Advanced Manufacturing of 4-Aminothiophene[3,2-d]pyrimidine-7-carboxylic Acid for Kinase Inhibitors
The pharmaceutical industry's relentless pursuit of potent protein kinase inhibitors has placed a premium on the efficient synthesis of key heterocyclic scaffolds. Patent CN114213430A introduces a transformative preparation method for 4-aminothiophene[3,2-d]pyrimidine-7-carboxylic acid, a critical intermediate in the development of oncology therapeutics such as Belvarafenib. This technical breakthrough addresses the longstanding bottlenecks associated with traditional synthetic routes, offering a streamlined three-step pathway that significantly enhances process safety and scalability. By replacing hazardous high-pressure carbonylation steps with a robust oxidative strategy, this innovation provides a reliable protein kinase inhibitor intermediate supplier with a distinct competitive advantage in the global market. The structural integrity of the target molecule, as depicted in the patent, underscores its importance as a core building block for next-generation cancer therapies.
![Chemical structure of 4-aminothiophene[3,2-d]pyrimidine-7-carboxylic acid (Formula III)](/insights/img/4-aminothiophene-pyrimidine-synthesis-pharma-supplier-20260305041450-01.png)
Historically, the manufacturing of this pivotal scaffold has been plagued by complex multi-step sequences that rely on dangerous reagents and expensive catalysts. Conventional methodologies, such as those documented in Org. Process Res. Dev. 2021, typically involve a tedious sequence starting from thiophene amino carboxylic acid methyl ester. This legacy approach necessitates bromination, followed by a high-pressure carbon monoxide insertion catalyzed by palladium acetate and BINAP ligands. Such conditions not only impose severe safety risks due to the handling of toxic CO gas under pressure but also introduce significant cost burdens related to precious metal recovery and specialized equipment requirements. Furthermore, the lengthy reaction sequence inherently accumulates impurities, complicating downstream purification and reducing overall yield efficiency.
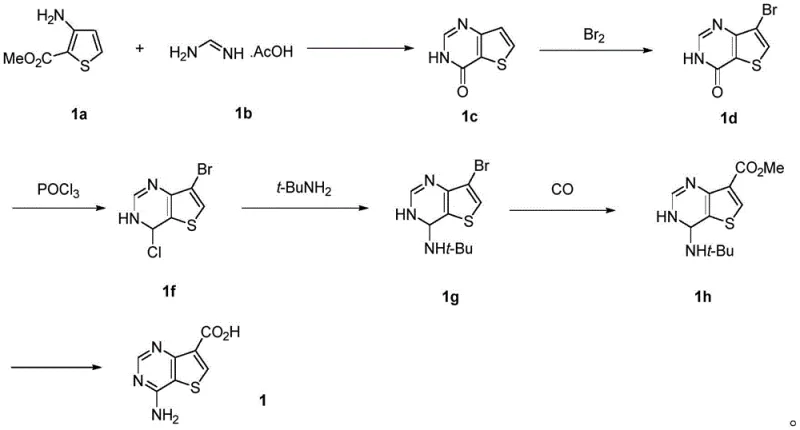
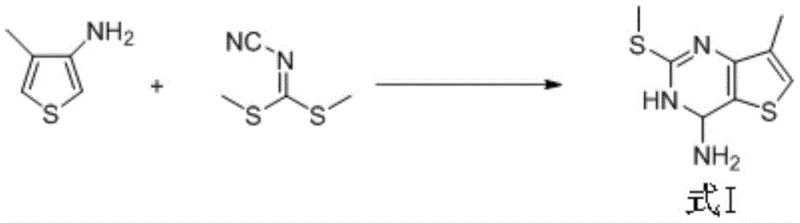
In stark contrast, the novel approach disclosed in the patent simplifies the entire synthesis into just three strategic transformations, fundamentally altering the economic and safety profile of production. The new route initiates with a direct condensation of 4-methyl-3-thiophenamine and N-cyanoimino-S,S-dimethyl dithiocarbonate, bypassing the need for halogenated precursors entirely. This is followed by a selective reductive desulfurization and a final oxidation step to install the carboxylic acid moiety. By eliminating the carbonylation step, the process removes the dependency on high-pressure reactors and toxic gases, thereby facilitating cost reduction in pharmaceutical manufacturing. The streamlined nature of this pathway ensures a shorter lead time for high-purity intermediates, allowing supply chain managers to respond more agilely to market demands without compromising on quality or regulatory compliance.
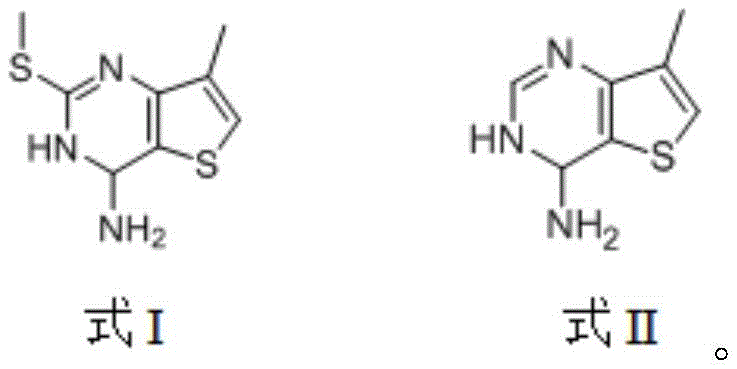
The mechanistic elegance of this new synthesis lies in its precise control over regioselectivity and functional group tolerance during the initial cyclization. The first step involves a nucleophilic addition where the amino group of 4-methyl-3-thiophenamine attacks the electrophilic carbon of the dithiocarbonate, followed immediately by an intramolecular Friedel-Crafts reaction. This tandem process occurs efficiently at elevated temperatures between 150°C and 170°C in polar aprotic solvents like DMF or acetonitrile. The reaction kinetics are optimized to favor the formation of the stable first intermediate (Formula I) while minimizing side reactions, ensuring a high-purity output that simplifies subsequent processing. This mechanistic insight is crucial for R&D directors aiming to replicate the process, as it highlights the importance of thermal energy in driving the cyclization to completion without the need for additional Lewis acid catalysts.
Following the construction of the pyrimidine ring, the process employs a highly effective desulfurization strategy to remove the methylthio group, a transformation that is often challenging in heterocyclic chemistry. Using Raney nickel as a heterogeneous catalyst in alcoholic solvents, the methylthio moiety is cleanly removed to yield the second intermediate (Formula II). This step is particularly advantageous because it avoids the use of harsh acidic or basic conditions that could degrade the sensitive thiophene ring. The final oxidation step utilizes common oxidants like potassium permanganate or activated manganese dioxide to convert the methyl group directly into a carboxylic acid. This direct oxidation is remarkably selective, preserving the amino group and the heterocyclic core, which demonstrates a superior impurity control mechanism compared to multi-step functional group interconversions found in older literature.
How to Synthesize 4-Aminothiophene[3,2-d]pyrimidine-7-carboxylic Acid Efficiently
Implementing this synthesis requires careful attention to reaction parameters, particularly temperature control during the initial cyclization and the choice of oxidant in the final step. The patent outlines a robust protocol that balances reaction speed with product stability, ensuring that the intermediates can be isolated and stored if necessary. For technical teams looking to adopt this methodology, the following guide summarizes the critical operational phases derived from the patent examples. Detailed standardized synthesis steps are provided in the section below to ensure reproducibility and adherence to quality standards.
- Perform nucleophilic addition and intramolecular Friedel-Crafts reaction between 4-methyl-3-thiophenamine and N-cyanoimino-S,S-dimethyl dithiocarbonate at 150-170°C to obtain the first intermediate.
- Execute reductive desulfurization of the first intermediate using Raney nickel in ethanol or methanol at 70-80°C to generate the second intermediate.
- Conduct oxidation of the second intermediate using potassium permanganate or activated manganese dioxide in tetrahydrofuran to yield the final carboxylic acid product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the shift away from palladium-catalyzed carbonylation represents a substantial opportunity for cost optimization and risk mitigation. The elimination of precious metal catalysts and high-pressure gas infrastructure drastically reduces the capital expenditure required for setting up production lines. Moreover, the reliance on commodity chemicals such as 4-methyl-3-thiophenamine and common oxidants ensures a stable supply chain that is less susceptible to the volatility often seen with specialized organometallic reagents. This stability is paramount for supply chain heads who prioritize continuity and predictability in the sourcing of critical oncology intermediates.
- Cost Reduction in Manufacturing: The removal of expensive palladium catalysts and phosphine ligands like BINAP significantly lowers the raw material costs per kilogram of product. Additionally, the simplified workup procedures, which rely on precipitation and filtration rather than complex chromatography, reduce solvent consumption and waste disposal costs. This lean manufacturing approach translates into significant margin improvements for large-scale producers without sacrificing product quality.
- Enhanced Supply Chain Reliability: By utilizing readily available starting materials and avoiding reagents with long lead times or strict transportation regulations (such as high-pressure CO cylinders), the new route enhances overall supply chain resilience. The ability to source inputs from multiple vendors reduces the risk of single-point failures, ensuring that production schedules remain uninterrupted even during global supply disruptions.
- Scalability and Environmental Compliance: The process operates under atmospheric pressure and avoids the generation of heavy metal waste streams associated with palladium catalysis. This aligns perfectly with modern green chemistry principles and stringent environmental regulations, making the scale-up from pilot plant to commercial production smoother and more compliant. The reduced environmental footprint also simplifies the permitting process for new manufacturing facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on process feasibility and product specifications.
Q: How does this new synthesis route improve safety compared to conventional methods?
A: The novel route eliminates the need for high-pressure carbon monoxide and palladium-catalyzed carbonylation, which are significant safety hazards in traditional synthesis. Instead, it utilizes standard atmospheric pressure oxidation and reduction steps.
Q: What are the purity levels achievable with this 3-step process?
A: The patent data indicates that the final product can achieve purity levels of up to 99% through simple purification steps like hexane precipitation and filtration, minimizing the need for complex chromatography.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process avoids expensive transition metal catalysts like palladium and BINAP ligands, and uses readily available solvents like ethanol and THF, making it highly scalable and cost-effective for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Aminothiophene[3,2-d]pyrimidine-7-carboxylic Acid Supplier
As the demand for targeted kinase inhibitors continues to surge, the ability to manufacture high-quality intermediates efficiently becomes a strategic asset. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging advanced synthetic methodologies like the one described in CN114213430A to deliver superior value to our global partners. Our facility boasts extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous volume requirements of clinical and commercial stages alike. With stringent purity specifications and rigorous QC labs, we guarantee that every batch of 4-aminothiophene[3,2-d]pyrimidine-7-carboxylic acid meets the highest international standards for pharmaceutical applications.
We invite procurement leaders and R&D directors to collaborate with us to optimize their supply chains for kinase inhibitor projects. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume needs. We encourage you to reach out today to request specific COA data and route feasibility assessments, ensuring that your project moves forward with the most efficient and reliable chemical foundation possible.
