Scalable Synthesis of 4-Chloro-2-methylbenzoic Acid: A Breakthrough in Organolithium Carboxylation Technology
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to access complex aromatic building blocks with high efficiency and purity. A pivotal advancement in this domain is detailed in Chinese patent CN1310700A, which discloses a highly effective preparation method for o-alkylated benzoic acid derivatives. This technology addresses a long-standing synthetic challenge by enabling the direct conversion of aryl bromides into valuable benzoic acid structures using secondary or tertiary organolithium compounds in the presence of carbon dioxide. Unlike traditional approaches that often necessitate expensive aryl iodides or suffer from poor conversion rates, this innovation leverages the unique reactivity profile of sterically hindered organolithium reagents. For global procurement and R&D teams, understanding this specific carboxylation pathway is essential, as it unlocks a cost-effective route to critical intermediates like 4-chloro-2-methylbenzoic acid, a key precursor in the synthesis of potent Na+/H+ antiporter inhibitors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of ortho-alkylated benzoic acid derivatives has been plagued by significant inefficiencies when relying on standard lithiation protocols. Classical organic synthesis literature, such as the works by H. Gilman, describes the lithiation of aromatic bromides followed by carboxylation; however, these methods typically yield disappointing results when applied to substrates containing sensitive functional groups or specific substitution patterns. For instance, attempting to synthesize 4-chloro-2-methylbenzoic acid using conventional primary organolithium reagents like n-butyllithium or methyllithium often results in yields ranging from 0% to 30%, rendering the process economically unviable for industrial scale-up. Furthermore, prior art solutions frequently resorted to the use of aryl iodides, such as 2-iodo-4-chlorotoluene, to achieve acceptable reactivity. While chemically effective, aryl iodides are considerably more expensive and less atom-economical than their bromide counterparts, creating a substantial cost burden for large-scale manufacturing operations that cannot be ignored by supply chain managers.
The Novel Approach
The methodology outlined in CN1310700A represents a paradigm shift by demonstrating that aryl bromides can be successfully metallated and carboxylated with exceptional efficiency when secondary or tertiary organolithium compounds are employed. By utilizing reagents such as sec-butyllithium, the process achieves yields as high as 90% for the conversion of 2-bromo-5-chlorotoluene to 4-chloro-2-methylbenzoic acid. This breakthrough eliminates the economic necessity of using aryl iodides, allowing manufacturers to leverage the lower cost and wider availability of aryl bromides without compromising on output. The reaction operates under mild conditions, typically between -100°C and +50°C, and utilizes common inert solvents like methyl tert-butyl ether (MTBE) or tetrahydrofuran (THF). This operational simplicity, combined with the dramatic improvement in yield, establishes a superior technical foundation for the reliable production of high-purity pharmaceutical intermediates.
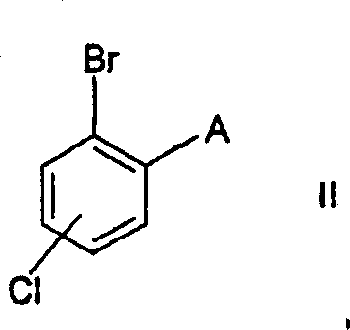
Mechanistic Insights into Sec-Butyllithium Catalyzed Carboxylation
The success of this novel synthetic route hinges on the precise mechanistic interaction between the aryl bromide substrate and the secondary organolithium metallation agent. When sec-butyllithium is introduced to the reaction mixture, it facilitates a rapid and selective lithium-halogen exchange or direct ortho-lithiation, depending on the specific substrate electronics, generating a highly reactive aryl lithium intermediate. Unlike primary organolithiums, which may promote unwanted side reactions such as Wurtz-type coupling or nucleophilic attack on the alkyl substituents, the steric bulk of the sec-butyl group modulates the basicity and nucleophilicity of the reagent. This modulation ensures that the metallation occurs selectively at the desired position adjacent to the alkyl group, preserving the integrity of the chloro-substituent on the aromatic ring. The subsequent introduction of carbon dioxide acts as an electrophile, trapping the aryl lithium species to form the carboxylate salt, which is then protonated during the workup phase to yield the final benzoic acid derivative.
From an impurity control perspective, the choice of sec-butyllithium significantly minimizes the formation of by-products that typically complicate downstream purification. In conventional processes using n-butyllithium, the low yields are often accompanied by complex impurity profiles resulting from incomplete reaction or decomposition of the intermediate. In contrast, the high conversion rates observed with sec-butyllithium (70%-90%) mean that the crude product contains a much higher proportion of the desired 4-chloro-2-methylbenzoic acid. This purity advantage simplifies the isolation process, often allowing for direct precipitation via acidification without the need for energy-intensive and solvent-heavy recrystallization steps. For R&D directors focused on impurity profiling, this cleaner reaction profile translates to a more robust and predictable manufacturing process that meets stringent quality specifications for pharmaceutical applications.
How to Synthesize 4-Chloro-2-methylbenzoic Acid Efficiently
Implementing this advanced carboxylation strategy requires strict adherence to the optimized parameters defined in the patent to ensure maximum yield and safety. The process involves dissolving the aryl bromide starting material, specifically 2-bromo-5-chlorotoluene, in an inert solvent such as MTBE and cooling the mixture to cryogenic temperatures, typically around -18°C. The careful addition of sec-butyllithium is critical, followed by the controlled introduction of the reaction mixture into a stream of carbon dioxide-saturated solvent. This specific sequence of addition and temperature management is designed to stabilize the reactive intermediates and prevent thermal runaway or decomposition. While the general concept of organolithium chemistry is well-known, the specific operational details regarding stoichiometry, addition rates, and quenching protocols provided in the patent are essential for successful replication. The detailed standardized synthesis steps for this high-efficiency route are provided in the guide below.
- Dissolve 2-bromo-5-chlorotoluene in methyl tert-butyl ether (MTBE) and cool the solution to approximately -18°C.
- Add sec-butyllithium dropwise to the cooled solution over 30 minutes under inert atmosphere to effect lithiation.
- Transfer the reaction mixture into CO2-saturated MTBE, maintain temperature between -15°C and -5°C, and saturate again with gaseous CO2.
- Quench the reaction with aqueous NaOH, separate phases, acidify the aqueous layer with HCl to pH 1-2, and isolate the precipitated product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this sec-butyllithium-mediated carboxylation process offers tangible strategic benefits that extend beyond simple chemical yield. The primary advantage lies in the raw material cost structure; by validating the use of aryl bromides over aryl iodides, manufacturers can access a significantly cheaper and more abundant feedstock supply chain. Aryl bromides are commodity chemicals with stable pricing, whereas aryl iodides often face supply volatility and premium pricing due to the cost of iodine. Furthermore, the high selectivity of the reaction reduces the consumption of solvents and energy associated with extensive purification procedures, contributing to a leaner and more sustainable manufacturing footprint. These factors collectively enhance the overall cost competitiveness of the final pharmaceutical intermediate.
- Cost Reduction in Manufacturing: The elimination of expensive aryl iodide precursors serves as a major driver for cost optimization in fine chemical manufacturing. By switching to aryl bromides, the direct material cost is drastically reduced without the penalty of low yields that previously plagued bromide-based routes. Additionally, the high yield of 90% minimizes waste generation and maximizes the throughput of existing reactor capacity, effectively lowering the cost per kilogram of the active intermediate. The ability to avoid complex purification steps like repeated recrystallization further reduces utility costs and solvent procurement expenses, creating a compound positive effect on the overall production economics.
- Enhanced Supply Chain Reliability: Relying on widely available aryl bromides mitigates the risk of supply chain disruptions associated with specialized or niche reagents. Since 2-bromo-5-chlorotoluene and similar bromides are produced on a large industrial scale, securing long-term supply contracts is far more feasible compared to sourcing specific aryl iodides. This stability ensures consistent production schedules and reduces the lead time for high-purity pharmaceutical intermediates, allowing downstream drug manufacturers to plan their inventory with greater confidence. The robustness of the process also means that production is less susceptible to batch failures caused by reagent variability, ensuring a steady flow of materials to the market.
- Scalability and Environmental Compliance: The process is inherently scalable, having been designed for industrial application with a one-pot synthesis approach that simplifies equipment requirements. The use of common solvents like MTBE and the avoidance of heavy metal catalysts align well with modern environmental regulations and green chemistry principles. Reduced solvent usage for purification and higher atom economy contribute to a lower environmental impact, facilitating easier regulatory approval and compliance with increasingly strict waste disposal standards. This scalability ensures that the transition from laboratory pilot batches to multi-ton commercial production can be achieved smoothly and safely.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this organolithium carboxylation technology. These answers are derived directly from the experimental data and comparative examples provided in patent CN1310700A, offering clarity on why this method outperforms historical precedents. Understanding these nuances is vital for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Why is sec-butyllithium preferred over n-butyllithium for this synthesis?
A: According to patent CN1310700A, conventional primary organolithium reagents like n-butyllithium result in negligible yields (0-30%) for this specific substrate. Secondary organolithiums like sec-butyllithium significantly improve yields up to 90% by optimizing the metallation kinetics and reducing side reactions.
Q: Can aryl iodides be used instead of aryl bromides?
A: While aryl iodides are more reactive, they are substantially more expensive. This novel process specifically enables the use of cheaper and more readily available aryl bromides (like 2-bromo-5-chlorotoluene) without sacrificing yield, offering a distinct economic advantage.
Q: What are the downstream applications of 4-chloro-2-methylbenzoic acid?
A: This intermediate is critical for synthesizing Na+/H+ antiporter inhibitors, specifically N-diaminomethylene-2-methyl-4-(1-pyrrolyl)-5-methylsulfonylbenzamide, which has significant pharmaceutical relevance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Chloro-2-methylbenzoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthetic routes in the competitive landscape of pharmaceutical intermediate production. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN1310700A are fully realized in practical, large-scale operations. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 4-chloro-2-methylbenzoic acid meets the exacting standards required for downstream drug synthesis. Our commitment to process excellence allows us to deliver high-purity intermediates that support the development of life-saving medications.
We invite global partners to collaborate with us to leverage this advanced technology for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your project benefits from the most cost-effective and reliable sourcing strategy available in the market.
