Advanced Chemical Synthesis of Epidaunorubicin Intermediates for Scalable Oncology Drug Manufacturing
Advanced Chemical Synthesis of Epidaunorubicin Intermediates for Scalable Oncology Drug Manufacturing
The pharmaceutical industry is constantly seeking more robust and reliable pathways for the production of critical oncology therapeutics. A significant breakthrough in this domain is detailed in patent CN109293613B, which discloses a novel synthesis method for a key intermediate of epirubicin, specifically an epidaunorubicin intermediate compound. Epirubicin, a potent anthracycline antibiotic, is widely utilized in the treatment of breast cancer, lung cancer, and various solid tumors. Its mechanism involves intercalating between DNA nucleobase pairs and inhibiting topoisomerase II. While traditionally produced via semi-synthesis from fermentation-derived daunorubicin, this patent introduces a fully chemical synthetic route that addresses long-standing issues regarding purity and supply consistency. By utilizing (2R, 6S)-6-methoxy-2-methyl-6H-pyran-3-one as a starting material, the process achieves high stereochemical control and operational simplicity.
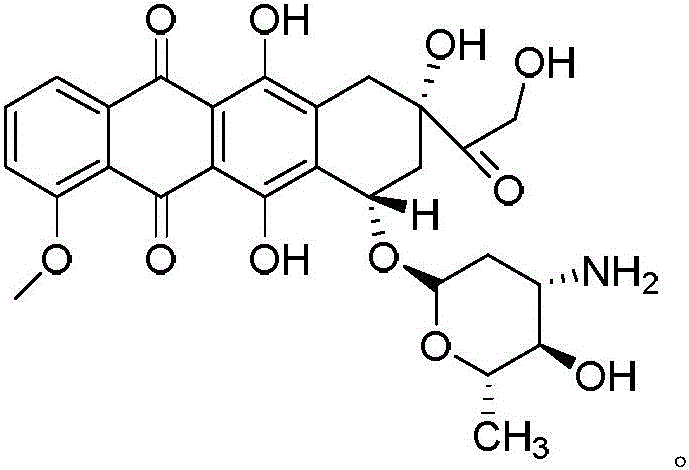
This technological advancement represents a paradigm shift for manufacturers of high-purity pharmaceutical intermediates. The ability to synthesize the crucial glycosidic portion of the molecule chemically, rather than relying on biological fermentation, offers unprecedented control over the impurity profile. For R&D directors and process chemists, this means access to a precursor that minimizes the risk of biological contaminants and ensures a consistent quality attribute profile essential for regulatory approval. The patent outlines a comprehensive pathway that transforms simple pyranone derivatives into complex chiral building blocks, setting a new standard for cost reduction in pharmaceutical intermediates manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of epirubicin has relied heavily on daunorubicin obtained through microbial fermentation. While effective, this biological approach is fraught with inherent limitations that impact both cost and quality. Fermentation processes are notoriously sensitive to environmental variables, leading to significant batch-to-batch variability in yield and impurity profiles. The crude fermentation broth typically contains a complex mixture of structurally similar analogs, necessitating rigorous and expensive purification steps, often involving large-scale resin column chromatography. This not only extends the production lead time significantly but also introduces potential safety threats due to residual solvents or leachables from the resin. Furthermore, the low efficiency of fermentation means that vast amounts of raw materials are required to produce relatively small quantities of the active pharmaceutical ingredient, creating a bottleneck in the commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
In stark contrast, the method described in patent CN109293613B utilizes a total chemical synthesis strategy that bypasses the unpredictability of fermentation. The core innovation lies in the construction of the chiral amino sugar moiety from scratch using (2R, 6S)-6-methoxy-2-methyl-6H-pyran-3-one. This route is characterized by its short step count and mild reaction conditions, which are critical for maintaining the integrity of sensitive functional groups. The process involves a logical sequence of double bond reduction, halogenation, ammoniation, chiral resolution, and protection/deprotection steps. By avoiding biological systems, manufacturers can achieve a much cleaner reaction profile, with the patent reporting final product purities reaching up to 99.3% and maximum single impurities below 0.5%. This level of purity drastically simplifies downstream processing, eliminating the need for extensive resin column separations and thereby enhancing overall process economics.
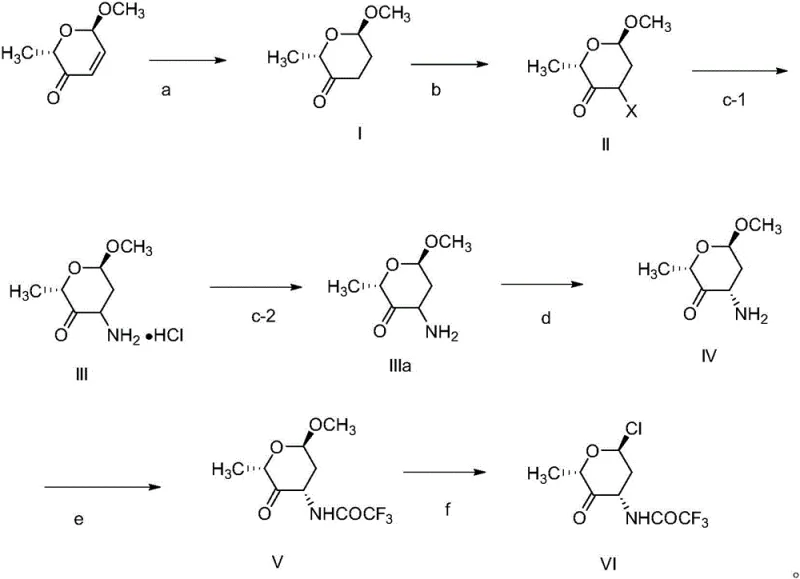
Mechanistic Insights into Chiral Resolution and Functionalization
The success of this synthetic route hinges on precise stereochemical control, particularly during the formation of the amino sugar ring. A critical step in the mechanism is the chiral resolution of the racemic amine intermediate. The patent details the use of specific chiral resolving agents such as L-tyrosine, L-pyroglutamic acid, or L-dibenzoyltartaric acid. These agents form diastereomeric salts with the amine, which possess different solubility properties in alcoholic solvents. By carefully controlling the temperature and solvent composition during crystallization, the desired (2S, 4S, 6R) enantiomer can be selectively precipitated while the unwanted isomer remains in solution. This kinetic and thermodynamic control allows for the isolation of the chiral amine with an enantiomeric excess (ee) value exceeding 99.8%, which is paramount for the biological activity of the final drug.
Following resolution, the mechanism proceeds through a series of functional group transformations designed to activate the sugar for coupling. The introduction of a halogen atom at the anomeric position (via chlorination of the methoxy group in Formula V to yield Formula VI) creates a highly reactive glycosyl donor. This activation is achieved under mild conditions using reagents like acetyl chloride/thionyl chloride or boron trichloride. The presence of the trifluoroacetyl protecting group on the nitrogen atom is also mechanistically significant; it not only prevents side reactions during the chlorination but also modulates the electronic properties of the ring to favor the desired stereochemistry during the subsequent glycosylation with the anthraquinone aglycone. This careful orchestration of protecting group chemistry and activation strategies ensures high regioselectivity and stereoselectivity throughout the synthesis.
How to Synthesize Epidaunorubicin Intermediate Efficiently
The synthesis of the key intermediate (Formula VI) requires strict adherence to the optimized reaction parameters outlined in the patent to ensure high yield and purity. The process begins with the catalytic hydrogenation of the starting pyranone, followed by alpha-halogenation to install the necessary leaving group. Subsequent ammoniation converts the halide to an amine, which is then subjected to the critical chiral resolution step. Once the correct enantiomer is isolated, it is protected and activated to form the final chloro-sugar intermediate. For a detailed breakdown of the specific reagents, stoichiometry, and workup procedures required to execute this synthesis in a GMP environment, please refer to the standardized protocol below.
- Reduction and Halogenation: Start with (2R, 6S)-6-methoxy-2-methyl-6H-pyran-3-one, reduce the double bond using Pd/C, and perform alpha-halogenation to introduce the leaving group.
- Ammoniation and Chiral Resolution: React the halogenated ketone with an ammoniation reagent to form the amine, then resolve the racemic mixture using chiral acids like L-tyrosine to isolate the desired enantiomer.
- Protection and Chlorination: Protect the amino group with trifluoroacetyl, followed by chlorination of the methoxy group to yield the final reactive intermediate (Formula VI).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from fermentation-based sourcing to this chemical synthesis route offers substantial strategic benefits. The primary advantage lies in the decoupling of production from biological constraints. Fermentation facilities are capital-intensive and subject to biological contamination risks that can halt production for weeks. In contrast, chemical synthesis can be performed in standard multipurpose reactors, offering greater flexibility and resilience against supply disruptions. This shift significantly enhances supply chain reliability, ensuring a steady flow of critical intermediates even during periods of high market demand or biological instability.
- Cost Reduction in Manufacturing: The elimination of fermentation and the associated downstream purification steps, such as resin column chromatography, leads to significant cost savings. Chemical synthesis typically utilizes cheaper, commodity-grade starting materials and solvents compared to the specialized media required for fermentation. Furthermore, the higher yields and purity reported in the patent reduce the volume of waste generated and the energy consumption per kilogram of product. By streamlining the process into fewer steps with simpler workups, manufacturers can achieve a leaner cost structure without compromising on quality standards.
- Enhanced Supply Chain Reliability: Relying on chemical synthesis mitigates the risks associated with biological variability. Fermentation yields can fluctuate due to strain degradation or environmental factors, whereas chemical reactions are reproducible and scalable. This consistency allows for more accurate forecasting and inventory planning. Additionally, the raw materials for this chemical route are readily available from the fine chemical sector, reducing dependency on a limited number of fermentation suppliers and diversifying the supply base for critical oncology ingredients.
- Scalability and Environmental Compliance: The mild reaction conditions described in the patent, often conducted at room temperature or with moderate heating, facilitate easier scale-up from pilot plant to commercial production. The process avoids the use of heavy metal catalysts in the resolution steps, relying instead on organic acids, which simplifies waste treatment and disposal. This aligns well with modern green chemistry principles and environmental regulations, reducing the burden on wastewater treatment facilities and lowering the overall environmental footprint of the manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. Understanding these details is crucial for evaluating the feasibility of adopting this route for your specific manufacturing needs. The answers are derived directly from the experimental data and claims presented in the patent documentation.
Q: What are the advantages of this chemical synthesis route over traditional fermentation?
A: This chemical route eliminates the reliance on fermentation-derived daunorubicin, which often suffers from low efficiency, high impurity profiles, and batch-to-batch variability. The patented method offers higher purity (up to 99.3%), milder reaction conditions, and avoids complex resin column separations.
Q: How is chirality controlled in the synthesis of the sugar moiety?
A: Chirality is strictly controlled through a resolution step using chiral reagents such as L-tyrosine, L-pyroglutamic acid, or L-dibenzoyltartaric acid. This ensures the formation of diastereomeric salts that can be separated to achieve an ee value exceeding 99.8%.
Q: Is this intermediate suitable for large-scale commercial production?
A: Yes, the process is designed for industrialization with short reaction steps, convenient post-treatment, and high yields. The use of common organic solvents and mild temperatures facilitates safe scale-up from pilot to commercial tonnage.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Epidaunorubicin Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of life-saving oncology drugs. Our team of expert process chemists has extensively analyzed the route disclosed in CN109293613B and possesses the technical capability to optimize and scale this synthesis for commercial production. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including chiral purity analysis, to guarantee that every batch meets global regulatory standards.
We invite you to collaborate with us to leverage this advanced synthetic technology for your epirubicin projects. By partnering with our technical procurement team, you can obtain a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our manufacturing expertise can drive efficiency and reliability in your supply chain.
