Scalable Synthesis of Cefpirome Intermediate: A Technical Breakthrough for API Manufacturing
Introduction to Advanced Cefpirome Intermediate Manufacturing
The pharmaceutical landscape for fourth-generation cephalosporins demands increasingly efficient and environmentally sustainable synthetic routes. Patent CN102391288B introduces a transformative preparation method for the key cefpirome intermediate, specifically the halogen acid salt of (6R, 7R)-7-amino-3-[(2,3-cyclopentene-pyridine)methyl]ceph-3-ene-4-carboxylic acid. This technology addresses critical bottlenecks in traditional manufacturing, such as low yields, complex purification, and significant iodine waste. By leveraging a sophisticated sequence of silylation, iodination, and oxidative crystallization, this method delivers a high-purity intermediate essential for producing cefpirome sulfate, a potent broad-spectrum antibiotic. For R&D directors and procurement specialists, understanding this pathway is crucial for securing a reliable cefpirome intermediate supplier capable of meeting stringent quality and cost targets.
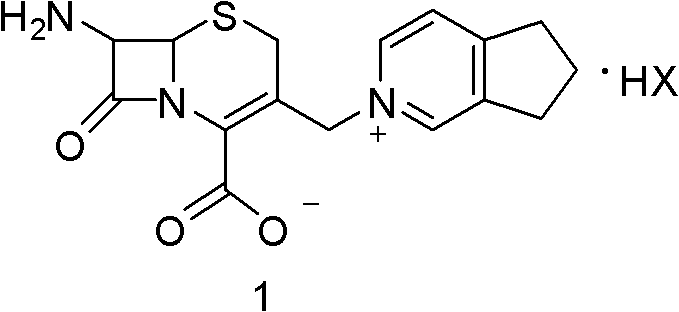
The structural integrity of the beta-lactam ring is paramount throughout this synthesis. The target molecule, often referred to as Compound 1 in technical literature, serves as the pivotal building block for the final active pharmaceutical ingredient. The innovation lies not just in the formation of the quaternary ammonium salt but in the strategic management of the protecting groups and the iodine byproducts. Traditional routes often struggle with the stability of the cephem nucleus under harsh acidic conditions required for deprotection. This patent outlines a refined approach that maintains stereochemical integrity while streamlining the isolation of the final salt form, ensuring that the resulting API precursor meets the rigorous specifications required for global regulatory submission.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods for synthesizing this critical intermediate have long been plagued by inefficiencies that drive up costs and complicate supply chains. Historically, the preparation involved converting 7-amino-cephalosporanic acid into a dihydriodate, which was considered second-rate quality. To make this usable for the next synthetic step, manufacturers were forced to employ tedious anion-exchange resin column chromatography to purify and convert the dihydriodate into the desired dihydrochloride. This additional unit operation is not only time-consuming but also introduces significant yield losses due to adsorption and elution inefficiencies. Furthermore, the conventional process disperses expensive iodine into the waste liquid as iodide ions, making recovery difficult and leading to substantial raw material wastage. The environmental burden of treating iodine-rich wastewater, combined with the high cost of iodine which can account for 15-20% of production costs, renders these older methods economically unsustainable for modern large-scale operations.
The Novel Approach
The methodology disclosed in CN102391288B represents a paradigm shift by integrating deprotection and crystallization into a synchronized, single-step operation. Instead of relying on resin columns, the novel process utilizes an oxidant to convert iodide ions directly into elemental iodine, which partitions into the organic phase. This clever chemical maneuver allows for the direct crystallization of the high-purity mono-hydrochloride or hydriodate monohydrate from the reaction mixture. By eliminating the need for column chromatography and reducing the volume of crystallization solvents, the process drastically simplifies the workflow. The result is a robust manufacturing route that minimizes the discharge of three wastes (waste water, gas, and solids) and facilitates the easy recovery of iodine. This approach not only enhances the overall molar yield to over 91% but also ensures a purity profile exceeding 98%, making it immediately suitable for downstream acylation without further purification.
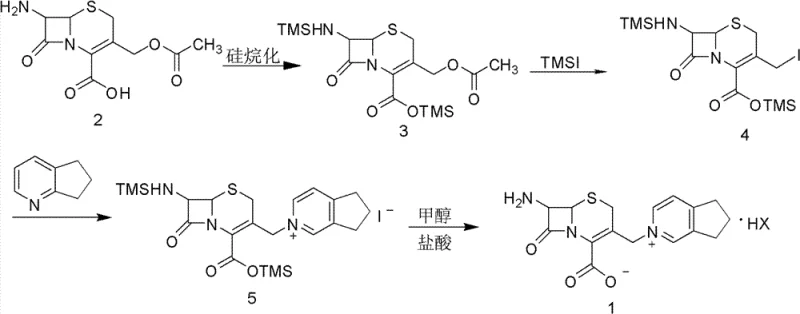
Mechanistic Insights into Silylation and Oxidative Crystallization
The core of this synthetic strategy relies on a precise sequence of protection and activation steps. Initially, 7-amino-cephalosporanic acid undergoes silylation using reagents such as hexamethyldisilazane (HMDS) or N,O-bis(trimethylsilyl)acetamide (BSA) in a methylene chloride solvent. This step protects both the amino and carboxyl groups, forming the bis-silylated intermediate (Compound 3), which is crucial for preventing side reactions during the subsequent iodination. Following this, trimethylsilyl iodide (TMSI) is introduced to effect the iodination at the 3-position of the cephem ring. The presence of an acid binding agent, such as N,N-diethylaniline or triethylamine, is vital here to neutralize the acid generated and drive the equilibrium forward. The subsequent addition of 2,3-cyclopentenopyridine results in a quaternization reaction, attaching the essential pyridinium side chain to form Compound 5. This multi-step cascade is performed with tight temperature control, typically maintaining the quaternization between 5-10°C to preserve the delicate beta-lactam structure.
The true genius of this mechanism, however, is revealed in the workup phase. In traditional methods, the removal of silyl groups and the separation of iodine byproducts are distinct, messy operations. Here, the addition of an oxidant—preferably hydrogen peroxide (40-50wt%) or ferric chloride—serves a dual purpose. First, it facilitates the deprotection of the silyl groups in the presence of methanol and acid. Second, and more critically, it oxidizes the free iodide ions (I-) generated during the reaction back into elemental iodine (I2). Since elemental iodine is highly soluble in organic solvents but insoluble in the aqueous acidic phase where the product salt crystallizes, this allows for a clean phase separation. The product precipitates as a high-purity salt, while the valuable iodine remains in the organic layer for recovery. This mechanistic insight explains how the process achieves such high purity (>98%) and yield while simultaneously solving the environmental issue of iodine waste.
How to Synthesize Cefpirome Intermediate Efficiently
Implementing this synthesis requires strict adherence to the optimized parameters regarding reagent ratios and temperature profiles to maximize yield and safety. The process begins with the reflux of 7-amino-cephalosporanic acid with HMDS, followed by the controlled addition of TMSI and the pyridine derivative. The critical innovation occurs during the quenching and crystallization stage, where the choice of oxidant and acid concentration dictates the final crystal form and purity. Operators must carefully manage the addition of methanol and the oxidant to ensure complete deprotection without degrading the cephem nucleus. For a detailed breakdown of the specific operational parameters, reagent quantities, and safety precautions required to execute this synthesis in a GMP environment, please refer to the standardized protocol below.
- Protect 7-amino-cephalosporanic acid using hexamethyldisilazane (HMDS) in methylene chloride under reflux to form the silylated intermediate.
- React the silylated intermediate with trimethylsilyl iodide (TMSI) and 2,3-cyclopentenopyridine to introduce the pyridinium side chain.
- Add an oxidant like hydrogen peroxide and hydrochloric acid to deprotect, oxidize iodide ions, and crystallize the final mono-hydrochloride salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented technology translates into tangible strategic advantages beyond mere technical elegance. The primary driver of value is the drastic reduction in raw material costs, specifically regarding iodine. In conventional processes, iodine is effectively lost to the wastewater stream, representing a recurring and volatile cost center. By enabling the recovery of iodine through oxidation, this method stabilizes the bill of materials and insulates the production cost from fluctuations in iodine market prices. Furthermore, the elimination of the anion-exchange resin column removes a significant bottleneck in the production schedule. Resin columns require regeneration, have limited lifespans, and slow down batch turnover. Removing this step accelerates the manufacturing cycle time, allowing for increased throughput without expanding facility footprint.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived from fundamental changes in the material flow rather than marginal efficiency tweaks. By recovering expensive iodine reagents that would otherwise be discarded as waste, the net consumption of raw materials is significantly lowered. Additionally, the process avoids the use of large volumes of acetone and other solvents typically required for multiple recrystallization steps in older methods. The reduction in solvent usage lowers both the direct purchase cost of chemicals and the indirect costs associated with solvent recovery and distillation. Moreover, the simplified workflow reduces labor hours and utility consumption per kilogram of product, contributing to a leaner cost structure that enhances competitiveness in the global API market.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by complex processes that have multiple points of failure. The traditional reliance on ion-exchange resins introduces a dependency on consumable materials that can degrade or vary in performance, potentially leading to batch failures. The new method's reliance on standard chemical reagents and straightforward crystallization makes the supply chain more robust. The simplified post-processing also means that equipment cleaning and changeover times are reduced, increasing the available capacity for production. This reliability is crucial for meeting the just-in-time delivery expectations of downstream pharmaceutical manufacturers who cannot afford interruptions in their antibiotic production lines.
- Scalability and Environmental Compliance: As regulatory pressure on pharmaceutical manufacturing intensifies, the environmental profile of a synthesis route becomes a key qualification criterion. This method's ability to concentrate iodine waste for easy recovery and its reduction in overall solvent discharge make it inherently more compliant with strict environmental regulations. The process generates less hazardous waste, simplifying the permitting process for plant expansion. From a scalability perspective, the reaction conditions are mild and do not require exotic catalysts or extreme pressures, making the transfer from pilot scale to commercial tonnage straightforward. This ease of scale-up ensures that suppliers can rapidly respond to surges in demand for cefpirome without compromising on quality or safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. They are derived from the specific improvements highlighted in the patent data, focusing on purity, yield, and operational simplicity. Understanding these details helps stakeholders evaluate the feasibility of adopting this technology for their own supply chains or assessing the quality of intermediates sourced from partners utilizing this method.
Q: How does this new method improve iodine recovery compared to traditional processes?
A: Traditional methods often lose iodine as iodide ions in waste liquid. This novel process uses an oxidant (e.g., hydrogen peroxide) to convert iodide ions back to elemental iodine, which concentrates in the organic phase, allowing for efficient recovery and significantly reducing raw material costs and environmental impact.
Q: What is the purity level achievable with this synthetic route?
A: The method described in patent CN102391288B achieves a molar yield greater than 91% and a product purity exceeding 98% for the intermediate. The subsequent cefpirome sulfate produced from this intermediate demonstrates HPLC purity greater than 99%, eliminating the need for complex purification steps like anion-exchange resin columns.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is designed for industrial suitability. It simplifies post-processing by combining deprotection and crystallization, reduces the use of massive amounts of crystallization solvents, and minimizes three-waste discharge, making it highly scalable and environmentally compliant for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefpirome Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent laboratory data to commercial reality requires deep technical expertise and robust infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this novel synthesis are fully realized in practice. We maintain stringent purity specifications and operate rigorous QC labs equipped to verify the identity and potency of every batch, guaranteeing that our cefpirome intermediates meet the exacting standards required for fourth-generation cephalosporin manufacturing. Our commitment to quality assurance ensures that the high purity (>98%) and yield (>91%) reported in the patent are consistently delivered to our clients.
We invite pharmaceutical partners to leverage our technical capabilities to optimize their antibiotic supply chains. Whether you are looking to reduce costs in cefpirome manufacturing or secure a stable source of high-purity intermediates, our team is ready to assist. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our advanced manufacturing processes can support your long-term strategic goals.
