Advanced Synthesis of 3 Beta-Ursodesoxycholic Acid for High-Purity Pharmaceutical Intermediates
The pharmaceutical industry continuously demands higher purity standards for active pharmaceutical ingredients (APIs) and their associated impurities to ensure patient safety and regulatory compliance. In this context, patent CN109912676B discloses a groundbreaking preparation method for 3 beta-ursodesoxycholic acid, a critical impurity reference standard for Ursodeoxycholic Acid (UDCA). UDCA is a first-line medication for treating liver and gallbladder diseases, with a massive market scale. As regulatory bodies increasingly scrutinize impurity profiles, the ability to synthesize specific isomers like the 3-beta variant efficiently is paramount. This novel methodology addresses the longstanding challenges of low yields and harsh reaction conditions found in traditional routes, offering a robust pathway for producing high-purity pharmaceutical intermediates. By leveraging selective protection strategies and advanced stereoselective reduction techniques, this process represents a significant leap forward in bile acid derivative synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
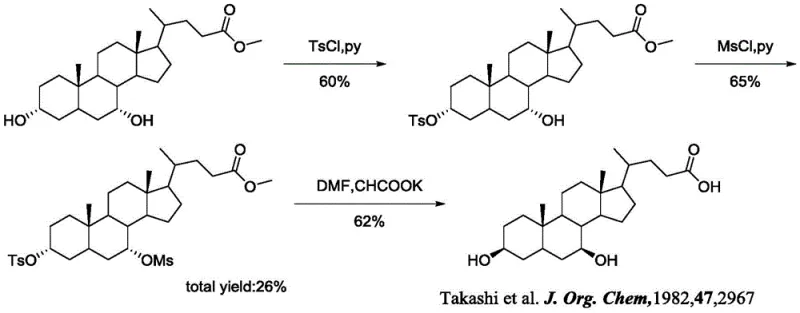
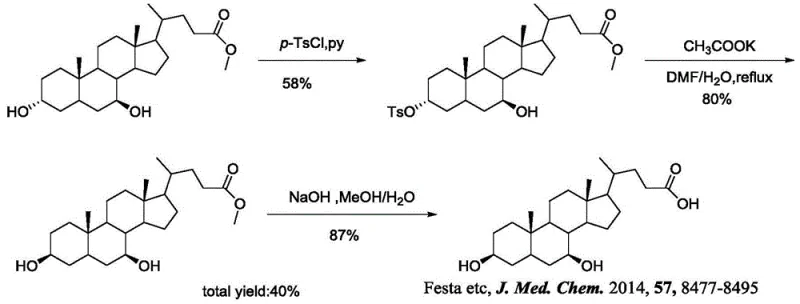
Historically, the synthesis of 3 beta-ursodesoxycholic acid has been plagued by inefficiencies that hinder commercial viability and research scalability. Early attempts, such as the method reported by Takashi et al. in 1982, utilized chenodeoxycholic acid as a starting material. While chemically feasible, this route suffered from a dismal total yield of merely 26% over three steps. The process involved the use of harsh sulfonyl chlorides (TsCl, MsCl) and required rigorous conditions that often led to numerous by-products, complicating purification and driving up costs. Similarly, a more recent approach by Festa et al. in 2014 attempted to improve upon this by using ursodeoxycholic acid methyl ester. Although this method achieved a slightly better total yield of 40%, it still relied on severe reaction conditions that posed safety risks and operational difficulties in a manufacturing setting. These legacy methods illustrate the critical need for a more efficient, high-yielding synthetic strategy.
The Novel Approach
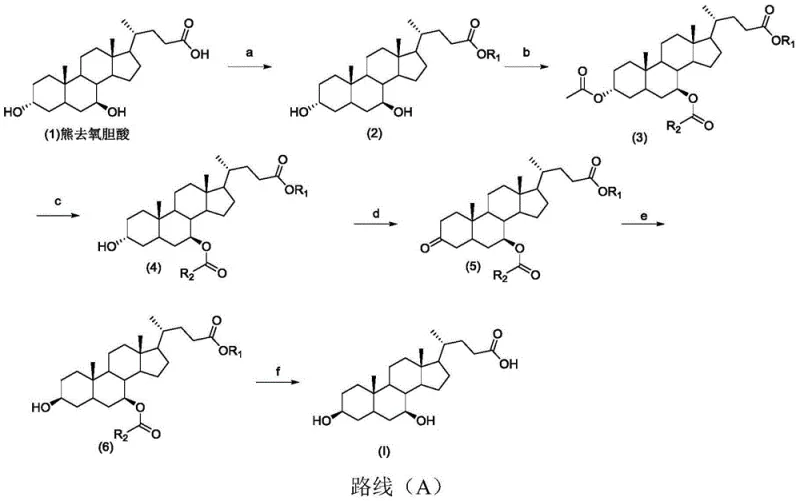
In stark contrast to these predecessors, the method disclosed in patent CN109912676B introduces a sophisticated six-step sequence that achieves a remarkable total yield of 57%. This new route begins with the abundant and cost-effective ursodeoxycholic acid. The innovation lies in the precise orchestration of protection and deprotection steps. By initially esterifying the carboxyl group and acetylating both hydroxyl groups, the molecule is primed for selective manipulation. The key breakthrough is the selective removal of the 3-position protecting group, followed by oxidation and a highly stereoselective reduction. This approach not only maximizes material throughput but also ensures the correct stereochemical outcome at the C3 position. The use of mild reagents and ambient to moderate temperatures throughout the sequence drastically reduces energy consumption and safety hazards, marking a substantial improvement over the aggressive chemistry of the past.
Mechanistic Insights into Stereoselective Reduction and Oxidation
The core chemical ingenuity of this process is centered on the transformation of the 3-alpha-hydroxyl group to the 3-beta configuration. This is accomplished through an oxidation-reduction sequence that effectively inverts the stereochemistry. In step (d), the intermediate bearing a free 3-hydroxyl group is oxidized using IBX (2-Iodoxybenzoic acid) in DMSO. IBX is a hypervalent iodine reagent known for its ability to oxidize alcohols to ketones under mild, non-acidic conditions, preserving the integrity of other sensitive functional groups in the steroid backbone. This oxidation generates a 3-keto intermediate, erasing the original stereochemical information at the C3 position and creating a planar carbonyl group ready for nucleophilic attack.
Following oxidation, step (e) executes the critical stereoselective reduction. The protocol employs a unique combination of Raney Nickel, Sodium Borohydride (NaBH4), and Potassium tert-butoxide (t-BuOK) in isopropanol. The presence of the bulky tert-butoxide base and the heterogeneous Raney Nickel catalyst creates a steric environment that directs the hydride delivery from the less hindered face of the molecule. This specific reagent system favors the formation of the equatorial 3-beta-hydroxyl group over the axial 3-alpha isomer. This level of stereocontrol is essential for generating the specific impurity reference standard required for quality control in UDCA manufacturing. The mechanism ensures high diastereoselectivity, minimizing the formation of unwanted isomers and simplifying downstream purification processes significantly.
How to Synthesize 3 Beta-Ursodesoxycholic Acid Efficiently
The synthesis of this valuable intermediate follows a logical progression of functional group manipulations designed to maximize yield and purity. The process initiates with the esterification of the starting material to protect the side chain, followed by global acetylation. The subsequent selective hydrolysis is the pivot point that allows for the specific modification of the A-ring. For laboratory or pilot-scale execution, strict adherence to the molar ratios and temperature controls specified in the patent is essential to replicate the 57% total yield. Detailed operational parameters, including solvent choices and reaction times for each of the six steps, are critical for success.
- Esterify ursodeoxycholic acid at the 24-position carboxyl group using methanol and sulfuric acid to form the methyl ester.
- Protect the 3-and 7-hydroxyl groups via acetylation using acetic anhydride and DMAP in pyridine.
- Selectively remove the 3-position acetyl group using sodium hydroxide in methanol to expose the 3-hydroxyl group.
- Oxidize the exposed 3-hydroxyl group to a ketone using IBX in DMSO.
- Perform stereoselective hydrogenation reduction using Raney Nickel, Sodium Borohydride, and Potassium tert-butoxide to invert the stereochemistry to 3-beta.
- Hydrolyze the remaining ester protecting groups using sodium hydroxide to yield the final 3 beta-ursodesoxycholic acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route offers tangible strategic benefits beyond mere chemical elegance. The shift from low-yielding, hazardous legacy processes to this streamlined methodology directly impacts the cost structure and reliability of the supply chain. By improving the total yield from historical lows of 26-40% to 57%, the amount of raw material required to produce a kilogram of the final product is drastically reduced. This efficiency gain translates into significant cost reduction in pharmaceutical intermediates manufacturing, as less starting material is wasted, and fewer resources are consumed in purification and waste disposal.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by its high atom economy and the use of inexpensive, commodity-grade reagents. Unlike older methods that relied on specialized sulfonyl chlorides or severe conditions requiring expensive containment, this route utilizes common solvents like methanol and dichloromethane. The elimination of complex, multi-step purifications caused by by-product formation further lowers operational expenditures. The overall process design minimizes the number of isolation steps and maximizes the throughput per batch, leading to substantial cost savings without compromising quality.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by processes that rely on exotic or hard-to-source reagents. This synthesis mitigates that risk by depending on widely available chemicals such as acetic anhydride, sulfuric acid, and Raney Nickel. The robustness of the reaction conditions—operating primarily between 25°C and 50°C—means that production is less susceptible to disruptions caused by equipment limitations or safety shutdowns. This reliability ensures reducing lead time for high-purity pharmaceutical intermediates, allowing manufacturers to respond more agilely to market demand fluctuations.
- Scalability and Environmental Compliance: As the industry moves towards greener manufacturing, the mild nature of this process is a distinct advantage. The avoidance of heavy metals (other than the recyclable Raney Nickel) and toxic chlorinating agents aligns with modern environmental, health, and safety (EHS) standards. The simplified workup procedures, often involving basic extraction and crystallization, reduce the volume of organic waste generated. This environmental compatibility facilitates easier regulatory approval for commercial scale-up of complex pharmaceutical intermediates, ensuring long-term viability of the production line.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders evaluating its implementation. The following questions address common inquiries regarding the process's selectivity, scalability, and comparative advantages. These answers are derived directly from the experimental data and technical disclosures within the patent documentation, providing a factual basis for decision-making.
Q: What is the primary advantage of this new synthesis method over prior art?
A: The primary advantage is the significantly improved total yield of 57%, compared to 26% in the 1982 Takashi method and 40% in the 2014 Festa method. Additionally, the reaction conditions are much milder, avoiding harsh reagents and extreme temperatures.
Q: How is the 3-beta stereochemistry controlled in this process?
A: The 3-beta stereochemistry is achieved through a specific stereoselective reduction step. After oxidizing the 3-hydroxyl to a ketone, the molecule is reduced using a system of Raney Nickel, Sodium Borohydride, and Potassium tert-butoxide, which favors the formation of the 3-beta-hydroxyl configuration.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process is designed for scalability. It utilizes common industrial solvents like methanol and dichloromethane, operates at mild temperatures (mostly 25-50°C), and employs readily available reagents, making it highly suitable for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3 Beta-Ursodesoxycholic Acid Supplier
The development of efficient synthetic routes like the one described in CN109912676B underscores the complexity and sophistication required in modern fine chemical manufacturing. At NINGBO INNO PHARMCHEM, we specialize in translating such innovative laboratory protocols into robust, industrial-scale processes. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high yields and purity levels achieved in the lab are maintained in the plant. We operate stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of 3 beta-ursodesoxycholic acid meets the exacting standards required for pharmaceutical impurity reference materials.
We invite pharmaceutical companies and research institutions to collaborate with us to secure a stable supply of this critical intermediate. By leveraging our technical expertise, we can help you navigate the complexities of bile acid synthesis and optimize your supply chain. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to support your R&D and commercialization goals.
