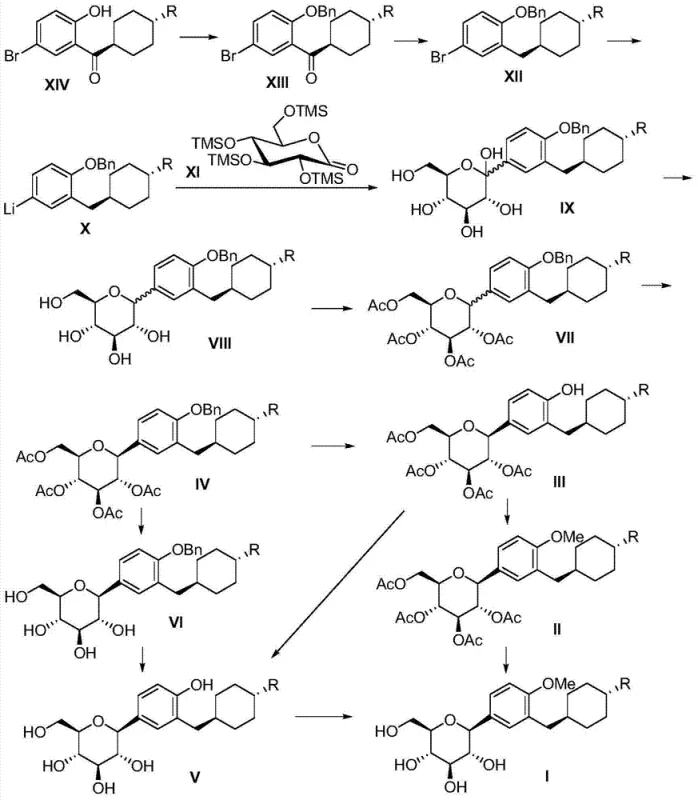
Advanced Manufacturing of High-Purity SGLT2 Inhibitor Intermediates via Optimized C-Glucoside Synthesis
Advanced Manufacturing of High-Purity SGLT2 Inhibitor Intermediates via Optimized C-Glucoside Synthesis
The global pharmaceutical landscape is increasingly focused on effective treatments for Type 2 diabetes, with SGLT2 inhibitors emerging as a critical therapeutic class due to their unique mechanism of action. Patent CN103524469A introduces a robust and innovative preparation method for (1S)-1,6-dideoxy-1-[4-hydroxy-3-(trans-4-substituted cyclohexyl)methyl phenyl]-D-glucopyranose compounds, which serve as pivotal intermediates in the synthesis of next-generation antidiabetic agents. This technology addresses significant bottlenecks in existing manufacturing protocols by optimizing the construction of the C-glycosidic bond and streamlining the protection-deprotection sequences. For R&D directors and procurement specialists, understanding this pathway is essential for securing a reliable supply of high-purity pharmaceutical intermediates that meet stringent regulatory standards. The disclosed method not only enhances chemical efficiency but also offers a scalable solution that mitigates the risks associated with complex multi-step syntheses in a commercial environment.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies, such as those disclosed in patent CN201010533301.2, have historically struggled with suboptimal conversion rates during the transformation of key precursors into the final C-glucoside structure. Specifically, the conversion of intermediate P3 to P4 in traditional routes often suffers from low productivity, which cascades into a poor overall line yield for the final target molecule. These inefficiencies are compounded by unfavorable circulation ratios and significant batch-to-batch fluctuations, creating substantial challenges for supply chain stability and cost predictability. The reliance on acid-catalyzed methyl glycoside steps in older protocols frequently leads to side reactions and difficult purification profiles, necessitating extensive chromatographic separations that are impractical for large-scale manufacturing. Consequently, manufacturers face elevated production costs and extended lead times, hindering the ability to respond rapidly to market demand for these vital diabetes therapeutics.
The Novel Approach
The inventive process detailed in CN103524469A overcomes these historical deficiencies by implementing a strategic benzyl protection group on the phenolic hydroxyl moiety early in the synthesis. This modification allows for the omission of the problematic acid-catalyzed methyl glycoside step, replacing it with a more efficient acetylation protocol that proceeds under milder conditions. By restructuring the synthetic sequence, the new method achieves a significantly higher overall yield and a superior circulation ratio, ensuring consistent output quality across multiple production batches. The route leverages robust chemical transformations that are inherently more stable and easier to control, thereby reducing the formation of impurities and simplifying downstream processing. This technological leap represents a paradigm shift in the manufacturing of SGLT2 inhibitor intermediates, offering a viable path toward cost-effective and high-volume production.
Mechanistic Insights into Lithiation and C-Glycosidic Bond Formation
The cornerstone of this synthesis is the formation of the critical carbon-carbon bond between the aromatic ring and the glucose moiety, achieved through a highly controlled organolithium reaction. The process initiates with the generation of an aryl lithium species from a bromo-substituted precursor using n-butyllithium at cryogenic temperatures of -78°C. This low-temperature condition is paramount for maintaining regioselectivity and preventing unwanted side reactions that could compromise the integrity of the sensitive functional groups present on the molecule. Once the lithiated intermediate is formed, it undergoes a nucleophilic attack on a protected D-glucono-1,5-lactone, establishing the C-glycosidic linkage with precise stereochemical control. The subsequent reduction of the resulting hemiketal using triethylsilane and boron trifluoride etherate ensures the formation of the desired alpha-anomer configuration, which is crucial for the biological activity of the final SGLT2 inhibitor.
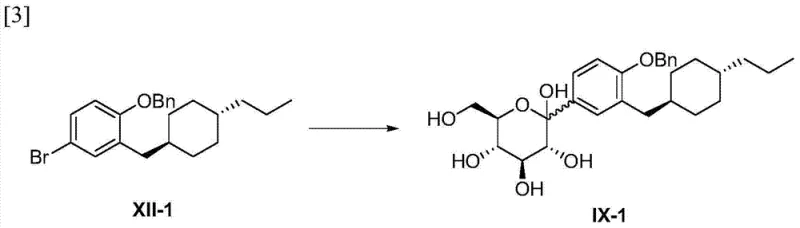
Impurity control is meticulously managed throughout the synthetic pathway through the strategic use of protecting groups and selective reaction conditions. The initial benzyl protection of the phenol prevents interference during the harsh lithiation conditions, while the acetylation of the sugar hydroxyls stabilizes the molecule against degradation during subsequent workup procedures. Furthermore, the use of specific reducing agents like triethylsilane allows for the selective reduction of the benzylic position without affecting other sensitive functionalities, thereby minimizing the generation of structural analogs. The final deprotection steps are carefully tuned to remove the benzyl and acetyl groups sequentially or concurrently without inducing epimerization or decomposition of the glucopyranose core. This rigorous attention to mechanistic detail ensures that the final product meets the high-purity specifications required for pharmaceutical applications, reducing the burden on quality control laboratories.
How to Synthesize (1S)-1,6-dideoxy-1-[4-hydroxy-3-(trans-4-substituted cyclohexyl)methyl phenyl]-D-glucopyranose Efficiently
The synthesis of this complex C-glucoside requires a disciplined approach to reaction parameters and reagent quality to ensure optimal outcomes. The process begins with the protection of the starting phenol, followed by reduction of the ketone to a methylene group to establish the trans-cyclohexyl architecture. The pivotal lithiation and coupling steps must be executed under strictly anhydrous conditions to prevent quenching of the reactive organolithium species. Following the coupling, a series of protection, reduction, and deprotection maneuvers are employed to reveal the final active pharmacophore. Detailed standardized operating procedures for each unit operation are critical for maintaining batch consistency and maximizing yield. For a comprehensive breakdown of the specific reaction conditions, stoichiometry, and workup protocols, please refer to the step-by-step guide below.
- Perform benzyl protection on the phenolic hydroxyl group of the bromo-ketone starting material using benzyl chloride and potassium carbonate in DMF.
- Reduce the ketone to a methylene group using triethylsilane and boron trifluoride etherate to form the trans-cyclohexyl methyl benzene derivative.
- Execute lithiation with n-BuLi at -78°C followed by coupling with a protected glucose lactone, then reduce and acetylate to form the final protected intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this novel synthesis route offers tangible strategic benefits that extend beyond simple chemical yield improvements. By eliminating inefficient steps and utilizing more robust reagents, the process inherently reduces the consumption of raw materials and solvents, leading to a leaner manufacturing footprint. The improved circulation ratio means that less material is lost to recycling loops or waste streams, directly translating to better resource utilization and lower variable costs per kilogram of product. Additionally, the stability of the intermediates allows for more flexible scheduling and inventory management, reducing the risk of production delays caused by unstable reagents or sensitive reaction conditions. This reliability is crucial for maintaining continuous supply lines to downstream API manufacturers who depend on timely deliveries to meet their own production targets.
- Cost Reduction in Manufacturing: The streamlined synthetic route eliminates the need for expensive and hazardous reagents associated with traditional acid-catalyzed glycosylation, resulting in substantial cost savings. By avoiding low-yield conversion steps, the overall material throughput is increased, meaning less starting material is required to produce the same amount of final product. The simplified purification processes reduce the consumption of chromatography media and solvents, further driving down operational expenditures. These efficiencies collectively contribute to a more competitive pricing structure for the final intermediate, allowing pharmaceutical partners to optimize their own cost of goods sold without compromising on quality standards.
- Enhanced Supply Chain Reliability: The use of commercially available and stable reagents such as triethylsilane and standard protecting group reagents ensures that the supply chain is not vulnerable to shortages of exotic catalysts. The robustness of the reaction conditions allows for manufacturing in a wider range of facilities, diversifying the potential supplier base and mitigating geopolitical or logistical risks. Consistent batch-to-batch performance reduces the likelihood of out-of-specification results that could halt production lines, ensuring a steady flow of materials to customers. This predictability enables long-term planning and secure contracting, which is essential for the multi-year development cycles typical of new drug candidates.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction conditions that can be safely translated from laboratory glassware to industrial reactors without significant re-engineering. The reduction in waste generation through higher yields and fewer purification steps aligns with modern green chemistry principles and environmental regulations. Minimizing the use of heavy metal catalysts or toxic solvents simplifies waste treatment and disposal, lowering the environmental compliance burden on manufacturing sites. This sustainable approach not only protects the environment but also future-proofs the supply chain against tightening regulatory frameworks regarding chemical manufacturing emissions and effluent discharge.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these SGLT2 inhibitor intermediates. The answers are derived directly from the technical specifications and experimental data provided in the patent literature, ensuring accuracy and relevance for industry professionals. Understanding these details is vital for assessing the feasibility of integrating this intermediate into your specific drug development pipeline. We encourage technical teams to review these points thoroughly to gain a deeper appreciation of the process capabilities and limitations.
Q: What represents the key improvement in this synthesis route compared to prior art?
A: The novel route eliminates the low-yield acid-catalyzed methyl glycoside conversion step found in previous methods, utilizing a benzyl protection strategy that significantly improves overall yield and circulation ratio.
Q: How is stereochemical control maintained during the C-C bond formation?
A: Stereocontrol is achieved through cryogenic lithiation at -78°C using n-BuLi, ensuring regioselective formation of the aryl lithium species before coupling with the chiral glucose lactone.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process utilizes standard reagents like triethylsilane and avoids unstable intermediates, making it highly amenable to scale-up from kilogram to metric ton quantities with consistent quality.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable SGLT2 Inhibitor Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the successful development of life-saving diabetes medications. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether you are in clinical trials or full-scale commercialization. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of (1S)-1,6-dideoxy-1-[4-hydroxy-3-(trans-4-substituted cyclohexyl)methyl phenyl]-D-glucopyranose meets the highest industry standards. Our commitment to technical excellence means we can navigate the complexities of this synthesis with precision, delivering materials that facilitate smooth downstream processing for our clients.
We invite you to engage with our technical procurement team to discuss how our optimized manufacturing capabilities can support your project goals. By requesting a Customized Cost-Saving Analysis, you can uncover specific opportunities to reduce your overall production expenses through our efficient synthesis route. We are prepared to provide specific COA data and route feasibility assessments tailored to your unique requirements, ensuring transparency and confidence in our partnership. Contact us today to secure a reliable supply of high-purity pharmaceutical intermediates that will accelerate your path to market.
