Advanced Synthesis of Tirofiban Intermediates: Scalable Routes for Global API Manufacturing
The pharmaceutical industry constantly seeks robust synthetic pathways for critical cardiovascular medications, and the production of Tirofiban hydrochloride stands as a prime example of this demand. Patent CN101898998B introduces a groundbreaking methodology for synthesizing 4-(4-pyridyl)-1-butanol, a pivotal intermediate in the manufacture of this potent antiplatelet agent. This specific compound serves as the structural backbone necessary for constructing the complex non-peptide thrombocyte IIb/IIIa receptor antagonist known commercially as Tirofiban. The significance of this patent lies not merely in the chemical transformation itself, but in its strategic departure from hazardous and inefficient legacy processes that have long plagued industrial production lines. By leveraging a novel salt-catalyzed de-esterification technique followed by a controlled reduction, the inventors have established a protocol that dramatically enhances safety profiles while maintaining exceptional product integrity. For global supply chain stakeholders, this represents a vital shift towards more sustainable and reliable pharmaceutical intermediate sourcing strategies.
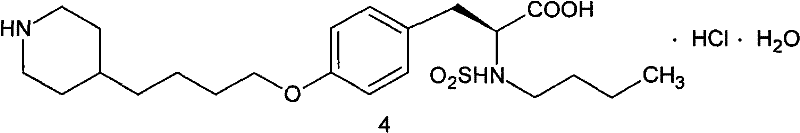
Tirofiban hydrochloride acts as a highly selective and specific antagonist for the thrombocyte IIb/IIIa receptor, offering reversible anticoagulation with a short transformation period and minimal antigenic response. Its clinical utility is profound, particularly in managing unstable angina pectoris, non-Q wave myocardial infarction, and during percutaneous coronary intervention procedures where preventing ischemic events is critical. The structural complexity of the final drug necessitates precise construction of its side chains, making the availability of high-quality precursors like 4-(4-pyridyl)-1-butanol absolutely essential. As depicted in the molecular architecture, the integration of the pyridyl-butyl chain is a defining feature that dictates the pharmacokinetic properties of the final API. Consequently, any improvement in the synthesis of this fragment directly correlates to improved efficiency in the overall drug manufacturing value chain.
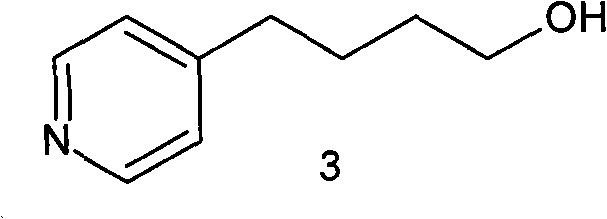
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this key pyridyl-butanol intermediate has been fraught with significant operational hazards and scalability bottlenecks that deterred widespread industrial adoption. One prominent prior art method, disclosed in CN1040534C, relies on the lithiation of 4-methylpyridine using n-Butyl Lithium, a reagent notorious for its pyrophoric nature and extreme sensitivity to moisture and air. This route necessitates cryogenic conditions and rigorous exclusion of atmospheric elements, creating substantial safety risks and demanding expensive, specialized reactor infrastructure that increases capital expenditure. Furthermore, the subsequent steps involving tetrahydropyran protection and deprotection add unnecessary complexity, extending the production cycle and introducing multiple points where yield loss can occur. Another existing pathway, described in CN100422147C, utilizes a Michael addition followed by hydrolysis and thermal decarboxylation, but this approach suffers from severe engineering challenges during the decarboxylation phase.
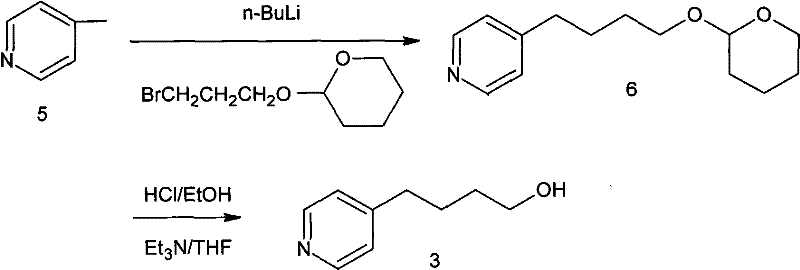
The decarboxylation step in traditional malonate-based routes is particularly problematic because it often requires heating solid reaction mixtures without solvent to drive off carbon dioxide, leading to poor heat transfer and potential runaway reactions. Additionally, the final reduction step in these legacy processes frequently employs Lithium Aluminum Hydride (LiAlH4), another hazardous reagent that poses significant fire risks and generates difficult-to-manage aluminum waste streams. These cumulative factors result in a production process that is not only costly due to low overall yields but also environmentally burdensome due to the generation of hazardous waste and the consumption of energy-intensive safety measures. For procurement managers, these inefficiencies translate into volatile pricing and unreliable delivery schedules, as manufacturers struggle to maintain consistent output under such demanding conditions. The industry urgently required a paradigm shift away from these dangerous reagents toward a more benign and operationally simple chemistry.
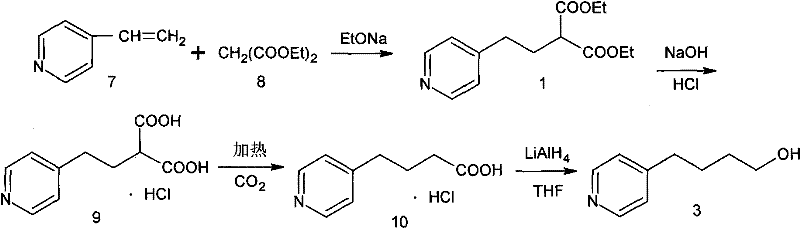
The Novel Approach
The methodology outlined in patent CN101898998B offers a transformative solution by condensing the synthesis into just two highly efficient steps starting from a diester intermediate, effectively bypassing the most dangerous and difficult stages of previous routes. Instead of relying on pyrophoric organolithium reagents or hazardous hydride reductions, this innovative process utilizes common inorganic salts such as sodium chloride or potassium chloride to catalyze the selective removal of one ester group in a polar aprotic solvent system. This de-esterification occurs smoothly in the liquid phase at elevated temperatures between 140°C and 180°C, eliminating the need for the treacherous solid-state decarboxylation that plagued earlier methods. The resulting mono-ester intermediate is obtained in high yield and, crucially, can be carried forward to the next step without rigorous purification, thereby streamlining the workflow and reducing solvent consumption. This represents a massive leap forward in cost reduction in API manufacturing by simplifying unit operations and minimizing waste generation.
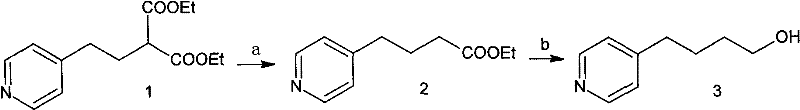
Following the de-esterification, the reduction of the mono-ester to the target alcohol is achieved using Sodium Borohydride in a mixed solvent system of lower alcohols, a stark contrast to the dangerous LiAlH4 used previously. Sodium Borohydride is significantly safer to handle, stable in alcoholic solutions, and generates manageable byproducts, which drastically lowers the barrier for safe commercial scale-up. The entire process achieves a total recovery rate exceeding 70% with a final product purity greater than 99.0% as determined by HPLC, demonstrating that safety improvements do not come at the expense of quality. By removing the need for extreme cryogenic conditions and hazardous reagents, this route facilitates the commercial scale-up of complex pharmaceutical intermediates with much greater confidence and regulatory compliance. It effectively resolves the long-standing tension between process safety and economic viability in the production of Tirofiban precursors.
Mechanistic Insights into Salt-Catalyzed Selective De-esterification
The core innovation of this patented process lies in the mechanistic nuance of using inorganic salts to promote selective hydrolysis or de-esterification within a high-boiling polar aprotic solvent like DMSO or DMF. In traditional organic synthesis, differentiating between two identical ester groups on a malonate derivative usually requires stoichiometric control of strong bases or acids, which often leads to over-hydrolysis or side reactions. However, the presence of salts like NaCl or KCl in a aqueous-DMSO mixture appears to modify the solvation shell and nucleophilicity of the water molecules, allowing for a controlled cleavage of only one ester bond under thermal conditions. This phenomenon allows the reaction to proceed at temperatures around 160°C without degrading the sensitive pyridyl ring or causing complete hydrolysis to the diacid. The mechanism likely involves the salt acting as a phase transfer catalyst or simply enhancing the ionic strength of the medium to favor the transition state for mono-hydrolysis, a subtle yet powerful manipulation of reaction kinetics. Understanding this interaction is vital for R&D directors aiming to replicate this success in other heterocyclic systems where selective functional group transformation is required.
Furthermore, the subsequent reduction mechanism utilizing Sodium Borohydride in tert-butanol and methanol ensures high chemoselectivity for the ester group while leaving the pyridine nitrogen intact. The choice of tert-butanol as a co-solvent is particularly ingenious, as it helps to solubilize the intermediate while moderating the reactivity of the borohydride species, preventing excessive gas evolution or thermal spikes. This controlled environment ensures that the reduction proceeds cleanly to the primary alcohol without reducing the aromatic pyridine ring, a common side reaction in less optimized protocols. The ability to perform this reduction on the crude mono-ester from the previous step indicates a high tolerance for residual salts and solvents, suggesting that the reaction mechanism is robust against minor impurities. This resilience is a key factor in achieving the reported high purity levels, as it minimizes the formation of complex byproduct mixtures that are difficult to separate. Such mechanistic stability is essential for maintaining batch-to-batch consistency in a GMP manufacturing environment.
How to Synthesize 4-(4-Pyridyl)-1-Butanol Efficiently
The execution of this synthesis requires careful attention to solvent ratios and temperature profiles to maximize the benefits of the salt-catalyzed de-esterification. Operators should dissolve the diester starting material in a mixture of DMSO and a small amount of water, adding the inorganic salt catalyst before heating the mixture to the optimal range of 140°C to 180°C. Monitoring the reaction via TLC or HPLC is recommended to ensure complete conversion to the mono-ester before proceeding to the workup, which involves simple extraction and concentration. The detailed standardized synthesis steps, including specific molar ratios and quenching procedures, are provided in the guide below to ensure reproducibility and safety during technology transfer.
- Dissolve the diester intermediate in DMSO or DMF with water and an inorganic salt like NaCl, then heat to 140-180°C to selectively remove one ester group.
- Isolate the resulting mono-ester intermediate, which can be used directly in the next step without further purification.
- Reduce the mono-ester using sodium borohydride in a mixed solvent system of tert-butanol and methanol or ethanol to obtain the final alcohol product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers tangible benefits that extend far beyond simple chemical yield improvements, fundamentally altering the cost structure and risk profile of the supply chain. By eliminating the need for n-Butyl Lithium and Lithium Aluminum Hydride, manufacturers can significantly reduce the costs associated with specialized storage, handling protocols, and hazardous waste disposal, leading to substantial overall cost savings. The simplification of the process from five steps down to essentially two major transformations reduces the production cycle time, allowing for faster turnaround on orders and improved responsiveness to market demand fluctuations. Moreover, the use of commodity chemicals like sodium chloride and sodium borohydride insulates the production process from the price volatility often seen with exotic organometallic reagents, ensuring more stable pricing for downstream customers. This stability is crucial for long-term supply agreements where budget predictability is a key decision-making factor for pharmaceutical buyers.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents such as n-Butyl Lithium and LiAlH4 removes the need for costly safety infrastructure and specialized waste treatment facilities. The ability to skip purification of the intermediate mono-ester saves significant amounts of solvents and processing time, directly lowering the variable cost per kilogram of the final product. Additionally, the higher overall yield means less raw material is wasted, improving the atom economy and further driving down the cost of goods sold. These efficiencies combine to create a much leaner manufacturing process that can offer competitive pricing without sacrificing margin.
- Enhanced Supply Chain Reliability: The robustness of the salt-catalyzed reaction conditions makes the process less susceptible to minor variations in raw material quality or environmental conditions, leading to fewer failed batches and more consistent output. Since the reagents used are widely available commodity chemicals, the risk of supply disruption due to vendor shortages is drastically minimized compared to routes relying on specialized organometallics. The simplified workflow also reduces the dependency on highly skilled operators for dangerous steps, making it easier to scale production across different manufacturing sites if needed. This reliability ensures that pharmaceutical companies can maintain their own production schedules without fear of intermediate shortages delaying final drug formulation.
- Scalability and Environmental Compliance: Moving from solid-state decarboxylation to a liquid-phase de-esterification solves major heat transfer issues, making the process inherently safer and easier to scale from pilot plant to multi-ton commercial production. The reduction in hazardous waste generation aligns with increasingly strict environmental regulations, reducing the compliance burden and potential liability for manufacturers. The use of safer reagents lowers the insurance premiums and safety audit costs associated with the facility, contributing to a more sustainable operational model. This environmental friendliness is becoming a key differentiator in supplier selection as global pharmaceutical companies prioritize green chemistry initiatives in their supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route, derived directly from the patent specifications and practical manufacturing considerations. These answers are designed to provide clarity on the operational advantages and safety improvements offered by this novel method compared to traditional approaches. Understanding these details is essential for technical teams evaluating the feasibility of adopting this process for their own supply chains.
Q: Why is the new salt-catalyzed de-esterification method superior to traditional decarboxylation?
A: Traditional methods often require heating solid diacids to induce decarboxylation, which creates heat transfer issues and safety risks on a large scale. The new method performs selective de-esterification in a liquid phase using common salts, ensuring better temperature control and scalability.
Q: What are the safety advantages of avoiding Lithium Aluminum Hydride (LiAlH4)?
A: LiAlH4 is pyrophoric and requires strictly anhydrous conditions, posing significant fire hazards and requiring specialized equipment. The patented process utilizes Sodium Borohydride (NaBH4), which is significantly safer, easier to handle, and compatible with alcoholic solvents, reducing operational risks.
Q: How does this process impact the purity of the final Tirofiban intermediate?
A: The optimized reaction conditions and the ability to use the crude mono-ester directly in the reduction step minimize exposure to impurities. The final distillation yields 4-(4-pyridyl)-1-butanol with HPLC purity greater than 99.0%, meeting stringent pharmaceutical standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-(4-Pyridyl)-1-Butanol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the production of life-saving cardiovascular medications like Tirofiban. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of global pharmaceutical clients. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of 4-(4-pyridyl)-1-butanol meets the highest standards of quality and consistency required for API synthesis. Our commitment to safety and efficiency mirrors the innovations found in patent CN101898998B, allowing us to deliver superior products reliably.
We invite you to contact our technical procurement team to discuss how our advanced manufacturing capabilities can support your specific project requirements. Request a Customized Cost-Saving Analysis today to understand how switching to our optimized supply chain can benefit your bottom line. We are ready to provide specific COA data and route feasibility assessments to demonstrate our capability as your trusted partner in fine chemical manufacturing. Let us help you secure a stable and cost-effective supply of this vital intermediate for your future production needs.
