction rather than being vented. Furthermore, the recyclability of the dioxane solvent means that raw material consumption is minimized, providing substantial cost savings over large-scale production runs without compromising the quality of the high-purity electronic chemical output.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route ensures a consistent and predictable output of 9,10-bis(chloromethyl)anthracene, which is vital for maintaining uninterrupted supply lines to downstream manufacturers of fluorescent materials. The use of readily available bulk chemicals like anthracene, paraformaldehyde, and commercial grade hydrochloric acid reduces dependency on specialized or scarce reagents that might face supply bottlenecks. The simplified operational protocol also means that production batches can be turned around more quickly and with fewer technical hurdles, effectively reducing lead time for high-purity intermediates and allowing suppliers to respond more agilely to market demand fluctuations.
- Scalability and Environmental Compliance: Scaling this process from laboratory to industrial tonnage is straightforward due to the homogeneous nature of the reaction mixture, which facilitates efficient heat transfer and mixing in large vessels. The reduced generation of corrosive waste streams simplifies effluent treatment, making it easier for facilities to meet stringent environmental discharge standards. This environmental compatibility not only mitigates regulatory risk but also enhances the corporate sustainability profile of the manufacturer, a growing requirement for partnerships with major multinational corporations in the electronics and pharmaceutical sectors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 9,10-bis(chloromethyl)anthracene, derived directly from the technical specifications and beneficial effects outlined in the patent literature. These insights are intended to clarify the operational advantages and quality benchmarks associated with this specific synthetic methodology for potential partners and technical evaluators.
Q: How does the saturated HCl/dioxane method improve upon traditional gas-phase chloromethylation?
A: The traditional method involves passing corrosive hydrogen chloride gas directly into the reaction system, which causes significant equipment degradation and safety hazards. The novel approach utilizes a pre-saturated solution of HCl in dioxane, which drastically reduces corrosion rates, simplifies operational procedures, and allows for solvent recovery, thereby lowering long-term capital expenditure.
Q: What purity levels can be achieved with this synthesis route?
A: Experimental data from the patent indicates that crude yields can reach over 80%, and subsequent recrystallization from toluene consistently produces 9,10-bis(chloromethyl)anthracene with a content of 99.5% to 99.7%. This high purity is critical for applications in organic electroluminescence and semiconductor materials where trace impurities can quench fluorescence.
Q: Is the dioxane solvent recoverable in this process?
A: Yes, one of the key economic advantages of this protocol is the recyclability of the dioxane solvent. Since the reaction medium is a liquid solution rather than a gas flow, the solvent can be separated during the workup and filtration stages, distilled, and reused in subsequent batches, contributing to substantial waste reduction and cost efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 9,10-Bis(chloromethyl)anthracene Supplier
At NINGBO INNO PHARMCHEM, we recognize that the integrity of your final optical or pharmaceutical product depends entirely on the quality of the intermediates you source. Our technical team has extensively analyzed the pathway described in CN102108041B and integrated its core principles into our own manufacturing protocols to ensure the delivery of superior materials. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that whether you require pilot quantities for R&D or bulk volumes for mass manufacturing, our capacity is aligned with your needs. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 9,10-bis(chloromethyl)anthracene meets the exacting standards required for high-performance electronic and fluorescent applications.
We invite you to collaborate with us to leverage these advanced synthetic capabilities for your next project. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our optimized processes can reduce your overall landed costs. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive efficiency and innovation in your supply chain.
