Scalable Synthesis of 4β-Amino-4'-demethyl-4-deoxypodophyllotoxin for Oncology Intermediates
The pharmaceutical industry continuously seeks robust synthetic routes for complex oncology intermediates, particularly those derived from the podophyllotoxin scaffold. Patent CN101223176B introduces a transformative methodology for preparing 4β-amino-4'-demethyl-4-deoxypodophyllotoxin, a critical precursor for next-generation anticancer agents. This technology addresses long-standing challenges in stereochemical control and process safety by replacing hazardous azide chemistry with a controlled thiourea-mediated cleavage of haloacetamido derivatives. The innovation lies not merely in the chemical transformation but in the strategic manipulation of solvent systems to drive equilibrium towards the desired amine without degradation. For R&D directors and procurement specialists, this represents a significant opportunity to streamline supply chains for high-value pharmaceutical intermediates. By leveraging this specific synthetic pathway, manufacturers can achieve molar yields exceeding 90% while bypassing the costly and time-consuming chromatographic purification steps that have historically plagued this chemical class.
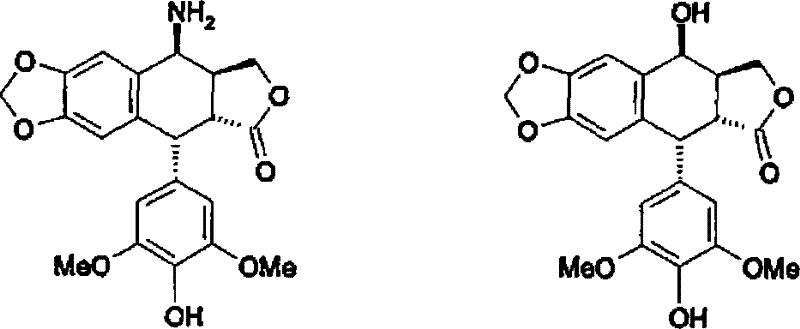
The evolution of synthetic strategies for 4-position modified podophyllotoxins highlights the limitations of conventional methodologies compared to this novel approach. Historically, the preparation of the target amino derivative relied heavily on the conversion of 4'-demethylepipodophyllotoxin into a 4β-azido intermediate, followed by catalytic reduction. While literature precedents such as J. Med. Chem. 1991 demonstrated partial success using sodium azide and trifluoroacetic acid, these methods suffered from poor stereoselectivity and the generation of alpha/beta mixtures. Furthermore, the reliance on azide chemistry introduces severe safety liabilities, including the risk of explosion during large-scale handling, which is unacceptable for modern GMP facilities. Even when stereoselectivity was improved using BF3 etherate or samarium iodide, the processes invariably required one or even two stages of chromatographic purification to isolate the product in acceptable purity. This dependency on chromatography creates a bottleneck in commercial scale-up of complex pharmaceutical intermediates, drastically increasing production costs and limiting throughput capacity.
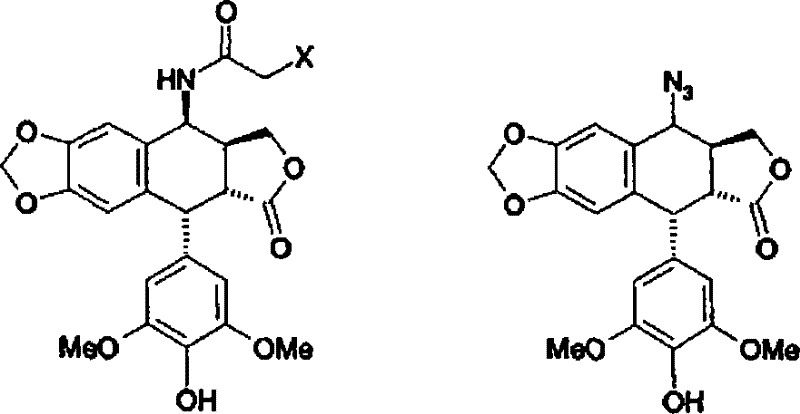
In stark contrast, the novel approach detailed in the patent utilizes a 4β-haloacetamido-4'-demethyl-4-deoxypodophyllotoxin intermediate, which is inherently safer and more stable than its azido counterpart. The breakthrough involves cleaving the chloroacetamido group using thiourea in a specifically defined acidic medium. Unlike classical organic synthesis teachings which suggest using ethanol and acetic acid mixtures, this invention discovered that such solvent systems are detrimental to the reaction progress. In traditional ethanol/acetic acid mixtures, the reaction stalls at the S-isothiourea salt stage, resulting in less than 10% conversion of the starting material even after prolonged reflux. The novel method overcomes this by employing pure weak acids, specifically glacial acetic acid, as the sole reaction medium. This adjustment prevents the stabilization of the unreactive intermediate salt, allowing the reaction to proceed to completion within just 2 hours at 80°C. The result is a direct precipitation of the product, which can be isolated via simple filtration, offering a dramatic improvement in operational efficiency and cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Thiourea-Mediated Amide Cleavage
Understanding the mechanistic nuances of this transformation is vital for R&D teams aiming to replicate or optimize the process. The core reaction involves the nucleophilic attack of thiourea on the carbonyl carbon of the haloacetamido group, leading to the displacement of the halide and the formation of an S-alkylisothiourea intermediate. In standard solvent systems containing water or alcohols, this S-alkylisothiourea salt becomes thermodynamically stable and resistant to further hydrolysis or aminolysis, effectively trapping the reaction. The patent data reveals that the presence of water or ethanol facilitates the crystallization or stabilization of this salt, preventing the release of the free amine. However, in a pure weak acid environment like glacial acetic acid, the solvation dynamics change significantly. The high concentration of protons and the specific dielectric properties of the pure acid likely promote the rapid decomposition of the S-isothiourea intermediate into the desired amine and the corresponding byproduct. This mechanistic pathway ensures that the reaction does not stall, driving the equilibrium forward until the starting material is fully consumed.

Furthermore, the stereochemical integrity of the molecule is preserved throughout this cleavage process, which is a critical quality attribute for biological activity. The 4β-configuration, established in the preceding Ritter reaction step where the haloacetonitrile reacts with the epipodophyllotoxin, remains intact during the thiourea treatment. This retention of configuration is essential because the biological target, topoisomerase II, is highly sensitive to the stereochemistry at the C4 position. The purity profile achieved through this mechanism is exceptional, with the patent reporting purity levels greater than 95% directly after filtration and drying. This high purity is attributed to the selective precipitation of the product hydrochloride salt from the reaction medium, leaving soluble impurities and byproducts in the mother liquor. For quality control teams, this means a simplified impurity profile and reduced burden on analytical validation, facilitating faster regulatory filings for the final drug substance.
How to Synthesize 4β-Amino-4'-demethyl-4-deoxypodophyllotoxin Efficiently
The implementation of this synthesis route requires precise adherence to the specified reaction parameters to avoid the pitfalls of the prior art. The process begins with the suspension of the 4β-haloacetamido precursor in pure glacial acetic acid, ensuring no water or alcohol contamination is present. The mixture is then heated to a temperature range of 60°C to 100°C, with 80°C identified as the optimal setpoint for balancing reaction rate and product stability. Thiourea is added in a molar ratio of 0.5 to 1 relative to the substrate, and the reaction is maintained under agitation. The visual observation of dissolution followed by reprecipitation serves as a key process indicator, signaling the progression from the starting material to the product salt. Detailed standardized operating procedures for this synthesis are outlined below to ensure reproducibility and safety.
- Suspend 4β-haloacetamido-4'-demethyl-4-deoxypodophyllotoxin in pure glacial acetic acid and heat the mixture to 80°C.
- Add thiourea to the heated suspension and maintain stirring at 80°C for approximately 2 hours to ensure complete conversion.
- Filter the precipitated product hot, wash with glacial acetic acid and diisopropyl ether, and dry under vacuum to obtain the hydrochloride salt.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this thiourea cleavage methodology offers substantial advantages that directly impact the bottom line and supply chain resilience. The most significant benefit is the elimination of chromatographic purification, which is traditionally one of the most expensive and scale-limiting steps in fine chemical manufacturing. By relying on precipitation and filtration, the process reduces solvent consumption, waste generation, and processing time, leading to significant cost savings. Additionally, the avoidance of hazardous azide reagents removes the need for specialized explosion-proof equipment and rigorous safety protocols associated with energetic materials. This simplification of the safety profile allows for more flexible manufacturing scheduling and reduces the regulatory burden related to process safety management. Consequently, this route supports a more agile and responsive supply chain capable of meeting fluctuating market demands for oncology intermediates.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the removal of chromatography and the use of commodity chemicals. Traditional methods require large volumes of high-purity solvents for column loading and elution, alongside expensive silica gel or resin media, all of which contribute to high operational expenditures. In contrast, the new method utilizes glacial acetic acid, a low-cost bulk chemical, which can potentially be recovered and recycled. The high molar yield of over 90% further enhances cost efficiency by maximizing the output from each batch of starting material. These factors combine to lower the overall cost of goods sold (COGS), making the final API more competitive in the global marketplace without compromising on quality standards.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the reliance on specialized or hazardous reagents that may face shipping restrictions or availability issues. Azides, for instance, are subject to strict transport regulations due to their explosive nature, which can lead to delays and increased logistics costs. By substituting azides with stable haloacetamido precursors and thiourea, the supply chain becomes more robust and less susceptible to regulatory bottlenecks. The reagents used in this process are widely available from multiple global suppliers, reducing the risk of single-source dependency. This diversification of the supply base ensures consistent raw material availability, enabling manufacturers to maintain steady production schedules and fulfill long-term contracts with confidence.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to industrial production often reveals hidden inefficiencies, particularly regarding waste management. The precipitation-based isolation method described in this patent is inherently scalable, as filtration equipment can be easily sized up without the linear increase in complexity seen with chromatography. Moreover, the reduction in solvent usage and the absence of heavy metal catalysts (often used in azide reductions) align with green chemistry principles. This results in a lower environmental footprint, simplifying waste treatment and disposal compliance. For manufacturing sites operating under strict environmental regulations, this cleaner process profile reduces the risk of non-compliance penalties and supports corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for their specific production needs. The answers highlight the critical parameters that distinguish this method from conventional approaches and underscore its suitability for industrial application.
Q: Why is the thiourea cleavage method superior to the traditional azide reduction route?
A: The traditional azide route involves hazardous, potentially explosive intermediates and typically requires complex chromatographic purification to remove impurities. The thiourea cleavage method described in CN101223176B utilizes stable haloacetamido precursors and achieves high purity (>95%) through simple precipitation and filtration, eliminating the need for column chromatography and significantly enhancing process safety.
Q: What are the critical reaction conditions for maximizing yield in this synthesis?
A: Critical conditions include the use of pure glacial acetic acid as both solvent and reagent, maintaining a reaction temperature of 80°C, and ensuring a reaction time of approximately 2 hours. The absence of water or other organic solvents like ethanol is crucial, as their presence leads to the formation of stable S-isothiourea salts that halt the reaction and reduce yield to less than 10%.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the process is highly scalable. It avoids hazardous azide chemistry, uses inexpensive and readily available reagents like thiourea and acetic acid, and relies on precipitation for isolation rather than resource-intensive chromatography. These factors collectively reduce operational complexity and waste generation, making it ideal for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4β-Amino-4'-demethyl-4-deoxypodophyllotoxin Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving oncology therapies. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex synthetic routes like the thiourea cleavage method are executed with precision and consistency. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Whether you require custom synthesis or large-scale supply, our infrastructure is designed to support your project from early-stage development through to commercial launch, guaranteeing a reliable source of critical pharmaceutical building blocks.
We invite you to engage with our technical procurement team to discuss how we can tailor our manufacturing capabilities to your specific requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how optimizing the synthesis route can reduce your overall project costs. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing you to make informed decisions based on verified performance metrics. Let us collaborate to accelerate your drug development timeline with a supply partner dedicated to excellence, safety, and innovation in fine chemical manufacturing.
