Industrial Scale-Up of High-Purity Delta9,11-Canrenone via Novel Dehydration Route
Introduction to the Breakthrough in Steroid Intermediate Manufacturing
The pharmaceutical industry is constantly seeking more efficient pathways for the production of critical hormonal intermediates, particularly those serving as precursors for advanced therapeutics like eplerenone. A recent technological advancement disclosed in patent CN112062805B presents a paradigm shift in the synthesis of Delta9,11-canrenone, a pivotal intermediate in the manufacture of selective aldosterone receptor antagonists. This novel methodology replaces the cumbersome, multi-step biological fermentation and chemical modification sequences traditionally employed with a streamlined, purely chemical synthetic route. By leveraging a strategic dehydration-epoxidation sequence, this process not only simplifies the operational workflow but also significantly enhances the structural integrity of the final product. For R&D directors and supply chain managers alike, this represents a move towards more predictable, scalable, and cost-effective manufacturing protocols that align with modern green chemistry principles while maintaining rigorous quality standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of Delta9,11-canrenone has relied heavily on a convoluted pathway that begins with the synthesis of canrenone, followed by a biological fermentation step to introduce the 11-hydroxyl group. This biotransformation is inherently variable, subject to the whims of microbial metabolism, and often results in inconsistent batch-to-batch yields. Following fermentation, the process necessitates a series of harsh chemical treatments, specifically sulfoacid esterification and subsequent desulfonation, to generate the desired 9,11-double bond. These acidic and basic conditions are notoriously detrimental to the 17-position lactone ring, a sensitive structural motif essential for the biological activity of the final drug. The exposure of the lactone ring to such aggressive reagents frequently leads to hydrolysis, ring-opening, and the formation of difficult-to-remove impurities, thereby dragging down the overall mass yield and complicating downstream purification efforts.
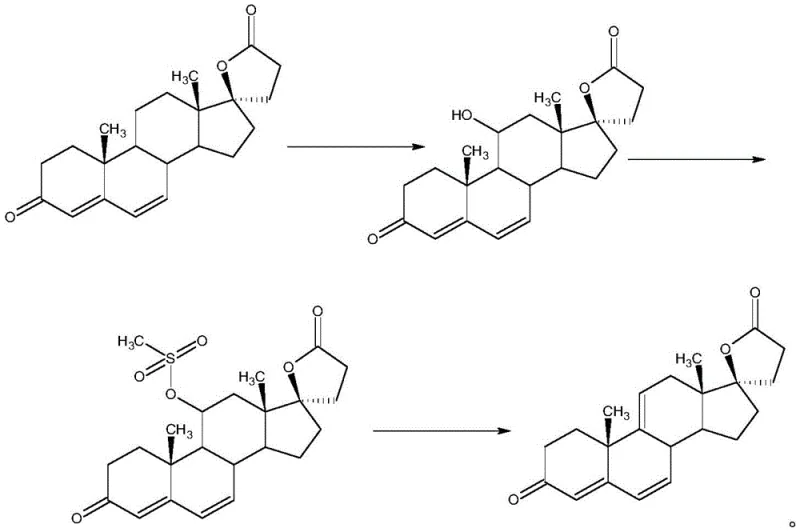
The Novel Approach
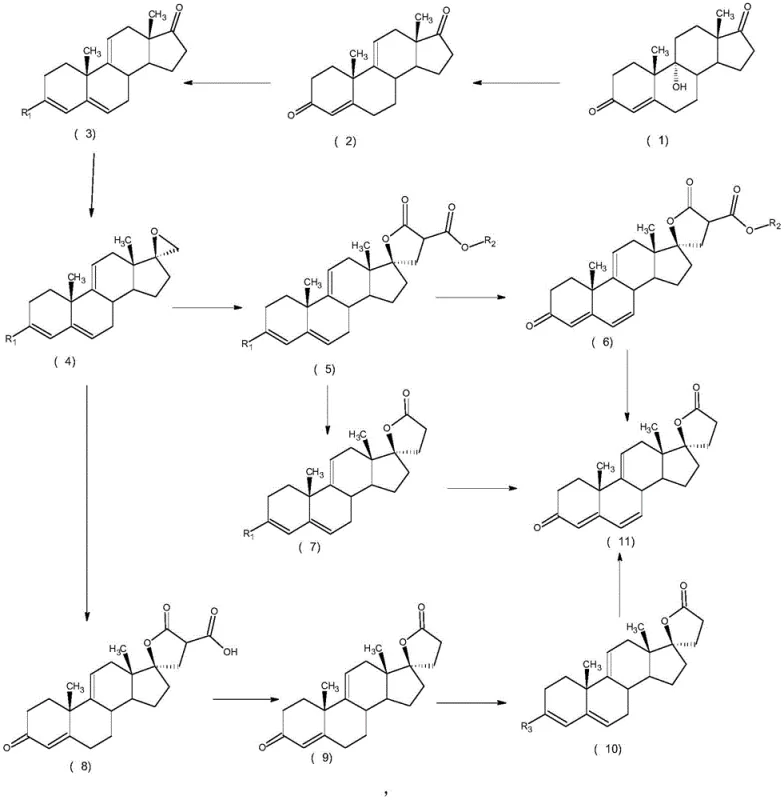
In stark contrast, the innovative process outlined in the patent data reverses the logical order of bond construction to circumvent these stability issues. Instead of building the lactone ring first and then attempting to modify the steroid nucleus, this method prioritizes the formation of the 9,11-double bond at the earliest possible stage. Starting from the readily available 9α-hydroxy-4-androstene-3,17-dione, the protocol employs a mild acid-catalyzed dehydration to instantly establish the crucial diene system. Only after this robust carbon skeleton is secured does the synthesis proceed to protect the 3-position carbonyl and construct the 17-position lactone ring via epoxidation and condensation. This strategic reordering ensures that the sensitive lactone functionality is never exposed to the harsh conditions required for double bond formation, effectively eliminating a major source of degradation and impurity generation.
Mechanistic Insights into the Dehydration-Epoxidation Cascade
The core of this high-efficiency process lies in its precise control over reaction kinetics and functional group compatibility. The initial dehydration step utilizes dilute sulfuric acid, typically at concentrations between 20% and 80%, maintained at a moderate temperature range of 10°C to 60°C. This specific window is critical; it provides sufficient proton activity to facilitate the elimination of the 9α-hydroxyl group without promoting the isomerization of the double bonds or the degradation of the steroid backbone. Following this, the 3-position ketone is masked using protecting agents such as trimethyl orthoformate or pyrrolidine in the presence of catalysts like pyridine hydrochloride. This protection is vital as it prevents unwanted side reactions at the A-ring during the subsequent nucleophilic attacks at the D-ring.
The construction of the lactone ring is achieved through a sophisticated sequence involving Corey-Chaykovsky epoxidation followed by malonate condensation. The epoxidation at the 17-position is executed using trimethyl sulfonium halides and strong bases like sodium methoxide or potassium tert-butoxide in polar aprotic solvents such as DMSO or THF. This generates a reactive spiro-epoxide intermediate which is then opened by a malonic diester nucleophile. The final transformation involves an oxidative decarboxylation, often employing reagents like tetrachloro-o-benzoquinone or dichlorodicyanobenzoquinone. This step simultaneously removes the extra carboxyl groups introduced by the malonate and oxidizes the system to restore the conjugated enone system, locking the molecule into the thermodynamically stable Delta9,11-canrenone configuration with exceptional stereochemical fidelity.
How to Synthesize Delta9,11-Canrenone Efficiently
Implementing this synthesis requires strict adherence to the specified stoichiometric ratios and thermal profiles to maximize the reported total mass yield of over 80%. The process is designed to be operationally simple, relying largely on standard unit operations such as extraction, filtration, and crystallization, which facilitates easy technology transfer from the laboratory to the pilot plant. The detailed standardized synthesis steps, including specific solvent volumes, reagent grades, and workup procedures, are outlined below to guide process engineers in replicating these high-yield results.
- Dehydrate 9α-hydroxy-4-androstene-3,17-dione using dilute sulfuric acid at 10-60°C to form the 9,11-double bond.
- Protect the 3-position carbonyl using trimethyl orthoformate or pyrrolidine to form the enol ether or enamine derivative.
- Perform Corey-Chaykovsky epoxidation at the 17-position using trimethyl sulfonium halide and base, followed by condensation with malonic diester and oxidative decarboxylation to close the lactone ring.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from a fermentation-dependent process to this fully chemical synthetic route offers profound strategic advantages. The reliance on biological systems introduces significant volatility into the supply chain, including long lead times for culture development and susceptibility to contamination. By replacing this with a robust chemical process using commodity chemicals like sulfuric acid and malonic esters, manufacturers can achieve a much higher degree of supply continuity and predictability. Furthermore, the simplification of the workflow directly translates to reduced capital expenditure, as there is no longer a need for specialized fermentation infrastructure or the extensive wastewater treatment systems often associated with biological processes.
- Cost Reduction in Manufacturing: The economic benefits of this route are driven by the drastic shortening of the synthetic timeline and the elimination of expensive enzymatic steps. By avoiding the fermentation, sulfonation, and desulfonation triad, the process removes multiple unit operations that previously consumed significant energy, labor, and solvent resources. The use of inexpensive, bulk-available reagents for the dehydration and lactone formation steps further drives down the raw material cost per kilogram. Additionally, the high purity of the crude product (>95% in many examples) minimizes the need for costly chromatographic purification, allowing for simpler recrystallization techniques that are far more economical at scale.
- Enhanced Supply Chain Reliability: Chemical synthesis offers a level of controllability that biological fermentation simply cannot match. The reaction times are measured in hours rather than days, and the outcome is determined by precise physical parameters rather than biological variability. This reliability allows for tighter production scheduling and faster response times to market demand fluctuations. Moreover, the starting material, 9α-hydroxy-4-androstene-3,17-dione, is a stable, commercially accessible steroid intermediate, ensuring that the supply chain is not bottlenecked by the availability of specialized microbial strains or fermentation substrates.
- Scalability and Environmental Compliance: From an environmental and safety perspective, this process is markedly superior. The avoidance of strong sulfonating agents reduces the generation of hazardous acidic waste streams, simplifying effluent treatment and lowering compliance costs. The reaction conditions are generally mild, with most steps occurring below 100°C, which reduces energy consumption for heating and cooling. The high atom economy of the condensation and dehydration steps, coupled with the ability to recover and recycle solvents like methanol and dichloromethane, aligns perfectly with modern sustainability goals and regulatory frameworks for pharmaceutical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthetic pathway. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear picture of the operational realities and potential hurdles for adoption.
Q: What is the primary advantage of this new synthesis route over the traditional fermentation method?
A: The new chemical route eliminates the need for biological fermentation and subsequent sulfonation/desulfonation steps. By forming the 9,11-double bond early via acid-catalyzed dehydration, it avoids the hydrolysis of the sensitive 17-position lactone ring that often occurs during the harsh acidic or basic conditions of the traditional sulfonation process, resulting in significantly higher purity and yield.
Q: What are the typical reaction conditions for the key dehydration step?
A: The dehydration of the starting material, 9α-hydroxy-4-androstene-3,17-dione, is conducted using dilute sulfuric acid with a volume concentration between 20% and 80%. The reaction temperature is strictly controlled between 10°C and 60°C to ensure selectivity and prevent degradation of the steroid backbone.
Q: How does this process impact the overall production cost for eplerenone intermediates?
A: This process drastically reduces costs by shortening the synthetic route and utilizing cheap, commercially available reagents like dilute sulfuric acid and malonic diesters. Furthermore, the elimination of fermentation tanks and the simplification of purification steps (often just filtration and washing) lead to substantial operational expenditure savings.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Delta9,11-Canrenone Supplier
As the global demand for eplerenone and related cardiovascular therapeutics continues to rise, securing a stable supply of high-quality intermediates is paramount. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific solvent systems and thermal requirements of this dehydration-epoxidation route, ensuring that every batch meets stringent purity specifications. With our rigorous QC labs and commitment to process optimization, we guarantee a product profile that supports the most demanding regulatory filings.
We invite forward-thinking pharmaceutical partners to collaborate with us on optimizing this value chain. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, ensuring that your supply of this critical steroid intermediate is both economically viable and technically secure for the long term.
