Advanced Synthesis of N-[8-(2-hydroxybenzoyl)amino]octanoic Acid for Oral Peptide Delivery
Advanced Synthesis of N-[8-(2-hydroxybenzoyl)amino]octanoic Acid for Oral Peptide Delivery
The rapid expansion of the oral peptide therapeutic market, driven largely by the success of oral semaglutide, has placed immense strategic value on high-quality absorption enhancers. Central to this technology is Monosodium N-[8-(2-hydroxybenzoyl)amino]caprylate, widely known as SNAC, which facilitates the gastric absorption of large molecule drugs. Patent CN113045445A introduces a groundbreaking manufacturing methodology for the key precursor, N-[8-(2-hydroxybenzoyl)amino]octanoic acid, addressing critical bottlenecks in purity and cost that have historically constrained supply chains. This technical disclosure outlines a robust five-step synthetic route originating from salicylamide, offering a viable pathway for reliable pharmaceutical intermediate supplier networks to meet the surging global demand for oral delivery excipients.
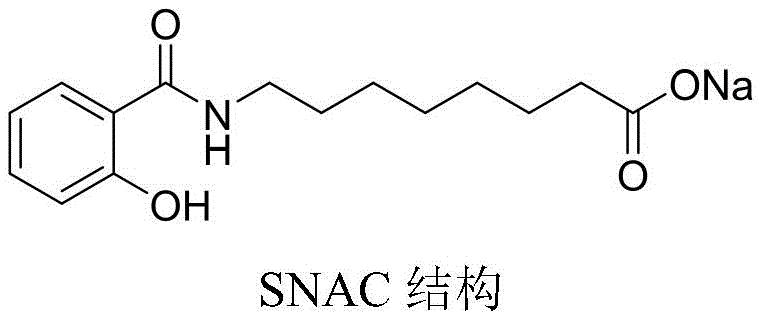
The significance of this chemical entity cannot be overstated, as it serves as the foundational building block for one of the most commercially successful oral biologics currently on the market. The ability to produce the acid precursor with high efficiency and minimal impurity burden directly correlates to the downstream quality of the final drug product. By leveraging the novel cyclization and substitution strategies detailed in the patent, manufacturers can achieve liquid phase purities exceeding 99.7 percent, a specification that is critical for regulatory compliance in GMP environments. This report analyzes the technical merits of this innovation and its implications for cost reduction in API manufacturing and supply chain stability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-[8-(2-hydroxybenzoyl)amino]octanoic acid has been plagued by complex purification requirements and significant impurity risks. Prior art methods often necessitate the preparation and isolation of 2-(6-halogenated hexyl) malonate as a discrete intermediate. This specific compound is thermally unstable and prone to decomposition, requiring high-temperature reduced-pressure distillation for purification, which imposes heavy capital expenditure on equipment and energy consumption. Furthermore, the harsh conditions required for this distillation frequently lead to the formation of stubborn byproducts, such as the impurity designated as Formula VI in the patent literature.
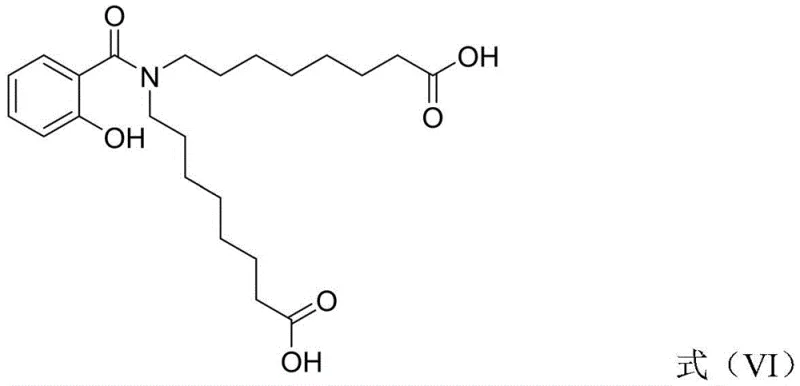
The presence of such impurities creates a cascade of quality control issues, as they are structurally similar to the target molecule and difficult to remove via standard crystallization or chromatography. This not only lowers the overall yield of the process but also introduces variability that is unacceptable for pharmaceutical grade materials. The reliance on expensive and difficult-to-source starting materials like ethyl 6-bromohexanoate further exacerbates the cost profile, making conventional routes economically unattractive for large-scale commercialization. Consequently, there has been a persistent industry need for a streamlined approach that bypasses these hazardous and inefficient purification steps.
The Novel Approach
The methodology disclosed in patent CN113045445A fundamentally reengineers the synthetic logic to circumvent these historical pitfalls. Instead of isolating the problematic malonate intermediate, the new process employs a telescoped strategy where the alkyl chain extension occurs directly on a protected benzoxazine scaffold. This innovative sequence begins with the cyclization of salicylamide to form a stable 2H-1,3-benzoxazine-2,4(3H)-dione, which serves as a robust platform for subsequent functionalization. By deferring the introduction of the malonate moiety until after the initial alkylation, the process avoids the thermal stress that typically generates Formula VI impurities.
Moreover, the patent highlights the versatility of the nucleophilic substitution steps, noting that the second step can be conducted under solvent-free conditions. This green chemistry feature not only reduces the environmental footprint by minimizing solvent waste but also significantly lowers the operational costs associated with solvent recovery and disposal. The final product, N-[8-(2-hydroxybenzoyl)amino]octanoic acid, is obtained through a straightforward hydrolysis and decarboxylation sequence that delivers high purity without the need for complex chromatographic separations. This streamlined approach represents a substantial advancement in process chemistry, aligning perfectly with the goals of modern sustainable manufacturing.
Mechanistic Insights into the Five-Step Synthetic Route
The core of this technological breakthrough lies in the precise orchestration of five distinct chemical transformations, each optimized for yield and selectivity. The journey begins with the activation of salicylamide using carbonyldiimidazole (CDI) in polar aprotic solvents like DMF or DMA. This cyclization reaction efficiently constructs the benzoxazine ring system, protecting the phenolic hydroxyl group and activating the nitrogen for alkylation. The resulting intermediate, 2H-1,3-benzoxazine-2,4(3H)-dione, is isolated as a high-purity solid, setting a clean foundation for the subsequent chain elongation steps.
Following cyclization, the process undergoes a critical nucleophilic substitution where the benzoxazine nitrogen attacks a dihalohexane, such as 1,6-dibromohexane. This step is catalyzed by phase transfer catalysts like tetrabutylammonium bromide, which facilitate the reaction even in the absence of bulk solvent. The resulting N-alkylated benzoxazine derivative then undergoes a second nucleophilic substitution with dialkyl malonate. This clever maneuver extends the carbon chain by three carbons while introducing the ester functionalities required for the final acid group, all while maintaining the integrity of the benzoxazine ring.
![Target molecular structure of N-[8-(2-hydroxybenzoyl)amino]octanoic acid (Formula I)](/insights/img/n-8-2-hydroxybenzoyl-amino-octanoic-acid-synthesis-pharma-supplier-20260309090439-03.webp)
The final stages involve the strategic deconstruction of the temporary protecting groups and the adjustment of the oxidation state. Alkaline hydrolysis serves a dual purpose: it opens the benzoxazine ring to regenerate the free phenolic hydroxyl group and simultaneously hydrolyzes the malonate esters to carboxylic acids. The resulting malonic acid derivative is then subjected to thermal decarboxylation in a high-boiling solvent like xylene. This final step cleanly removes one carboxyl group as carbon dioxide, yielding the target N-[8-(2-hydroxybenzoyl)amino]octanoic acid with the correct eight-carbon chain length. The mechanistic elegance of this route ensures that impurities are minimized at every stage, resulting in a final product that meets stringent purity specifications.
How to Synthesize N-[8-(2-hydroxybenzoyl)amino]octanoic Acid Efficiently
Implementing this synthesis requires careful attention to reaction parameters, particularly temperature control during the cyclization and decarboxylation phases. The patent provides detailed experimental examples demonstrating that molar yields can consistently exceed 85 percent for the final step when optimal conditions are maintained. For R&D teams looking to replicate or scale this process, the key lies in the efficient workup procedures, such as the use of specific solvent mixtures for recrystallization that maximize recovery while excluding trace byproducts. The detailed standardized synthesis steps are outlined below to guide process development.
- Cyclization of salicylamide with CDI to form 2H-1,3-benzoxazine-2,4(3H)-dione.
- Nucleophilic substitution with dihalohexane to introduce the alkyl chain.
- Secondary nucleophilic substitution with dialkyl malonate to extend the carbon backbone.
- Alkaline hydrolysis to open the benzoxazine ring and hydrolyze esters.
- Thermal decarboxylation in xylene to yield the final N-[8-(2-hydroxybenzoyl)amino]octanoic acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology offers compelling economic and logistical benefits. The shift away from complex distillation-dependent intermediates translates directly into a more resilient supply chain that is less susceptible to equipment failures or bottlenecks. By utilizing commodity chemicals like salicylamide and diethyl malonate, manufacturers can secure raw material sourcing at significantly lower price points compared to specialized brominated esters. This fundamental change in the bill of materials drives down the overall cost of goods sold, enabling more competitive pricing for the final absorption enhancer.
- Cost Reduction in Manufacturing: The elimination of high-vacuum distillation steps removes a major capital and operational expense from the production line. Traditional methods require specialized glass-lined reactors capable of withstanding high temperatures and deep vacuums, which are expensive to maintain and operate. In contrast, the new process relies on standard agitation and heating, allowing for the use of general-purpose reactor trains. Furthermore, the potential for solvent-free operation in the alkylation step drastically reduces the volume of organic solvents that must be purchased, stored, and subsequently treated as waste, leading to substantial savings in both material costs and environmental compliance fees.
- Enhanced Supply Chain Reliability: Sourcing strategies are greatly simplified by the use of widely available starting materials. Salicylamide is a bulk chemical produced by numerous suppliers globally, ensuring that production is never halted due to the scarcity of a niche precursor. This diversification of the supply base mitigates the risk of single-source dependency, a critical factor for pharmaceutical companies aiming to ensure continuous availability of their oral formulations. The robustness of the synthetic route also means that batch-to-batch variability is minimized, reducing the likelihood of failed quality control tests that could delay shipments to downstream customers.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, with reaction conditions that are easily transferable from pilot plant to multi-ton commercial production. The reduction in hazardous waste generation, particularly through the avoidance of difficult-to-treat distillation residues, aligns with increasingly strict global environmental regulations. This eco-friendly profile not only reduces disposal costs but also enhances the corporate sustainability metrics of the manufacturing entity. The ability to produce high-purity intermediates with a smaller environmental footprint positions suppliers as preferred partners for multinational corporations with rigorous ESG mandates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of this critical pharmaceutical intermediate. These answers are derived directly from the experimental data and technical disclosures found within the patent documentation, providing clarity on the feasibility and advantages of the new synthetic route. Understanding these details is essential for stakeholders evaluating the integration of this technology into their existing manufacturing portfolios.
Q: How does this new method improve impurity control compared to prior art?
A: The patented method avoids the isolation of 2-(6-halogenated hexyl) malonate, an intermediate that traditionally requires high-temperature vacuum distillation and often generates difficult-to-remove impurities like Formula VI. By performing the malonate substitution directly on the benzoxazine intermediate, the process significantly reduces impurity formation and simplifies purification.
Q: What are the cost advantages of using salicylamide as the starting material?
A: Salicylamide is a commercially abundant and inexpensive raw material compared to specialized brominated esters required in older routes. Additionally, the second step of this process can be performed under solvent-free conditions, drastically reducing solvent procurement and recovery costs while minimizing environmental waste.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process is designed for industrial scalability. It eliminates the need for complex high-vacuum distillation equipment for intermediate purification and utilizes standard reagents like potassium carbonate and common solvents. The high molar yields reported in the examples demonstrate robust performance suitable for metric-ton scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-[8-(2-hydroxybenzoyl)amino]octanoic Acid Supplier
As the demand for oral peptide therapeutics continues to accelerate, the need for a dependable source of high-quality absorption enhancer precursors has never been greater. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging advanced process chemistry to deliver N-[8-(2-hydroxybenzoyl)amino]octanoic acid that meets the most exacting standards. Our facility boasts extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can support your clinical trials through to full market launch without interruption. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch performs consistently in your final formulation.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our innovative production capabilities can add value to your oral drug delivery projects.
