Advanced Synthesis of Tetramisole Intermediates: A Breakthrough in Yield and Process Simplicity for Global Supply Chains
Advanced Synthesis of Tetramisole Intermediates: A Breakthrough in Yield and Process Simplicity for Global Supply Chains
The pharmaceutical and veterinary industries are constantly seeking more efficient pathways to produce broad-spectrum anthelmintics, and the recent disclosure in patent CN112194639A presents a significant technological leap forward in the synthesis of tetramisole intermediates. This patent details a novel preparation method for the hydroxyl salt of 2-imino-3-(2-hydroxy-2-phenyl)ethyl thiazoline, a critical precursor in the manufacture of tetramisole and its active levamisole resolution. Unlike legacy methods that struggle with low conversion rates and complex purification burdens, this innovative approach leverages a direct acetophenone-based condensation strategy that streamlines the entire production workflow. By fundamentally re-engineering the bond formation steps, the process achieves a remarkable yield improvement, pushing production efficiency to levels previously unattainable with conventional styrene oxide routes. For global procurement leaders and R&D directors, this represents not just a chemical optimization, but a strategic opportunity to secure a more stable and cost-effective supply of high-value veterinary and pharmaceutical actives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of tetramisole intermediates has relied heavily on the styrene oxide-ethanolamine addition route, a method that, while established, is fraught with significant inefficiencies that impact both cost and environmental footprint. The primary drawback of this traditional pathway is the inherent formation of isomers during the reaction process, which necessitates a complex and energy-intensive hydrolysis rearrangement step to convert these unwanted byproducts into the target molecule. Furthermore, the overall yield of the hydroxyl salt in these conventional processes typically hovers around 60%, meaning that nearly half of the valuable starting materials are lost to waste streams or require expensive recycling protocols. This low atom economy translates directly into higher raw material costs and increased pressure on waste treatment facilities, creating a bottleneck for manufacturers aiming to scale production sustainably. Additionally, the reliance on styrene oxide introduces safety and handling concerns due to its reactivity and toxicity, further complicating the operational safety profile of the manufacturing plant.
The Novel Approach
In stark contrast, the methodology outlined in the patent utilizes acetophenone as the foundational building block, initiating a reaction sequence that bypasses the problematic isomerization issues entirely. By reacting acetophenone with an inorganic base and 2-chloroethylamine hydrochloride in an aprotic solvent, the process generates the necessary amino-alcohol intermediate with exceptional selectivity and minimal byproduct formation. This strategic shift allows the subsequent cyclization with thiourea to proceed with much higher fidelity, resulting in a final hydroxyl salt yield that consistently exceeds 90%. The simplicity of the work-up procedure, which involves straightforward filtration and crystallization rather than complex chromatographic separations, drastically reduces the processing time and solvent consumption. This novel approach effectively decouples production capacity from the limitations of legacy chemistry, offering a robust platform for cost reduction in API manufacturing while simultaneously enhancing the purity profile of the final intermediate to over 99%.
Mechanistic Insights into Acetophenone-Based Condensation and Cyclization
The core of this technological advancement lies in the precise orchestration of nucleophilic substitution and heterocyclic ring closure under controlled thermal conditions. The first stage involves the generation of an enolate or activated ketone species from acetophenone using a strong inorganic base such as potassium hydroxide or sodium carbonate in solvents like acetonitrile or DMF. This activated species then undergoes nucleophilic attack by the amine group of 2-chloroethylamine, forming the carbon-nitrogen bond that establishes the side chain required for the thiazoline ring. The reaction is maintained at temperatures between 80°C and 120°C to ensure complete conversion while minimizing degradation, resulting in an oily intermediate compound that is isolated via reduced pressure concentration. This step is critical because it sets the stereochemical and structural foundation for the final product, ensuring that the hydroxyl group and the amine chain are correctly positioned for the subsequent cyclization event without the formation of regio-isomers.
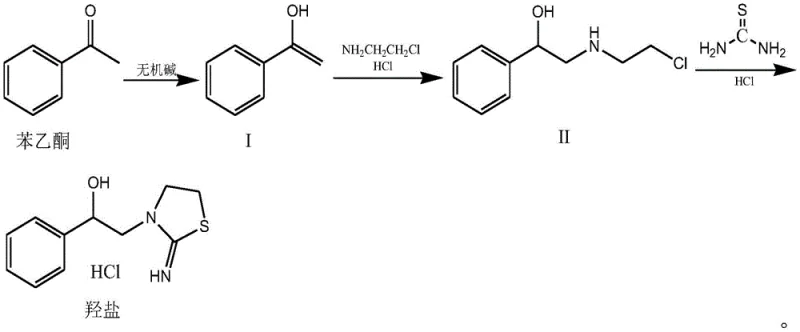
Following the formation of the amino-alcohol intermediate, the process transitions into the cyclization phase where thiourea acts as the sulfur and nitrogen source for the thiazoline ring construction. The reaction mixture is acidified to a pH range of 0.5 to 1.5 using hydrochloric acid, which protonates the amine and activates the thiourea for nucleophilic attack on the adjacent carbon centers. Heating the mixture to an internal temperature of 108-110°C facilitates the distillation of water and drives the equilibrium towards the formation of the cyclic thiazoline structure. This high-temperature reflux condition is essential for overcoming the activation energy barrier of the ring closure, ensuring that the reaction proceeds to completion within a 6 to 10-hour window. The result is a highly crystalline hydrochloride salt that can be easily purified by washing with ethanol, demonstrating a level of impurity control that is superior to the messy reaction profiles of older methods.
How to Synthesize Tetramisole Intermediate Hydroxyl Salt Efficiently
Implementing this synthesis route requires careful attention to solvent selection and temperature ramping to maximize the yield benefits described in the patent data. The process is designed to be operationally simple, utilizing standard reactor equipment capable of handling elevated temperatures and vacuum distillation, making it accessible for most fine chemical manufacturing facilities. Operators must ensure that the molar ratios of the inorganic base to acetophenone are maintained between 1.1:1 and 2.2:1 to fully drive the initial condensation without excessive base residue. Detailed standardized synthesis steps, including specific charging orders and cooling rates for crystallization, are provided in the technical guide below to ensure reproducibility at scale.
- React acetophenone with an inorganic base and 2-chloroethylamine hydrochloride in an aprotic solvent at 80-120°C to form the amino-alcohol intermediate.
- Dissolve the resulting oily compound in water, adjust pH to 0.5-1.5 with hydrochloric acid, and add thiourea.
- Heat the mixture to 108-110°C for reflux and distillation, then cool to crystallize the final hydroxyl salt product with >90% yield.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers compelling economic and logistical advantages that extend far beyond simple yield metrics. By shifting to a process that eliminates the need for expensive chiral catalysts or complex rearrangement steps, manufacturers can achieve significant cost savings in raw material procurement and utility consumption. The use of commodity chemicals like acetophenone and thiourea ensures that the supply chain is not vulnerable to the volatility often associated with specialized reagents, thereby enhancing the reliability of the pharmaceutical intermediates supplier network. Furthermore, the high purity of the crude product (>97% by GC analysis before final crystallization) reduces the burden on downstream purification units, allowing for faster batch turnover and improved asset utilization rates across the production facility.
- Cost Reduction in Manufacturing: The elimination of the isomer rearrangement step found in traditional styrene oxide routes removes a major cost center associated with energy consumption and extended reaction times. By achieving yields greater than 90% compared to the historical 60%, the effective cost per kilogram of the active intermediate is drastically lowered, as less raw material is wasted and fewer batches are needed to meet production targets. Additionally, the avoidance of transition metal catalysts means there is no need for expensive heavy metal scavenging resins or rigorous testing for residual metals, further simplifying the cost structure and reducing the financial risk of batch rejection due to specification failures.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials such as acetophenone and inorganic bases like potassium carbonate ensures that production is not held hostage by the supply constraints of niche reagents. This commoditization of the input stream allows for reducing lead time for high-purity veterinary drug precursors, as suppliers can maintain larger safety stocks of raw materials without fear of obsolescence. The robustness of the reaction conditions, which tolerate a range of aprotic solvents including acetonitrile and DMF, provides flexibility in sourcing; if one solvent becomes scarce or expensive due to market fluctuations, the process can be adapted to use alternatives without compromising the integrity of the final product.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from laboratory glassware to multi-ton industrial reactors without the need for specialized high-pressure equipment or cryogenic cooling. The simplified work-up procedure, which relies on filtration and crystallization rather than extractive work-ups with large volumes of organic solvents, significantly reduces the generation of hazardous waste streams. This aligns with modern green chemistry principles and helps manufacturers meet increasingly stringent environmental regulations, avoiding potential fines and shutdowns while positioning the company as a sustainable partner for commercial scale-up of complex anthelmintic intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis pathway, drawing directly from the experimental data and claims presented in the patent documentation. Understanding these nuances is critical for technical teams evaluating the feasibility of technology transfer and for commercial teams negotiating supply agreements based on this new standard of quality. The answers provided reflect the specific advantages of the acetophenone route over legacy methods, focusing on yield, purity, and operational simplicity.
Q: How does this new acetophenone route compare to the traditional styrene oxide method?
A: The traditional styrene oxide-ethanolamine addition route typically yields around 60% and suffers from isomer formation requiring complex hydrolysis rearrangement. In contrast, the patented acetophenone route achieves yields exceeding 90% with purity over 99%, eliminating the need for difficult isomer separation and significantly reducing energy consumption.
Q: What are the critical reaction conditions for the cyclization step?
A: The cyclization with thiourea requires precise control of acidity and temperature. The process mandates adjusting the pH to between 0.5 and 1.5 using hydrochloric acid and maintaining a reaction temperature of 108-110°C during the distillation and reflux phase to ensure complete conversion to the thiazoline ring structure.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is specifically designed for industrial scalability. It utilizes common, commercially available raw materials like acetophenone and thiourea, avoids expensive transition metal catalysts, and relies on standard unit operations such as filtration and crystallization, making it highly robust for metric-ton scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetramisole Intermediate Hydroxyl Salt Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to a superior synthetic route requires a partner with deep technical expertise and proven manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a GMP-compliant environment. Our state-of-the-art facilities are equipped with rigorous QC labs and stringent purity specifications, guaranteeing that every batch of tetramisole intermediate meets the highest standards required for human and veterinary pharmaceutical applications. We understand the critical nature of supply continuity in the anthelmintic market and are committed to delivering consistent quality that supports your regulatory filings and commercial launch timelines.
We invite you to engage with our technical procurement team to discuss how this advanced manufacturing process can be integrated into your supply chain to drive efficiency and reduce costs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic impact of switching to this high-yield route. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions based on hard data and our proven track record of excellence in fine chemical synthesis.
