Revolutionizing Peptide Intermediate Manufacturing: The NdSe2-Catalyzed Route to Fmoc-D-Pro-D-Pro-OH
Revolutionizing Peptide Intermediate Manufacturing: The NdSe2-Catalyzed Route to Fmoc-D-Pro-D-Pro-OH
The landscape of peptide intermediate manufacturing is undergoing a significant transformation driven by the need for higher purity, reduced costs, and scalable processes. A pivotal advancement in this domain is detailed in Chinese Patent CN110156873B, which discloses a novel preparation method for Fmoc-D-Pro-D-Pro-OH, a critical building block in the synthesis of complex therapeutic peptides. This patent introduces a groundbreaking catalytic system utilizing Neodymium Selenide (NdSe2) to facilitate the direct coupling of activated proline esters with proline sodium salts. Unlike conventional methods that suffer from cumbersome protection-deprotection sequences or reliance on expensive solid supports, this innovation offers a streamlined, two-step liquid-phase synthesis. By leveraging rare-earth catalysis, the process achieves exceptional purity levels exceeding 98.5% while operating under mild room temperature conditions. For global pharmaceutical manufacturers, this represents a paradigm shift towards more efficient and economically viable production strategies for dipeptide intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Fmoc-D-Pro-D-Pro-OH has been plagued by significant inefficiencies inherent to both traditional liquid-phase and solid-phase methodologies. In conventional liquid-phase synthesis, the strategy typically involves a laborious sequence where the Fmoc protecting group must be removed and subsequently reintroduced, leading to extended reaction times and increased consumption of expensive Fmoc reagents. Furthermore, these older liquid-phase routes often struggle with yield limitations, frequently falling below 50%, and fail to achieve the stringent purity standards required for modern API manufacturing, often capping at purities below 90%. On the other hand, solid-phase synthesis, while capable of higher purity, introduces its own set of economic and logistical burdens. The reliance on polymeric resins significantly inflates raw material costs, and the process necessitates the use of activated esters in vast excess—sometimes several hundred percent—to drive the reaction to completion. This wastefulness results in overall yields that can be abysmally low, often reported at less than 30% based on the activated ester, making it an unsustainable option for large-scale commercial production.
The Novel Approach
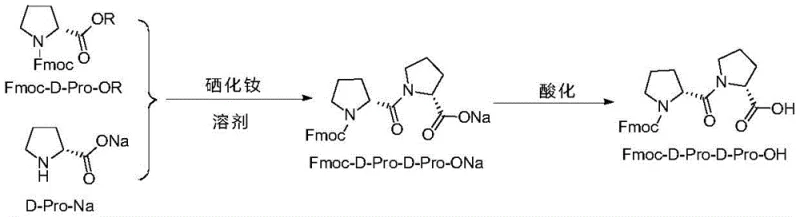
The methodology outlined in patent CN110156873B fundamentally disrupts these established norms by introducing a direct coupling mechanism that bypasses the need for resin support or repetitive protection cycles. The core of this innovation lies in the reaction between an activated ester, specifically Fmoc-D-Pro-OR, and the sodium salt of D-proline (D-Pro-Na) in the presence of the NdSe2 catalyst. This approach condenses the synthesis into a highly efficient workflow where the coupling occurs rapidly at room temperature, eliminating the energy costs associated with heating or cryogenic cooling. The result is a dramatic improvement in process metrics; the new method consistently delivers yields exceeding 69% and purities surpassing 98.5%. By avoiding the use of solid supports and minimizing reagent excess, this novel route not only simplifies the downstream purification process but also aligns perfectly with the principles of green chemistry, offering a robust solution for the industrial manufacture of high-purity pharmaceutical intermediates.
Mechanistic Insights into NdSe2-Catalyzed Peptide Coupling
The introduction of Neodymium Selenide (NdSe2) as a catalyst in peptide bond formation represents a sophisticated application of rare-earth chemistry to solve persistent selectivity issues. In the absence of this catalyst, the reaction between the activated ester and the amine salt is prone to competing side reactions, including hydrolysis of the activated ester and potential racemization, which severely compromises the optical purity of the final dipeptide. The NdSe2 catalyst likely functions by coordinating with the carbonyl oxygen of the activated ester and the nitrogen of the incoming amine, thereby lowering the activation energy for the nucleophilic attack while simultaneously stabilizing the transition state. This coordination sphere effectively shields the reactive intermediates from solvent interference and suppresses the formation of oligomeric by-products. The patent data explicitly highlights that omitting NdSe2 leads to a scenario where side reaction rates exceed the main reaction rate, resulting in a crude product with purity as low as 89.12%. In contrast, the catalyzed pathway ensures that the main coupling reaction dominates, facilitating the formation of the desired amide bond with high fidelity and minimal impurity generation.
Furthermore, the choice of solvent and reaction conditions plays a pivotal role in the mechanistic success of this protocol. The use of protic solvents like methanol or ethanol, which are typically avoided in peptide coupling due to the risk of transesterification, is rendered viable here due to the specific catalytic environment created by NdSe2. The catalyst appears to direct the nucleophilicity of the proline sodium salt specifically towards the activated carbonyl of the Fmoc-proline ester, outcompeting the solvent molecules. This selectivity allows for the use of inexpensive, environmentally benign solvents that are easy to recover and recycle on an industrial scale. Additionally, the reaction proceeds efficiently at room temperature over a period of 8 to 72 hours, suggesting a kinetic profile that is manageable and safe for large-scale reactors. The subsequent acidification step, performed at a pH of ≤ 2, ensures the quantitative conversion of the intermediate sodium salt into the free acid form, precipitating the product in a highly pure crystalline state that requires minimal further processing.
How to Synthesize Fmoc-D-Pro-D-Pro-OH Efficiently
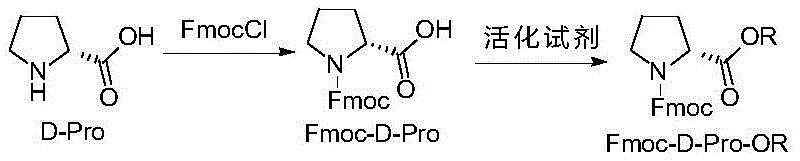
The synthesis of Fmoc-D-Pro-D-Pro-OH via this patented route is designed for operational simplicity and robustness, making it highly attractive for process chemists aiming to transfer technology from the lab to the pilot plant. The procedure begins with the preparation of the activated ester, Fmoc-D-Pro-OR, where R can be a succinimidyl (Su), benzotriazolyl (NB), or other leaving group, with the benzotriazolyl ester being particularly preferred for its balance of stability and reactivity. This activated species is then combined with D-Pro-Na and the NdSe2 catalyst in a suitable alcohol solvent. The reaction mixture is stirred at ambient temperature, allowing the coupling to proceed to completion without the need for complex thermal control systems. Following the reaction, a simple filtration step removes insoluble impurities, and the filtrate is subjected to acidification to isolate the product. The detailed standardized synthetic steps, including specific molar ratios and purification protocols, are provided in the guide below to ensure reproducibility and compliance with quality standards.
- Coupling Reaction: Mix Fmoc-D-Pro-OR activated ester, D-Pro-Na sodium salt, and NdSe2 catalyst in an alcoholic solvent at room temperature for 24 hours.
- Acidification and Extraction: Filter the reaction mixture, acidify the filtrate to pH ≤ 2 using hydrochloric acid, and extract the product into an organic phase like ethyl acetate.
- Purification: Concentrate the organic phase, induce crystallization with n-hexane, and perform sequential slurry purification with ether and water to achieve >98.5% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the NdSe2-catalyzed synthesis route offers compelling economic and logistical advantages that directly impact the bottom line. The most significant benefit is the drastic reduction in raw material costs achieved by eliminating the need for expensive polymeric resins and avoiding the massive excess of activated esters required in solid-phase synthesis. In traditional methods, reagent waste is a major cost driver, but this new liquid-phase approach operates with much higher atom economy, utilizing stoichiometric amounts of reactants more efficiently. Moreover, the ability to conduct the reaction at room temperature translates into substantial energy savings, as there is no requirement for energy-intensive heating mantles or cryogenic cooling units. This reduction in utility consumption, combined with the use of common solvents like ethanol or methanol which are cheaper and easier to source than specialized aprotic solvents, results in a significantly lower cost of goods sold (COGS) for the final intermediate.
- Cost Reduction in Manufacturing: The elimination of solid-phase resins and the reduction in reagent excess lead to a profound decrease in material expenditures. By avoiding the deprotection-reprotection cycles typical of older liquid-phase methods, the process also saves on the consumption of Fmoc-Cl and other protecting group reagents. The simplified workflow reduces labor hours and reactor occupancy time, further driving down operational expenses and enhancing overall manufacturing profitability.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as ethanol, hydrochloric acid, and commercially available proline salts ensures a stable and resilient supply chain. Unlike specialized resins or exotic coupling reagents that may face availability bottlenecks, the raw materials for this process are widely sourced from multiple global suppliers. This diversification mitigates the risk of supply disruptions and allows for more flexible procurement strategies, ensuring continuous production schedules even during market volatility.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from gram-scale laboratory experiments to multi-ton commercial production without the engineering challenges associated with solid-phase filtration and swelling. The use of greener solvents and the generation of fewer hazardous by-products simplify waste treatment protocols, aiding in compliance with increasingly stringent environmental regulations. This ease of scale-up ensures that supply can be rapidly ramped up to meet surging demand for peptide therapeutics without compromising quality or safety.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These answers are derived directly from the experimental data and technical specifications provided in patent CN110156873B, offering clarity on the practical aspects of adopting this technology. Understanding these details is crucial for R&D teams evaluating the feasibility of the process and for quality assurance personnel establishing control strategies for the new manufacturing method.
Q: Why is Neodymium Selenide (NdSe2) critical in this synthesis?
A: According to patent CN110156873B, NdSe2 acts as a crucial catalyst that suppresses side reactions. Without it, the reaction yields drop and purity falls below 90%, whereas the catalyzed route achieves over 98.5% purity.
Q: How does this method compare to traditional solid-phase synthesis?
A: Traditional solid-phase synthesis requires expensive resins and large excesses of reagents, often resulting in yields below 30%. This new liquid-phase method eliminates resin costs and operates with stoichiometric efficiency, significantly reducing material costs.
Q: What are the storage and handling requirements for the catalyst?
A: The process utilizes Neodymium Selenide under mild room temperature conditions. While specific handling protocols depend on the grade, the reaction itself avoids extreme temperatures or pressures, simplifying equipment requirements for scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fmoc-D-Pro-D-Pro-OH Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of next-generation peptide therapeutics. Our technical team has thoroughly analyzed the NdSe2-catalyzed pathway described in CN110156873B and possesses the expertise to implement this advanced chemistry at an industrial scale. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of Fmoc-D-Pro-D-Pro-OH. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch meets the >98.5% purity benchmark required for sensitive peptide synthesis applications. We are committed to delivering not just a chemical product, but a comprehensive supply solution that supports your drug development timeline.
We invite pharmaceutical partners to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By switching to our optimized manufacturing process, you can realize significant reductions in procurement costs while securing a supply chain that is both robust and compliant. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us demonstrate how our mastery of rare-earth catalyzed peptide coupling can become a strategic advantage for your organization, driving efficiency and quality in your peptide manufacturing operations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
