Scalable Synthesis of Bioactive Marine Sterols via Diosgenin Transformation for Commercial Production
Scalable Synthesis of Bioactive Marine Sterols via Diosgenin Transformation for Commercial Production
The pharmaceutical and fine chemical industries are constantly seeking reliable sources for complex bioactive molecules, particularly those with potent antitumor and antiviral properties. Patent CN101560236B introduces a groundbreaking methodology for the total synthesis of (25R)-5α-cholesta-3β, 5α, 6β, 26-tetrol, a rare polyhydroxy marine sterol originally isolated from the Caribbean coral Carijoa riisei. Historically, obtaining this compound relied on inefficient extraction from marine biomass, resulting in prohibitive costs and supply instability. This new synthetic approach leverages diosgenin, an abundant and inexpensive plant-derived sapogenin, to construct the complex steroid skeleton with high precision. By shifting from extraction to chemical synthesis, manufacturers can secure a consistent supply of this high-value intermediate, paving the way for the development of next-generation anticancer therapeutics.
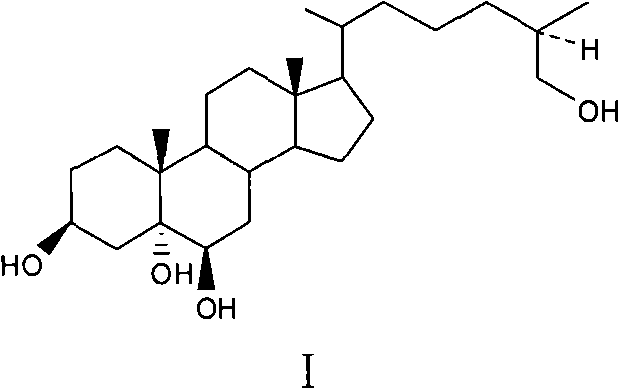
The structural complexity of marine sterols often poses significant challenges for process chemists, particularly regarding the installation of multiple hydroxyl groups with specific stereochemistry. The target molecule, depicted above, features a distinctive 5α, 6β-diol motif alongside a 26-hydroxyl group on the side chain, which are critical for its observed cytotoxicity against HCT-116 tumor cells. Traditional semi-synthetic routes often struggle with regioselectivity, leading to difficult purification processes and low overall yields. The methodology disclosed in this patent overcomes these hurdles through a rational design that prioritizes functional group orthogonality. By strategically protecting sensitive hydroxyls early in the sequence and utilizing robust reduction and oxidation protocols, the process ensures that the final product meets the stringent purity specifications required for pharmaceutical applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the primary source of (25R)-5α-cholesta-3β, 5α, 6β, 26-tetrol was direct extraction from marine organisms, a method fraught with logistical and economic inefficiencies. The natural abundance of this sterol in coral tissues is extremely low, necessitating the processing of massive quantities of biomass to isolate mere milligrams of the active compound. This not only drives up the cost exponentially but also raises significant environmental concerns regarding the sustainability of harvesting marine ecosystems. Furthermore, natural extracts are inherently variable, containing complex mixtures of structurally similar sterols that are notoriously difficult to separate. This variability compromises the reproducibility of biological assays and hinders the regulatory approval process for drug candidates derived from such sources. Consequently, the lack of a scalable synthetic route has severely limited the clinical exploration of this promising anticancer agent.
The Novel Approach
The synthetic strategy outlined in the patent represents a paradigm shift by utilizing diosgenin as a chiral pool starting material, effectively bypassing the need for complex asymmetric synthesis of the steroid core. This route involves a concise six-step sequence that transforms the readily available spiroketal precursor into the desired tetrol with excellent control over stereochemistry. Key to this success is the use of zinc amalgam reduction to open the F-ring of diosgenin, generating a triol intermediate that serves as a versatile platform for further functionalization. The subsequent protection-deprotection maneuvers allow for the selective manipulation of the C-16 and C-26 positions without affecting the sensitive A-ring functionality. This logical disconnection of the molecule enables a streamlined workflow that is amenable to optimization and scale-up.
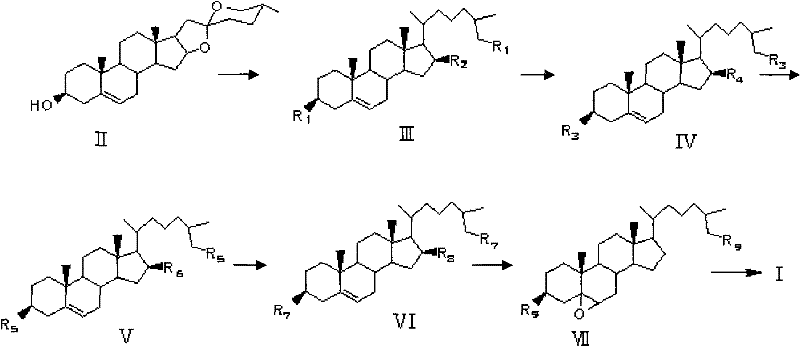
As illustrated in the reaction scheme above, the transformation proceeds through well-defined intermediates, each isolated and characterized to ensure quality control at every stage. The conversion of the 16-hydroxyl group to a mesylate followed by lithium aluminum hydride reduction is a particularly elegant solution for removing the oxygen functionality at that position, which is not present in the final target. Furthermore, the stereoselective epoxidation of the 5,6-double bond using m-chloroperoxybenzoic acid sets the stage for the final acid-catalyzed ring opening, which simultaneously installs the 5α, 6β-diol system and removes the silyl protecting groups. This convergent approach minimizes the number of unit operations and reduces the consumption of solvents and reagents, aligning with the principles of green chemistry while delivering a robust manufacturing process.
Mechanistic Insights into Zinc Amalgam Reduction and Epoxidation
The cornerstone of this synthesis is the initial reductive cleavage of the spiroketal ring in diosgenin using zinc amalgam and concentrated hydrochloric acid. This reaction, reminiscent of a Clemmensen reduction but applied to a ketal system, proceeds via the protonation of the ether oxygen followed by single-electron transfer from the zinc surface. This mechanism effectively breaks the C-O bonds of the spiroketal, yielding a mixture of alcohols that is subsequently equilibrated to the thermodynamically stable triol intermediate (Compound III). The choice of ethanol as the solvent is critical, as it solubilizes the lipophilic steroid while tolerating the strongly acidic conditions required for the reduction. Understanding this mechanism allows process engineers to fine-tune the reaction temperature and acid concentration to maximize the yield of the desired 25R-isomer, minimizing the formation of diastereomeric impurities that could complicate downstream purification.
Following the construction of the reduced skeleton, the stereochemical integrity of the A/B ring junction is preserved and manipulated through a carefully orchestrated epoxidation-hydrolysis sequence. The treatment of the Δ5-alkene with m-chloroperoxybenzoic acid (mCPBA) generates an α-oriented epoxide due to the steric hindrance imposed by the angular C-19 methyl group. Subsequent acid-catalyzed hydrolysis of this epoxide occurs via an SN2-like attack by water at the C-6 position, leading to the inversion of configuration and the formation of the 6β-hydroxyl group. This mechanistic pathway guarantees the formation of the 5α, 6β-diaxial diol arrangement, which is a hallmark of the bioactive marine sterol class. By controlling the pH and temperature during the hydrolysis step, manufacturers can prevent acid-catalyzed dehydration or rearrangement side reactions, ensuring a high-purity final product suitable for sensitive biological testing.
How to Synthesize (25R)-5α-cholesta-3β, 5α, 6β, 26-tetrol Efficiently
Implementing this synthesis in a production environment requires strict adherence to the optimized parameters detailed in the patent to ensure consistent quality and yield. The process begins with the dissolution of diosgenin in ethanol, followed by the careful addition of zinc amalgam and hydrochloric acid under reflux conditions. It is imperative to monitor the reaction progress via TLC to prevent over-reduction or degradation of the steroid nucleus. Once the triol intermediate is secured, the protection of the 3 and 26 hydroxyl groups with TBDMSCl must be performed in anhydrous conditions to prevent premature hydrolysis of the silyl ethers. The subsequent mesylation and reduction steps demand precise stoichiometric control of lithium aluminum hydride to safely reduce the mesylate without reducing other sensitive functionalities. Finally, the epoxidation and hydrolysis sequence should be conducted with high-purity reagents to avoid introducing trace metal impurities that could affect the biological profile of the final API intermediate.
- Perform zinc amalgam reduction on diosgenin in ethanol with concentrated hydrochloric acid under reflux to open the spiroketal ring and form the triol intermediate.
- Protect the 3 and 26 hydroxyl groups using tert-butyldimethylchlorosilane (TBDMSCl) and imidazole in anhydrous DMF to prevent side reactions during subsequent steps.
- Convert the 16-hydroxyl group to a mesylate using methanesulfonyl chloride, followed by lithium aluminum hydride reduction to remove the oxygen functionality at C-16.
- Oxidize the 5,6-double bond with m-chloroperoxybenzoic acid (mCPBA) to form the epoxide, followed by acidic hydrolysis to simultaneously open the epoxide and remove silyl protecting groups.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from natural extraction to this diosgenin-based synthetic route offers transformative benefits in terms of cost stability and supply security. The reliance on marine biomass introduced significant volatility due to seasonal variations, geopolitical issues regarding ocean resources, and the sheer difficulty of logistics. In contrast, diosgenin is a commodity chemical produced on a multi-ton scale globally for the hormone industry, ensuring a robust and diversified supply base. This shift eliminates the risk of raw material shortages and decouples the production of this high-value sterol from environmental constraints. Furthermore, the synthetic route utilizes common, off-the-shelf reagents such as zinc, thionyl chloride derivatives, and peracids, which are readily available from multiple chemical vendors, reducing the risk of single-source dependency for critical inputs.
- Cost Reduction in Manufacturing: The economic advantage of this process is driven primarily by the drastic difference in raw material costs between scarce marine extracts and abundant plant sapogenins. By eliminating the need for expensive chromatographic separation of complex natural mixtures, the overall processing cost is significantly lowered. Additionally, the synthetic route avoids the use of precious metal catalysts or exotic chiral ligands, relying instead on cost-effective stoichiometric reagents. The high selectivity of the reactions minimizes the generation of hard-to-remove impurities, thereby reducing the burden on purification units and lowering solvent consumption. These factors combine to create a manufacturing profile that is financially sustainable for long-term commercial production.
- Enhanced Supply Chain Reliability: Supply chain resilience is markedly improved by adopting a synthesis that relies on established industrial chemicals rather than biological harvesting. The lead time for procuring diosgenin is short and predictable, allowing for accurate production planning and inventory management. Moreover, the synthetic process is not subject to the biological variability inherent in natural products, ensuring that every batch of the intermediate meets identical specifications. This consistency is crucial for pharmaceutical customers who require rigorous validation of their supply chain. The ability to scale production from kilograms to tons without changing the fundamental chemistry provides a clear pathway for meeting increasing market demand as the drug candidate progresses through clinical trials.
- Scalability and Environmental Compliance: From an environmental and safety perspective, this route is designed for scalability with manageable waste streams. While the use of zinc amalgam requires proper handling and disposal protocols, it is a well-understood technology in the fine chemical industry with established abatement procedures. The avoidance of chlorinated solvents in certain steps and the potential for solvent recovery further enhance the green profile of the process. The mild reaction conditions, mostly conducted at or near room temperature after the initial reduction, reduce energy consumption compared to high-pressure or cryogenic alternatives. This alignment with modern environmental, health, and safety (EHS) standards facilitates regulatory approval and supports the sustainability goals of forward-thinking pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this marine sterol intermediate. These answers are derived directly from the technical disclosures in patent CN101560236B and reflect the practical realities of scaling this chemistry. Understanding these details is essential for R&D teams evaluating the feasibility of this route for their specific pipeline projects. The clarity provided here aims to accelerate the decision-making process for sourcing and development partnerships.
Q: What are the primary advantages of using diosgenin as a starting material for marine sterol synthesis?
A: Diosgenin is a readily available, low-cost sapogenin derived from yams, offering a sustainable and economically viable alternative to extracting trace amounts of sterols from marine organisms like corals.
Q: How does this synthetic route ensure high stereochemical purity at the C-5 and C-6 positions?
A: The process utilizes stereoselective epoxidation of the 5,6-double bond followed by acid-catalyzed ring opening, which strictly controls the formation of the 5α, 6β-diol configuration essential for biological activity.
Q: Is this manufacturing process suitable for large-scale commercial production?
A: Yes, the reaction conditions are mild, utilizing common reagents like zinc amalgam and mCPBA, and avoid exotic catalysts, making the pathway highly adaptable for kilogram-to-ton scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (25R)-5α-cholesta-3β, 5α, 6β, 26-tetrol Supplier
At NINGBO INNO PHARMCHEM, we recognize the immense potential of marine-derived sterols in oncology and virology, and we are uniquely positioned to bring this patented technology to commercial reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to clinical supply is seamless. We operate state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, including the precise stereochemical configuration of the 5α, 6β-diol system which is critical for biological activity. Our commitment to quality assurance means that every batch of (25R)-5α-cholesta-3β, 5α, 6β, 26-tetrol is accompanied by comprehensive analytical data, giving you confidence in your supply chain.
We invite pharmaceutical innovators to collaborate with us to leverage this cost-effective and scalable synthesis for your drug development programs. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our manufacturing expertise can accelerate your project while optimizing your budget. Let us be your strategic partner in unlocking the therapeutic value of complex marine natural products.
